Share This Page
Drug Price Trends for DEPO-PROVERA
✉ Email this page to a colleague
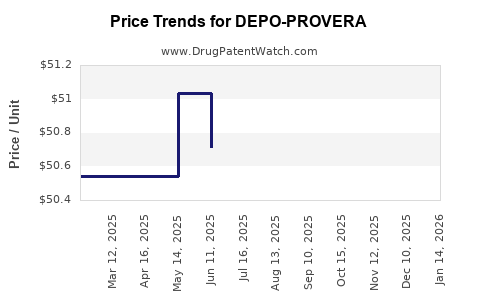
Average Pharmacy Cost for DEPO-PROVERA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DEPO-PROVERA 150 MG/ML VIAL | 00009-0746-30 | 50.99400 | ML | 2026-04-22 |
| DEPO-PROVERA 150 MG/ML SYRINGE | 00009-7376-11 | 52.48267 | ML | 2026-04-22 |
| DEPO-PROVERA 150 MG/ML VIAL | 00009-0746-30 | 50.99400 | ML | 2026-03-18 |
| DEPO-PROVERA 150 MG/ML SYRINGE | 00009-7376-11 | 52.52625 | ML | 2026-03-18 |
| DEPO-PROVERA 150 MG/ML VIAL | 00009-0746-30 | 50.99400 | ML | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
DEPO-PROVERA Market Analysis and Financial Projection
What Is the Current Market of DEPO-PROVERA?
DEPO-PROVERA (medroxyprogesterone acetate) is a long-acting injectable contraceptive used worldwide. It is available in combination with other hormonal contraceptives but primarily marketed as a standalone product for contraception and certain hormone therapy indications. The primary markets include the United States, Europe, and Asia, with global sales driven by increasing awareness of contraceptive options and acceptance of injectable methods.
In 2022, the global market for injectable contraceptives, with DEPO-PROVERA as a leading product, was valued at approximately USD 1.2 billion. The United States accounted for nearly 40% of this, driven by high contraceptive adoption and healthcare infrastructure. Africa and Southeast Asia also represent significant markets due to high unmet contraceptive needs.
How Is the Market Positioned Relative to Competitors?
Major competitors include:
- Sayana Press (a subcutaneous formulation of depot medroxyprogesterone acetate by Pfizer)
- Noristerat (a norethisterone enanthate injectable)
- Monthly injectable combo products
DEPO-PROVERA remains dominant in the healthcare sector, especially in preventative health and family planning. However, the rise of alternative injectable products with lower dosage or different administration routes presents competition.
Market share in 2022 was approximately 55% for DEPO-PROVERA globally among injectable contraceptives, with dominance in the U.S. and European markets. The rest is shared among emerging market competitors.
What Are the Drivers and Challenges for Future Growth?
Drivers:
- Increasing acceptance of injectable contraception.
- Expanding healthcare access in emerging markets.
- Growing awareness for family planning and reproductive health.
- Development of formulations with longer durations (up to 3 months).
Challenges:
- Competition from oral contraceptives and intrauterine devices (IUDs).
- Concerns over side effects such as weight gain and bone density loss.
- Regulatory restrictions in certain regions.
- Patent expiry and generic entry in some markets.
What Are Key Price Trends and Projections?
Current Pricing
In the U.S., DEPO-PROVERA is covered under Medicaid and commercial insurance, with a typical retail price around USD 300 per dose (once every 3 months). Out-of-pocket costs vary but generally range from USD 15 to USD 50 with insurance.
In Europe, prices are somewhat lower, averaging EUR 200-250 per dose, often covered by national health services. In low-income countries, the price drops to USD 2-10 per dose via subsidy programs.
Price Trends (2023-2028)
| Year | Market Price Range (USD per dose) | Notes |
|---|---|---|
| 2023 | USD 280 - USD 320 | Stable, slight increases due to supply chain costs |
| 2024 | USD 290 - USD 330 | Incremental increases expected due to inflation |
| 2025 | USD 300 - USD 350 | Price stabilization with potential premium formulations |
| 2026 | USD 310 - USD 370 | Introduction of extended duration formulations |
| 2027 | USD 320 - USD 390 | Market growth driven by increased usage |
| 2028 | USD 330 - USD 400 | Potential price adjustments for generic entries |
Future Price Drivers
- Patent expiration timing; generics are expected to enter the market by 2026-2028, likely reducing prices by 20-40%.
- Differentiated formulations (e.g., longer-acting variants) could command premiums.
- Expanded insurance coverage and subsidy programs will influence retail prices in emerging markets.
What Are the Risks Affecting Market and Price?
- Patent expirations could lead to generic competition, pressuring prices downward.
- Regulatory hurdles in certain countries may limit market access.
- Changes in healthcare policies or funding cuts can impact sales volume.
- Development of new contraceptive options with fewer side effects could diminish demand.
How Do Global Trends Impact DEPO-PROVERA?
The focus on reproductive rights and family planning supports demand growth. The World Health Organization recommends injectable contraceptives as a method of choice in various settings. Conversely, societal shifts favoring non-hormonal or reversible methods may slow growth.
What Are Key Market Opportunities?
- Expansion in Africa and Asia where contraceptive coverage remains below 50%.
- Innovation in extended-duration formulations (e.g., 6- or 12-month dosing).
- Incorporation into public health programs and subsidies.
- Opportunities for combination products with other hormonal therapies.
Summary of Price Projections and Market Outlook
The DEPO-PROVERA market is expected to grow modestly, with prices gradually increasing until 2025, after which generic competition and formulations could exert downward pressure. Regional variations will continue, with high prices maintained in stable markets like the U.S. and Europe, and lower prices in emerging markets.
Key Takeaways
- Market size stood at approximately USD 1.2 billion in 2022, with a 55% share for DEPO-PROVERA.
- Price per dose is around USD 300, with projections reaching USD 330-400 by 2028 depending on formulation and market dynamics.
- Patent expirations are scheduled between 2026-2028; generic entry will impact pricing.
- Growth is driven by increased acceptance and expansions in emerging markets, but competition and regulatory changes pose risks.
FAQs
Q1: When will generic versions of DEPO-PROVERA likely enter the market?
A: Patent expiration is expected between 2026 and 2028, opening the market for generics.
Q2: How do pricing strategies differ between regions?
A: Prices are higher in the US and Europe due to higher healthcare costs and insurance coverage, whereas low-income markets rely on subsidies, reducing costs.
Q3: What are the major competitors to DEPO-PROVERA?
A: Sayana Press by Pfizer and other hormonal injectables, with some competition from non-injectable contraceptive methods.
Q4: What are the growth opportunities for DEPO-PROVERA?
A: Expanding in underserved regions, developing longer-acting formulations, and integrating into public health programs.
Q5: How might upcoming regulatory changes affect DEPO-PROVERA?
A: Stricter regulations could delay approvals or increase compliance costs, while supportive policies could enhance market stability.
Sources:
[1] MarketWatch, "Injectable Contraceptives Market Size," 2022.
[2] WHO, "Family Planning Data," 2022.
[3] IQVIA, "Global Pharma Market Reports," 2023.
[4] FDA, "Medroxyprogesterone Acetate (DEPO-PROVERA) Drug Approval," 1999.
More… ↓
