Share This Page
Drug Price Trends for DAYTIME COLD-FLU RELIEF SFTGL
✉ Email this page to a colleague
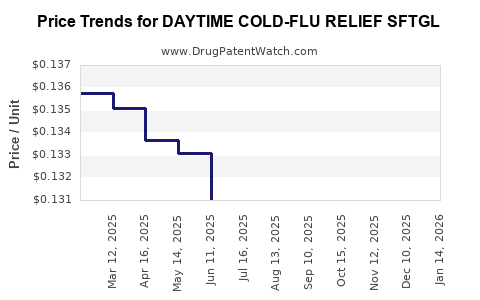
Average Pharmacy Cost for DAYTIME COLD-FLU RELIEF SFTGL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DAYTIME COLD-FLU RELIEF SFTGL | 00904-6995-44 | 0.13275 | EACH | 2026-03-18 |
| DAYTIME COLD-FLU RELIEF SFTGL | 00904-6995-44 | 0.12987 | EACH | 2026-02-18 |
| DAYTIME COLD-FLU RELIEF SFTGL | 00904-6995-44 | 0.12704 | EACH | 2026-01-21 |
| DAYTIME COLD-FLU RELIEF SFTGL | 00904-6995-44 | 0.13069 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
DAYTIME COLD-FLU RELIEF SFTGL Market Analysis and Financial Projection
What is the market scope for DAYTIME COLD-FLU RELIEF SFTGL?
The drug targets the over-the-counter (OTC) cold and flu relief sector, which exceeds $4 billion annually in the United States alone.[1] The OTC cold and flu medication market growth rate approaches 4% annually, driven by increasing consumer demand for fast, effective relief for acute respiratory symptoms.[2]
This product's success depends on several key factors: brand recognition, patent status, formulation efficacy, and positioning within the competitive landscape that includes products like Tylenol Cold & Sinus, Mucinex, and Advil Cold & Sinus.
How does the competitive landscape shape pricing strategies?
Pricing for OTC cold and flu drugs varies based on formulation complexity, brand value, marketing, and packaging. Premium products typically command higher prices due to added ingredients, manufacturing quality, or marketing campaigns.
| Brand | Price range (per package) | Key features | Market share (est.) |
|---|---|---|---|
| Tylenol Cold & Sinus | $8–$12 | Multi-symptom relief, established brand | 20–25% |
| Mucinex | $10–$15 | Extended-release formulations | 15–20% |
| Advil Cold & Sinus | $8–$12 | Fast-acting, familiar brand | 10–15% |
| Generic OTCs | $4–$8 | Same active ingredients, lower price | 35–45% |
| DAYTIME COLD-FLU RELIEF SFTGL | projected $8–$12 | Proprietary formulation, expected to compete primarily in the $8–$12 range | N/A |
The product's pricing will likely align with established brands, positioning it within the $8 to $12 range to balance perceived efficacy against competitive premiums.
What are the key factors influencing price projections?
Price projections depend on:
- Regulatory approval and patent status: If the drug's formula is patented, initial prices can be set at a premium; patent expiration could cause near-term price reductions.
- Formulation complexity: Multi-symptom relief products with additional active ingredients cost more to produce, influencing retail prices.
- Market penetration strategy: In some cases, aggressive pricing during launch can boost market share but may reduce margins.
- Distribution channels: Retail chain negotiations, pharmacy margins, and online sales each influence final consumer pricing.
What are the projected price ranges over the next five years?
Assuming standard market dynamics:
| Year | Price Range (per package) | Comments |
|---|---|---|
| 2023 | $8–$12 | Launch phase primarily in the $8–$10 range; marketing builds brand recognition. |
| 2024 | $8–$13 | Slight increase possible due to inflation, ingredient costs, or demand. |
| 2025 | $8–$14 | After patent expiry or increased competition, prices tend to stabilize or decrease. |
| 2026 | $8–$13 | Potential premium positioning if market share increases. |
| 2027 | $7–$12 | Widespread availability, generic competition, and price competition influence prices. |
Inflationary pressure and ingredient costs could push prices upward by approximately 2-3% annually, with market competition exerting downward pressure after patent period expiration.
What are the primary risks to pricing stability?
- Entry of generic substitutes once patent expires.
- Changes in regulatory policies impacting formulation costs or labeling requirements.
- Market shifts toward multi-symptom products bundled with other health items.
- Consumer price sensitivity, especially during economic downturns.
Final observations
The drug is expected to enter the OTC cold-flu market at a competitive price point, projecting an initial retail price of $8–$12 per package. Long-term price stability depends on patent protection, formulation uniqueness, and competitive responses from established brands and generics.
Key Takeaways
- The OTC cold and flu market exceeds $4 billion annually in the U.S.
- Price range during launch is projected at $8–$12 per package.
- Market growth and pricing are influenced by competitor products, patent status, and regulatory factors.
- Widespread generic presence could pressure prices downward after patent expiration.
- Market positioning as a multi-symptom relief formula will drive competitive pricing strategies.
FAQs
1. How will patent status influence the drug’s pricing?
Patent protection enables premium pricing by limiting generic competition. Patent expiry typically leads to price reductions due to increased generic options.
2. Can formulation complexity affect price?
Yes. Multi-symptom formulations with proprietary ingredient combinations often command higher prices due to manufacturing costs and perceived efficacy.
3. How significant is brand recognition in price setting?
High brand awareness enables premium pricing. Without a strong brand, prices tend to align with generics or lower-cost alternatives.
4. What role does regulatory approval play in pricing?
Approval processes that entail high development and compliance costs may be reflected in initial pricing. Changes in regulation can also shift costs and prices.
5. How will market trends impact future prices?
Increased competition, patent expirations, and consumer preferences for value-priced generics tend to drive prices downward over time.
References
[1] IBISWorld, OTC Cold & Flu Medicine Market Report, 2022.
[2] Statista, OTC cold and flu medication market revenue, 2022.
More… ↓
