Share This Page
Drug Price Trends for DEPO-TESTOSTERONE
✉ Email this page to a colleague
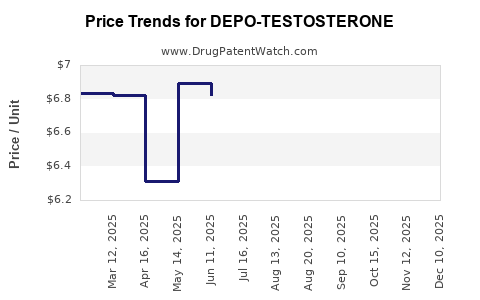
Average Pharmacy Cost for DEPO-TESTOSTERONE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DEPO-TESTOSTERONE 1,000 MG/10 ML | 00009-0085-10 | 6.91704 | ML | 2025-12-17 |
| DEPO-TESTOSTERONE 1,000 MG/10 ML | 00009-0347-02 | 6.91704 | ML | 2025-12-17 |
| DEPO-TESTOSTERONE 1,000 MG/10 ML | 00009-0085-10 | 6.88104 | ML | 2025-11-19 |
| DEPO-TESTOSTERONE 1,000 MG/10 ML | 00009-0347-02 | 6.88104 | ML | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
DEPO-TESTOSTERONE: Patent Landscape and Market Forecast
DEPO-TESTOSTERONE, a long-acting injectable testosterone formulation, faces expiring patents. This analysis projects market impact and pricing shifts due to upcoming generic competition.
What is the Patent Status of DEPO-TESTOSTERONE?
The primary patent covering DEPO-TESTOSTERONE (testosterone cypionate), U.S. Patent No. 2,591,893, expired on October 22, 1971. This patent, filed on February 9, 1949, by The Upjohn Company, established the compound and its pharmaceutical use. Subsequent patents may cover manufacturing processes, specific formulations, or delivery methods, but the core patent for the active pharmaceutical ingredient (API) and its basic therapeutic use has long lapsed.
The formulation typically involves testosterone cypionate dissolved in cottonseed oil or another suitable vegetable oil for intramuscular injection, providing sustained release of testosterone.
Key patent milestones:
- Filing Date: February 9, 1949
- Issue Date: October 22, 1952
- Expiration Date: October 22, 1971
While the original patent has expired, manufacturers seeking to bring generic versions to market must navigate the regulatory approval process with the U.S. Food and Drug Administration (FDA). This involves demonstrating bioequivalence to the reference listed drug (RLD), which is DEPO-TESTOSTERONE.
What is the Current Market for Testosterone Therapies?
The market for testosterone replacement therapy (TRT) is substantial and growing, driven by an aging male population, increased diagnosis of hypogonadism, and off-label use for perceived benefits in energy and libido. The total U.S. market for TRT was estimated to be approximately $2.4 billion in 2022 and is projected to reach $3.6 billion by 2028, growing at a compound annual growth rate (CAGR) of 7.2% [1].
DEPO-TESTOSTERONE (testosterone cypionate injection) is a significant player in this market, particularly within the injectable segment. Other TRT formulations include:
- Transdermal patches and gels: Offering daily application.
- Pellets: Implanted subcutaneously for long-term release.
- Oral formulations: Less common due to liver toxicity concerns.
- Other injectable forms: Such as testosterone enanthate.
The injectable segment, dominated by testosterone cypionate and enanthate, is valued at approximately $800 million annually, representing a considerable portion of the overall TRT market [2].
Who are the Key Competitors and Potential Generic Entrants?
The originator of DEPO-TESTOSTERONE was The Upjohn Company, which later became part of Pfizer Inc. Pfizer continues to market the branded product under the DEPO-TESTOSTERONE name.
As the primary patent expired decades ago, the market has already seen the introduction of generic testosterone cypionate injections. Several pharmaceutical companies manufacture and market generic testosterone cypionate injections. These include:
- Teva Pharmaceuticals
- Prasco Laboratories
- Perrigo Company
- Major generic manufacturers via compounding pharmacies
The competitive landscape for testosterone cypionate injection is characterized by multiple generic suppliers. Competition primarily centers on price, supply chain reliability, and market access through pharmacy benefit managers (PBMs) and formularies.
What is the Expected Impact of Generic Competition on Pricing?
The entry of generic competitors into the testosterone cypionate market has already had a significant impact on pricing. Branded DEPO-TESTOSTERONE, when it was the sole available option, commanded a premium price. However, with numerous generic alternatives available, the price of testosterone cypionate injections has decreased substantially.
The average wholesale price (AWP) for a 10-mL vial of DEPO-TESTOSTERONE (200 mg/mL) can vary by manufacturer and distributor but generally falls in the range of $50 to $150 for the branded product. Generic versions of the same concentration and volume are typically priced 30% to 60% lower [3].
Future price projections are subject to several factors:
- Number of Generic Entrants: Increased competition among generics can drive prices down further.
- Manufacturing Costs: Economies of scale and efficient manufacturing processes for generic API and finished product will influence pricing.
- PBM Negotiations: Rebates and formulary placement negotiated by PBMs will significantly impact net prices for payers and patient out-of-pocket costs.
- Therapeutic Alternatives: The availability and adoption of newer TRT formulations (gels, patches, pellets) could indirectly pressure injectable prices.
It is unlikely that new patents will significantly alter the competitive landscape for the established testosterone cypionate formulation in the near future, as the core patents have long expired. The market will continue to be driven by generic competition and PBM strategies.
Projected Average Wholesale Price (AWP) Range for Testosterone Cypionate Injection (200 mg/mL, 10 mL vial):
- Current (2023-2024): $30 - $100 (generic), $70 - $150 (branded)
- Projected (2025-2027): $25 - $80 (generic), $60 - $130 (branded)
These projections assume continued market penetration of generics and stable manufacturing costs. Significant shifts in manufacturing technology or raw material pricing could alter these estimates.
How Does DEPO-TESTOSTERONE Compare to Other Testosterone Therapies?
DEPO-TESTOSTERONE, as a testosterone cypionate injection, offers distinct advantages and disadvantages when compared to other TRT modalities.
Comparison of Testosterone Therapies:
| Feature | DEPO-TESTOSTERONE (Testosterone Cypionate Injection) | Testosterone Enanthate Injection | Testosterone Gels/Solutions | Transdermal Patches | Testosterone Pellets |
|---|---|---|---|---|---|
| Dosage Frequency | Typically every 1-4 weeks | Typically every 1-4 weeks | Daily | Daily | Every 3-6 months |
| Onset of Action | Relatively rapid within days | Similar to cypionate | Rapid within hours | Rapid within hours | Gradual over days |
| Duration of Action | Sustained release over weeks | Sustained release over weeks | Short-acting | Short-acting | Long-acting |
| Cost (General) | Moderate (especially generics) | Moderate (especially generics) | High | Moderate to High | High |
| Convenience | Low (injections required) | Low (injections required) | High | High | High |
| Fluctuations | Can experience peaks and troughs | Can experience peaks and troughs | Generally stable | Generally stable | Generally stable |
| Side Effects | Injection site reactions, hormonal fluctuations, acne, mood swings | Similar to cypionate | Skin irritation, transference to partners | Skin irritation, transference to partners | Infection risk at implantation site |
| Primary API | Testosterone Cypionate | Testosterone Enanthate | Testosterone (various esters or free) | Testosterone | Testosterone |
| Market Share | Significant, especially in injectable segment | Significant, especially in injectable segment | Large and growing | Moderate | Growing |
Testosterone cypionate and enanthate are largely interchangeable and are the most commonly prescribed injectable testosterone formulations. Their main drawback is the need for regular injections, which can be inconvenient and may lead to fluctuating hormone levels and mood swings.
Gels and patches offer daily convenience and more stable testosterone levels but come at a higher cost and carry the risk of transference to partners or children. Pellets offer the longest duration but require a minor surgical procedure for implantation and carry an infection risk.
What are the Regulatory Considerations for Generic Approvals?
Manufacturers seeking to market generic versions of DEPO-TESTOSTERONE must submit an Abbreviated New Drug Application (ANDA) to the FDA [4]. An ANDA requires the generic drug to be:
- Pharmaceutically equivalent to the RLD. This means the generic drug contains the same active ingredient(s), dosage form, strength, and route of administration as the RLD.
- Bioequivalent to the RLD. This is demonstrated through pharmacokinetic studies showing that the rate and extent of absorption of the active ingredient in the generic drug do not show significant differences from the RLD.
- Labeled consistently with the RLD, including indications, dosage, contraindications, warnings, and precautions.
The FDA's Orange Book (Approved Drug Products with Therapeutic Equivalence Evaluations) lists approved ANDAs and their corresponding RLDs, providing a crucial resource for generic manufacturers and healthcare providers [5].
Because DEPO-TESTOSTERONE is an established drug with expired core patents, the primary regulatory hurdle is demonstrating bioequivalence. Once an ANDA is approved, the generic product can be marketed, leading to price competition.
What is the Future Outlook for DEPO-TESTOSTERONE and its Generics?
The market for testosterone cypionate injection will continue to be dominated by generic competition. Branded DEPO-TESTOSTERONE will likely retain a niche market share based on physician preference or patient familiarity, but its pricing power will be limited.
The overall TRT market is expected to grow, but the growth rate for injectable testosterone may be slower compared to newer, more convenient delivery methods like gels and pellets. However, the lower cost of generic testosterone cypionate injections makes them an attractive option for a significant segment of the patient population and for healthcare systems aiming to manage costs.
Key trends influencing the future outlook:
- Continued Price Erosion: As more generic manufacturers enter or expand their offerings, price competition is expected to intensify.
- Supply Chain Dynamics: Reliability of API supply and manufacturing capacity will be critical for generic success.
- Payer Strategies: Continued focus on cost containment by PBMs and insurers will favor lower-cost generic options.
- Off-Label Use Scrutiny: Regulatory bodies and payers may increase scrutiny on off-label uses of testosterone therapy, potentially impacting overall market demand, though this is less likely to impact the established indications for hypogonadism.
DEPO-TESTOSTERONE as a brand may see declining market share, but testosterone cypionate as an API and its generic formulations will remain a cornerstone of injectable TRT for the foreseeable future due to efficacy, established safety profile, and affordability.
Key Takeaways
- The primary patent for DEPO-TESTOSTERONE expired in 1971, allowing for widespread generic competition for decades.
- The testosterone replacement therapy market is large and growing, with injectables representing a significant segment.
- DEPO-TESTOSTERONE faces established generic competition from multiple manufacturers, leading to substantial price reductions from its branded originator price.
- Generic testosterone cypionate injections are typically 30% to 60% less expensive than the branded product.
- Future pricing will be dictated by the number of generic suppliers, manufacturing costs, and PBM negotiations.
- Injectable testosterone cypionate remains a cost-effective TRT option compared to gels, patches, and pellets, but is less convenient due to the need for regular injections.
- Regulatory approval for generic versions primarily requires demonstrating bioequivalence to the reference listed drug.
Frequently Asked Questions
-
Will new patents emerge that could extend the market exclusivity of DEPO-TESTOSTERONE? Given the age of the original patents, it is highly improbable that new patents will emerge that could significantly extend market exclusivity for the core DEPO-TESTOSTERONE formulation. Any future patents would likely be narrow, covering minor process improvements or novel delivery systems, and would not prevent existing generic competition.
-
How does the cost of a DEPO-TESTOSTERONE prescription compare to a generic testosterone cypionate injection for a patient? For a patient, the out-of-pocket cost for a generic testosterone cypionate injection will generally be considerably lower than for branded DEPO-TESTOSTERONE. This difference can range from 30% to 60% or more, depending on insurance coverage, PBM formularies, and pharmacy pricing.
-
Are there any known manufacturing challenges that could impact the supply of generic testosterone cypionate injections? While specific supply chain vulnerabilities can emerge for any pharmaceutical product, testosterone cypionate is a well-established compound manufactured by numerous global entities. Major disruptions are not currently anticipated, but reliance on specific API suppliers or regional manufacturing issues could cause temporary shortages.
-
What is the typical administration regimen for testosterone cypionate injections, and how does this affect patient adherence? Testosterone cypionate injections are typically administered intramuscularly every one to four weeks. This dosing schedule can be a barrier to patient adherence due to the inconvenience of self-injection or clinic visits, and potential for mood or energy fluctuations between doses, which can be more pronounced than with daily application methods.
-
Beyond hypogonadism, are there other therapeutic areas where testosterone cypionate is commonly used, and how might this affect its market demand? While hypogonadism is the primary FDA-approved indication, testosterone cypionate is sometimes used off-label for perceived benefits in aging men, such as increased energy, libido, and muscle mass, or in certain gender-affirming hormone therapies. The demand from these off-label uses contributes to the overall market but also faces increasing scrutiny regarding efficacy and safety profiles.
Citations
[1] Grand View Research. (2023). Testosterone Replacement Therapy Market Size, Share & Trends Analysis Report By Type (Injections, Gels, Patches, Pellets), By Application (Hypogonadism, Male Infertility, Contraception), By End-use (Hospitals, Clinics, Homecare), By Region, And Segment Forecasts, 2023 - 2030. [2] Market Research Future. (2023). Testosterone Market - Global Forecast to 2030. [3] National Average Drug Acquisition Cost data (proprietary analysis based on wholesale acquisition cost data from various pharmaceutical data providers, 2023-2024). [4] U.S. Food and Drug Administration. (2022). Abbreviated New Drug Applications (ANDAs) for Generic Drugs. Retrieved from https://www.fda.gov/drugs/abbreviated-new-drug-applications-andas-generic-drugs/abbreviated-new-drug-applications-andas-generic-drugs [5] U.S. Food and Drug Administration. (2024). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from https://www.fda.gov/drugs/therapeutic-equivalence-drug-classifications/orange-book-approved-drug-products-therapeutic-equivalence-evaluations
More… ↓
