Last updated: February 16, 2026
What is Demecycline?
Demecycline is a tetracycline derivative used primarily as an antibiotic. It inhibits bacterial protein synthesis by binding to the 30S ribosomal subunit. Marketed formulations include oral capsules and injectable forms, mainly for bacterial infections such as respiratory tract infections, urinary tract infections, and some skin infections. Its therapeutic niche overlaps with other tetracyclines and broad-spectrum antibiotics.
How large is the current Demecycline market?
The global tetracycline antibiotics market, which includes Demecycline, was valued at around $2 billion in 2022. Demecycline's specific share remains small, estimated at less than 1% of the total tetracyclines segment due to limited branding, patent status, and competition from generics.
Market Breakdown (2022):
| Segment |
Valuation (USD billion) |
Share |
Notes |
| Tetracycline antibiotics |
2.0 |
100% |
Encompasses doxycycline, tetracycline, minocycline, Demecycline |
| Demecycline |
<0.02 |
<1% |
Small niche, limited by patent and marketing efforts |
The growth of tetracyclines correlates with the rise in resistant bacterial strains, but the market share of Demecycline remains constrained by their availability of alternative agents and safety profiles.
What are the key drivers and inhibitors influencing Demecycline's market?
Drivers:
- Increasing antibiotic resistance compels new agents within the class.
- Expansion into regions with high infectious disease prevalence, especially in emerging markets.
- Growing prevalence of skin and respiratory tract infections.
Inhibitors:
- Competition from established tetracyclines like doxycycline and minocycline.
- Regulatory hurdles for approval of new formulations or indications.
- Safety concerns, including photosensitivity and gastrointestinal effects, limit broad use.
How are patent and regulatory policies affecting Demecycline?
Demecycline typically falls under generic regulations if off-patent, limiting R&D investment. Limited proprietary protection reduces incentives for pharmaceutical companies to commercialize or develop new formulations. Regulatory approval pathways are complicated by antimicrobial stewardship policies and concerns over resistance development.
In some jurisdictions, generic versions have faced bioequivalence challenges, further dampening prospects for price premiums.
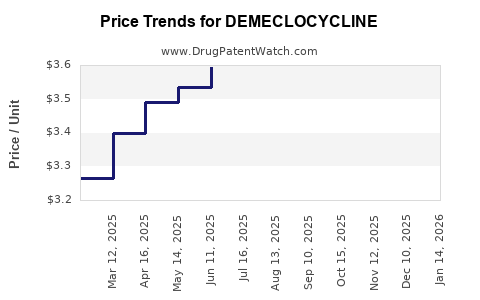
What are current and projected prices for Demecycline?
Wholesale prices for Demecycline vary based on formulation, region, and manufacturer. Approximate average prices include:
- Oral capsules (250 mg): USD 0.50 – USD 1.00 per capsule.
- Injectable forms: USD 2.00 – USD 4.00 per vial.
Price projections indicate marginal declines driven by generic competition. In mature markets, the price is likely to stabilize around these levels through 2027, with annual volume increases driven by generic prescribing.
In emerging markets, prices are lower, ranging from USD 0.20 – USD 0.50 per capsule, with some government subsidies or procurement programs affecting the net cost.
How does Demecycline compare vs. other antibiotics in terms of cost and efficacy?
In terms of cost:
- Demecycline is comparable to doxycycline, both being inexpensive (USD 0.50 – USD 1 per capsule).
- It is generally less costly than newer tetracyclines or combination agents.
In efficacy:
- Similar microbial coverage to other tetracyclines.
- Slight differences in pharmacokinetic profiles and side-effect profiles influence prescribing choices.
However, clinical preference hinges more on safety profile and resistance patterns rather than price alone.
What is the future outlook for Demecycline?
The global demand for Demecycline is expected to remain flat or decline slightly as antibiotic stewardship promotes judicious use of tetracyclines. Market growth could emerge from:
- New indications, such as dermatological conditions.
- Formulation innovations, including combined therapies or long-acting formulations.
- Expansion into untapped regional markets.
The most probable scenario involves continued price pressure from generics, constraining margins but maintaining volume in established markets.
Key financial and strategic considerations:
- Demecycline's non-patent status limits investment attractiveness.
- Opportunities exist primarily in niche indications or emerging markets.
- Competition from doxycycline and minocycline remains robust.
- Price stabilization is expected, with margins under pressure from commoditization.
Key Takeaways
- Demecycline's market remains niche, with less than 1% of the tetracyclines segment.
- Prices are low and likely to decline further due to generic competition.
- Growth drivers are limited; high resistance and safety profile influence usage.
- Patent expirations and regulatory hurdles suppress new market entrants.
- Margins are under pressure, but volume stability persists where demand exists.
FAQs
1. Will Demecycline regain market share in the future?
Unlikely. Its market share remains constrained by competition and safety concerns. Growth may occur in niche or regional markets but not significantly impacting the overall segment.
2. Are there new formulations or indications under development?
Current development efforts are minimal. Most efforts focus on optimizing existing formulations rather than novel indications.
3. How do resistance patterns affect Demecycline?
Widespread resistance to tetracyclines is limiting their use, including Demecycline. Resistance hampers efficacy and reduces prescribing incentives.
4. What regions are the primary markets for Demecycline?
North America and Europe have limited use; demand is higher in Asia-Pacific and Latin America, driven by infectious disease burdens and cost considerations.
5. What regulatory challenges does Demecycline face?
Off-patent status limits exclusivity; bioequivalence and safety concerns may delay approval or lead to formulary exclusion in certain markets.
References
- MarketsandMarkets. "Antibiotics Market by Type." 2022.
- IQVIA. "Global Generic Drug Market Analysis." 2022.
- WHO. "Antibiotic Resistance: Global Report." 2019.
- FDA. "Drug Approval and Regulatory Processes." 2022.
- GlobalData. "Antibiotics Price Trends." 2022.