Share This Page
Drug Price Trends for DEXTROAMP-AMPHETAM
✉ Email this page to a colleague
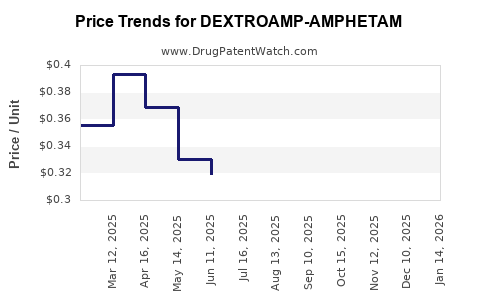
Average Pharmacy Cost for DEXTROAMP-AMPHETAM
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DEXTROAMP-AMPHETAM 12.5 MG TAB | 00555-0776-02 | 0.31653 | EACH | 2026-02-18 |
| DEXTROAMP-AMPHETAM 12.5 MG TAB | 16714-0805-01 | 0.31653 | EACH | 2026-02-18 |
| DEXTROAMP-AMPHETAM 12.5 MG TAB | 31722-0158-01 | 0.31653 | EACH | 2026-02-18 |
| DEXTROAMP-AMPHETAM 12.5 MG TAB | 11534-0193-01 | 0.31653 | EACH | 2026-02-18 |
| DEXTROAMP-AMPHETAMINE 5 MG TAB | 72516-0016-01 | 0.24545 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Dextroamphetamine-Amphetamine Market Analysis and Price Projections
This report analyzes the current market landscape and projects future pricing for dextroamphetamine-amphetamine, a stimulant medication primarily used for the treatment of Attention-Deficit/Hyperactivity Disorder (ADHD). The analysis considers patent expirations, generic competition, regulatory factors, and demand trends.
What is the Current Market Status of Dextroamphetamine-Amphetamine?
Dextroamphetamine-amphetamine, marketed under various brand names including Adderall, is a central nervous system stimulant. Its primary therapeutic application is in managing the symptoms of ADHD in children and adults. The medication's efficacy in improving focus, attention, and impulse control has established it as a cornerstone treatment for the disorder.
The market for dextroamphetamine-amphetamine is mature, characterized by a significant presence of both branded and generic formulations. Adderall, manufactured by Takeda Pharmaceutical Company, has historically dominated the market share. However, the expiration of key patents has paved the way for robust generic competition, which has exerted downward pressure on pricing.
The prevalence of ADHD has been a consistent driver of demand. According to the Centers for Disease Control and Prevention (CDC), an estimated 6.1 million children in the United States have been diagnosed with ADHD as of 2016-2019 [1]. This persistent and growing diagnosis rate ensures a steady patient pool for stimulant medications.
Key Market Participants:
- Branded Manufacturer: Takeda Pharmaceutical Company (Adderall)
- Major Generic Manufacturers: Teva Pharmaceuticals, Sandoz, Barr Pharmaceuticals (now part of Teva), Mallinckrodt Pharmaceuticals.
The market is highly regulated by the U.S. Drug Enforcement Administration (DEA) due to the Schedule II controlled substance status of amphetamines, imposing strict quotas on production and distribution [2]. This regulatory framework significantly influences supply availability.
What are the Primary Drivers and Restraints for Market Growth?
Market Drivers:
- Increasing ADHD Diagnosis Rates: Continued advancements in diagnostic methodologies and increased awareness among healthcare providers and the public contribute to a rising number of ADHD diagnoses. This directly translates to higher demand for stimulant medications.
- Off-Label Use for Narcolepsy: Dextroamphetamine-amphetamine is also FDA-approved for the treatment of narcolepsy, a chronic sleep disorder. While ADHD represents the larger market segment, narcolepsy treatment contributes to overall demand.
- Established Treatment Efficacy: Dextroamphetamine-amphetamine has a long history of clinical use and a well-documented efficacy profile, making it a preferred choice for many healthcare providers.
- Pediatric and Adult Patient Population: The sustained prevalence of ADHD across both pediatric and adult populations ensures a consistent demand for these medications.
Market Restraints:
- Strict DEA Quotas: The Schedule II classification of amphetamines subjects them to stringent DEA production quotas. These quotas can limit the overall supply of the drug, potentially leading to shortages and impacting market availability, irrespective of demand [2].
- Generic Competition and Price Erosion: The market has seen extensive generic penetration following patent expiries, leading to significant price reductions and increased competition among manufacturers.
- Side Effect Profile and Abuse Potential: Potential side effects, including cardiovascular issues, insomnia, and appetite suppression, alongside the inherent risk of misuse and addiction, can limit prescribing for some patient populations and necessitate careful monitoring.
- Development of Alternative Treatments: While stimulants remain primary treatments, the ongoing research into non-stimulant ADHD medications and alternative therapeutic approaches could, over the long term, impact market share.
What is the Patent Landscape for Dextroamphetamine-Amphetamine?
The original patents for Adderall have long expired, allowing for widespread generic manufacturing. However, specific formulations and extended-release mechanisms have been subject to subsequent patent protections.
Key Patent Milestones and Status:
- Original Composition of Matter Patents: Expired. This opened the door for generic versions of immediate-release formulations.
- Extended-Release Formulations (e.g., Adderall XR): Patents for extended-release technologies have been crucial in maintaining market exclusivity for branded products for a period. However, many of these key patents have also expired or are nearing expiration.
- For instance, Shire's (now Takeda's) Adderall XR faced patent challenges and generic approvals starting in the mid-2010s. The expiration of foundational patents related to its controlled-release technology allowed for the introduction of generic equivalents.
- Manufacturing Process Patents: These can offer some protection, but their impact on market entry for generics is typically less significant than composition of matter or formulation patents.
The genericization of dextroamphetamine-amphetamine has been a defining feature of its market trajectory. The primary patent expirations for Adderall XR were in the 2010s, leading to a surge in generic competition. This process is largely complete for the most common formulations.
Impact of Patent Expirations:
- Increased Generic Availability: Multiple manufacturers now produce generic dextroamphetamine-amphetamine, significantly expanding supply options.
- Price Compression: Generic entry is a primary driver of price reductions. The average selling price of generic dextroamphetamine-amphetamine has fallen considerably compared to the branded product before widespread genericization.
- Focus on Formulation and Delivery: With core patents expired, innovation has shifted towards novel delivery systems or combination therapies, although these often face their own patent challenges and market adoption hurdles.
How Have Regulatory Factors Impacted the Market?
Regulatory oversight by the U.S. Food and Drug Administration (FDA) and the DEA significantly shapes the dextroamphetamine-amphetamine market.
FDA Regulations:
- Approval and Post-Market Surveillance: The FDA approves all dextroamphetamine-amphetamine products, ensuring safety and efficacy standards are met. Post-market surveillance monitors for adverse events and manufacturing quality.
- Labeling and Indication Updates: The FDA mandates specific labeling requirements and can influence prescribing practices through guidance or changes to approved indications.
DEA Regulations (Controlled Substance Status):
- Schedule II Classification: Dextroamphetamine-amphetamine is classified as a Schedule II controlled substance under the Controlled Substances Act. This classification imposes strict regulations on manufacturing, distribution, prescribing, and dispensing [2].
- Production Quotas: The DEA sets annual aggregate production quotas for Schedule II substances, including amphetamines. These quotas are based on estimated medical, scientific, research, and industrial needs, as well as export requirements [3].
- Impact of Quotas: For years, DEA quotas have been cited as a significant factor contributing to shortages of stimulant medications, including dextroamphetamine-amphetamine. Manufacturers must operate within these limits, and if demand exceeds the quota, shortages can occur.
- 2023 Shortage Example: In 2023, reports indicated ongoing shortages of ADHD medications, including amphetamine products, attributed in part to DEA quota limitations and increased demand [4].
- Record Keeping and Security: DEA regulations require stringent record-keeping and security measures for all parties involved in the handling of Schedule II substances.
Impact of Regulatory Factors:
- Supply Chain Constraints: DEA quotas directly limit the total amount of dextroamphetamine-amphetamine that can be manufactured, acting as a ceiling on market supply.
- Increased Compliance Costs: Adhering to DEA and FDA regulations involves significant compliance costs for manufacturers, which can influence pricing strategies.
- Market Volatility: The potential for changes in DEA quotas or regulatory scrutiny can introduce uncertainty and volatility into the market.
- Barriers to Entry: The rigorous regulatory environment presents a high barrier to entry for new manufacturers, particularly for generic production.
What are the Current and Projected Pricing Trends for Dextroamphetamine-Amphetamine?
The pricing of dextroamphetamine-amphetamine is characterized by a significant divergence between branded and generic formulations, with ongoing downward pressure on generics due to intense competition.
Current Pricing Landscape:
- Branded (Adderall): The average wholesale price (AWP) for branded Adderall has historically been substantially higher. However, with generic availability, its market share and pricing power have diminished. Current list prices for branded Adderall can range from $200 to $500+ for a month's supply, depending on dosage and quantity, before insurance.
- Generic Dextroamphetamine-Amphetamine: Prices for generic formulations are significantly lower.
- Immediate-Release (IR): A month's supply of generic IR dextroamphetamine-amphetamine (e.g., 10mg, 30 tablets) can range from $20 to $70 out-of-pocket for cash-paying customers, with prices varying by pharmacy and dosage.
- Extended-Release (XR): Generic XR formulations are typically priced higher than IR but still considerably less than branded Adderall XR. A month's supply can range from $50 to $150 out-of-pocket.
- Wholesale Acquisition Cost (WAC) and Average Wholesale Price (AWP): These list prices are indicative, and actual transaction prices between manufacturers, wholesalers, and pharmacies are subject to rebates, discounts, and payer negotiations.
Factors Influencing Current Pricing:
- Generic Competition: The presence of numerous generic manufacturers creates a competitive pricing environment, driving down prices.
- DEA Quotas: While not directly setting prices, the supply limitations imposed by DEA quotas can, in instances of shortage, lead to temporary price spikes or increased acquisition costs for distributors and pharmacies, which may be passed on to consumers.
- Payer Contracts: Pharmaceutical benefit managers (PBMs) and insurance companies negotiate pricing with manufacturers, influencing the net price paid.
- Dosage and Formulation: Prices vary significantly based on dosage strength, tablet count, and whether the formulation is immediate-release or extended-release.
Price Projections:
The market for dextroamphetamine-amphetamine is expected to remain competitive, with pricing trends largely dictated by ongoing generic dynamics and regulatory influences.
Short-to-Medium Term (1-3 years):
- Continued Price Stability/Slight Decline for Generics: Generic dextroamphetamine-amphetamine prices are projected to remain relatively stable or experience a slight decline. Intense competition among generic manufacturers will limit significant price increases. The focus will be on market share retention rather than price hikes.
- Potential for Price Volatility due to Supply Issues: Persistent DEA quota limitations and potential manufacturing disruptions could lead to temporary price increases or out-of-stock situations, impacting availability and potentially driving up acquisition costs for pharmacies during shortages.
- Branded Product Pricing: The branded product (Adderall) will continue to occupy a niche, with pricing primarily driven by payer contracts and its specific market positioning rather than broad market trends.
Long Term (3-5+ years):
- Sustained Generic Dominance: Generics will continue to dominate the market. Pricing will remain a key competitive factor.
- Impact of New Formulations/Therapies: If significantly novel or improved ADHD therapies emerge and gain market traction, it could gradually impact the demand for traditional stimulants, indirectly influencing pricing dynamics. However, the entrenched nature of dextroamphetamine-amphetamine and the prevalence of ADHD suggest continued demand.
- Evolving Regulatory Landscape: Any shifts in DEA quota policies or FDA regulations could introduce new pricing pressures or supply dynamics. For example, a significant increase in DEA quotas could potentially alleviate supply constraints and lead to more stable pricing.
- Focus on Cost-Effectiveness: Healthcare systems and payers will continue to emphasize cost-effective treatments, supporting the long-term prevalence of generic dextroamphetamine-amphetamine at competitive price points.
Projected Price Ranges (Out-of-Pocket Estimates for 30-day supply):
- Generic IR Dextroamphetamine-Amphetamine: $20 - $60
- Generic XR Dextroamphetamine-Amphetamine: $50 - $130
- Branded Adderall (IR/XR): $200 - $500+ (highly variable with insurance)
These projections assume no major disruptions beyond current known factors like DEA quotas and standard generic competition. Significant shifts in manufacturing capacity, unforeseen regulatory changes, or the emergence of a disruptive new treatment could alter these trajectories.
What is the Competitive Landscape and Future Outlook?
The competitive landscape for dextroamphetamine-amphetamine is characterized by a mature generic market and a stable, albeit regulated, demand.
Current Competitive Dynamics:
- High Generic Penetration: The market is saturated with generic versions of dextroamphetamine-amphetamine from numerous manufacturers, including Teva, Sandoz, Mylan (now Viatris), and others.
- Price-Based Competition: With product differentiation largely eliminated for generic IR formulations, competition is primarily price-driven. Manufacturers compete on cost efficiency and distribution networks.
- Supply Chain Management: Manufacturers with robust supply chains and the ability to navigate DEA quotas effectively have a competitive advantage.
- Branded Product Niche: Takeda's Adderall and Adderall XR hold a brand premium and appeal to a segment of prescribers and patients who prefer the established brand or have specific payer coverage arrangements. However, their market share has been eroded by generics.
- DEA Quota Influence: The ability of manufacturers to secure adequate DEA production quotas is a critical competitive factor. Companies that consistently meet production targets and fulfill orders are better positioned.
Future Outlook:
The future outlook for dextroamphetamine-amphetamine remains stable, driven by the persistent prevalence of ADHD and its established therapeutic role. However, the market will continue to be shaped by regulatory constraints and generic competition.
- Sustained Demand: The long-term diagnosis trends for ADHD suggest a continued and stable demand for stimulant medications. This provides a solid foundation for the dextroamphetamine-amphetamine market.
- Regulatory Constraints as a Limiting Factor: DEA production quotas will continue to be a primary factor influencing market supply and potentially creating periodic shortages. This regulatory ceiling will prevent unchecked market expansion.
- Generic Market Dominance: The generic segment will continue to be the largest and most dynamic part of the market. Innovation within generics will likely focus on incremental improvements in delivery or formulation, but significant patent-protected innovations are unlikely for the core molecule.
- Potential for Market Consolidation: The highly competitive generic space may see further consolidation as smaller players struggle with regulatory compliance and pricing pressures.
- Focus on Supply Chain Resilience: Companies that can demonstrate reliable and consistent supply, even within quota limitations, will gain a competitive edge. This includes optimizing manufacturing processes and securing raw material inputs.
- Emergence of New ADHD Treatments: While stimulants are a first-line treatment, ongoing research into non-stimulant medications, digital therapeutics, and behavioral interventions may, over the long term, diversify ADHD treatment options and potentially shift market dynamics. However, the efficacy and rapid onset of action of amphetamines ensure their continued relevance.
The market is unlikely to experience dramatic growth spurts but rather steady demand within a tightly regulated supply environment. Key success factors will involve efficient manufacturing, strong regulatory compliance, and effective supply chain management to navigate quota limitations and meet patient needs.
Key Takeaways
- Dextroamphetamine-amphetamine's market is mature, driven by persistent ADHD diagnoses, but constrained by strict DEA production quotas.
- Original patents have expired, leading to intense generic competition that has significantly lowered prices compared to the branded product.
- DEA Schedule II classification and annual production quotas are critical factors limiting overall market supply and can lead to periodic shortages.
- Generic immediate-release formulations are projected to remain in the $20-$60 range for a 30-day supply, with extended-release versions between $50-$130.
- The competitive landscape is dominated by generic manufacturers competing primarily on price and supply reliability.
- Future market dynamics will be dictated by continued generic competition, regulatory supply limitations, and the steady underlying demand for ADHD treatment.
Frequently Asked Questions
1. What is the primary reason for shortages of dextroamphetamine-amphetamine? The primary reason for shortages of dextroamphetamine-amphetamine is the annual production quota set by the U.S. Drug Enforcement Administration (DEA) for Schedule II controlled substances. When demand exceeds the allocated quota, manufacturers cannot produce enough to meet market needs [2, 4].
2. How does the DEA quota system impact the price of dextroamphetamine-amphetamine? While DEA quotas do not directly set prices, they limit supply. When supply is artificially constrained below demand, it can lead to increased acquisition costs for distributors and pharmacies, which may then be passed on to consumers, causing temporary price spikes or increased volatility, especially during periods of shortage [2, 4].
3. Are there significant differences in efficacy between generic and branded dextroamphetamine-amphetamine? No. Generic dextroamphetamine-amphetamine products approved by the FDA are bioequivalent to their branded counterparts. This means they contain the same active ingredients, are in the same dosage form, have the same strength and route of administration, and are intended to have the same clinical effect when used under the same conditions.
4. What are the main therapeutic uses for dextroamphetamine-amphetamine? Dextroamphetamine-amphetamine is primarily approved for the treatment of Attention-Deficit/Hyperactivity Disorder (ADHD) in children and adults. It is also approved for the treatment of narcolepsy [1].
5. How has the patent expiration of Adderall XR affected its price? The expiration of key patents for Adderall XR allowed for the introduction of generic versions. This led to a significant decrease in the price of extended-release dextroamphetamine-amphetamine products, as generic manufacturers entered the market and engaged in price-based competition [5].
Citations
[1] Centers for Disease Control and Prevention. (2023, November 29). ADHD in children. Centers for Disease Control and Prevention. Retrieved from https://www.cdc.gov/ncbddd/adhd/features/adhd-in-children.html
[2] U.S. Drug Enforcement Administration. (n.d.). Drug Scheduling. U.S. Drug Enforcement Administration. Retrieved from https://www.dea.gov/drug-scheduling
[3] U.S. Drug Enforcement Administration. (2023, March 16). DEA proposes 2023 aggregate production quota for fentanyl, carfentanil, and other opioids. U.S. Drug Enforcement Administration. Retrieved from https://www.dea.gov/press-releases/2023/03/16/dea-proposes-2023-aggregate-production-quota-fentanyl-carfentanil-and-other
[4] U.S. Food & Drug Administration. (2023, October 31). Drug Shortages. U.S. Food & Drug Administration. Retrieved from https://www.fda.gov/drugs/drug-shortages/drug-shortages-last-updated-october-31-2023
[5] U.S. Food & Drug Administration. (n.d.). Orange Book Database. U.S. Food & Drug Administration. Retrieved from https://www.fda.gov/drugs/therapeutic-equivalence-approvals/orange-book-database
More… ↓
