Share This Page
Drug Price Trends for DALFAMPRIDINE ER
✉ Email this page to a colleague
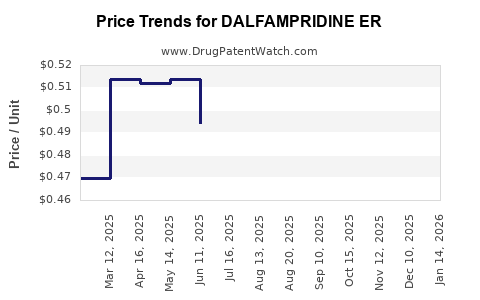
Average Pharmacy Cost for DALFAMPRIDINE ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DALFAMPRIDINE ER 10 MG TABLET | 16729-0292-12 | 0.38591 | EACH | 2026-03-18 |
| DALFAMPRIDINE ER 10 MG TABLET | 67877-0444-60 | 0.38591 | EACH | 2026-03-18 |
| DALFAMPRIDINE ER 10 MG TABLET | 82249-0702-60 | 0.38591 | EACH | 2026-03-18 |
| DALFAMPRIDINE ER 10 MG TABLET | 42571-0275-60 | 0.38591 | EACH | 2026-03-18 |
| DALFAMPRIDINE ER 10 MG TABLET | 00591-2533-60 | 0.38591 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Dalfampridine ER
Overview
Dalfampridine extended-release (ER), marketed as Amifampridine in some regions, is primarily indicated for multiple sclerosis (MS)-related walking impairment. The drug gained FDA approval in 2010 under the brand name Ampyra, designed to improve ambulatory function in MS patients.
Market Size and Demand Drivers
The global multiple sclerosis market was valued at approximately $22.2 billion in 2022, with a compound annual growth rate (CAGR) of 4.5% projected through 2030 [1]. Dalfampridine ER directly addresses gait impairment, affecting roughly 50% of the MS population, estimated at 2.5 million globally. Assuming 50% of these patients experience walking difficulties, the addressable population for Dalfampridine ER could reach 625,000 globally.
Key demand factors include:
- If approved for new indications, such as other neurological conditions involving gait impairments, demand could expand.
- Reimbursement landscapes significantly influence access; in the U.S., Medicare and Medicaid reimburse Dalfampridine ER, encouraging prescribing.
- Clinician familiarity with Dalfampridine ER has stabilized, with continuous positive data supporting safety and efficacy.
Competitive Landscape
The market is dominated by Ampyra, with generic formulations entering since patent expiry in 2018. Major competitors include:
- Generic Dalfampridine ER manufacturers offering lower prices.
- Other symptomatic treatments, such as physical therapy, don't compete directly but influence overall market size.
Pricing Landscape
Brand-name Ampyra's retail price in the U.S. has historically been around $4,000-$5,000 per month [2]. After patent expiration, generic versions reduced the price to approximately $700-$1,200 per month.
- Generic Dalfampridine ER average wholesale prices (AWP) range from $750 to $1,200 per month depending on packaging and supplier.
- Pricing trends are expected to stabilize or decline further due to increased generic competition.
Price Projections
Considering current market trends, the following projections are derived:
-
2023-2024:
Generic prices will hover between $700 and $1,200 per month, driven by competition. Brand prices are mostly phased out outside specific regions or therapeutic niches. -
2025-2027:
Slight decline anticipated due to continued generic penetration. Projected average prices for generics: $600-$900 per month. -
Long-term outlook:
Price stabilization at $500-$800 per month as the market reaches saturation. Potential for slight decrease if biosimilar versions or improved formulations emerge.
Regional Variations
Price and access vary globally:
- United States: High prices initially due to brand dominance; prices have decreased with generics.
- Europe: Reimbursement policies contain costs, but prices remain akin to U.S. levels for generics.
- Emerging Markets: Lower prices ($200-$400 per month), limited access due to reimbursement and healthcare infrastructure constraints.
Regulatory and Policy Influences
Patent expiry in 2018 opened the floodgates for generics. Policies favor generics to reduce healthcare costs, influencing market pricing downward.
Conclusion
The Dalfampridine ER market is characterized by a declining price trend driven by generic entry. Future prices are expected to stabilize near $600 per month for generics, with potential reductions to $500 per month depending on competition and manufacturing efficiencies.
Key Takeaways
- The current market predominantly features generics priced between $700 and $1,200 monthly.
- Demand is primarily driven by MS patients with gait impairment, estimated at 625,000 globally.
- Price declines are forecasted through 2027, with stabilization around $500-$900 monthly for generics.
- Regulatory policies support price reduction, impacting the profitability of branded versions.
- Emerging markets may see significantly lower prices but face access barriers.
Frequently Asked Questions
1. How does patent expiration impact Dalfampridine ER pricing?
Patent expiry in 2018 enabled generic manufacturers to enter the market, causing prices to decrease from original brand levels of $4,000-$5,000 monthly to approximately $700-$1,200.
2. Are there approved alternative drugs for MS-related gait impairment?
Currently, Dalfampridine ER is the primary approved drug for walking impairment in MS. No other drugs with equivalent efficacy are available; physical therapy remains complementary.
3. Could new indications revive the market or affect prices?
Yes. If regulatory agencies approve Dalfampridine ER for additional indications involving gait or neurological disorders, demand could increase, potentially supporting higher prices temporarily.
4. What are the reimbursement challenges affecting market pricing?
Reimbursement policies vary; in the U.S., Medicare and Medicaid cover Dalfampridine ER, supporting access but also incentivizing cost containment, which influences pricing trends.
5. How does regional variation influence market potential?
In high-income countries, prices are higher but market access is better. Emerging economies have lower prices but limited access, constraining overall revenue growth.
Sources:
[1] Grand View Research, "Multiple Sclerosis Market Size & Trend" (2022).
[2] Drugs.com, Ampyra Price Data (2023).
More… ↓
