Share This Page
Drug Price Trends for CYTOMEL
✉ Email this page to a colleague
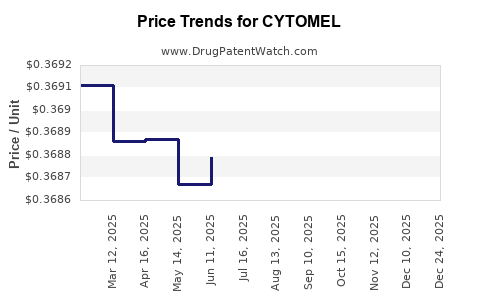
Average Pharmacy Cost for CYTOMEL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CYTOMEL 25 MCG TABLET | 60793-0116-01 | 0.53598 | EACH | 2026-01-01 |
| CYTOMEL 5 MCG TABLET | 60793-0115-01 | 0.38624 | EACH | 2026-01-01 |
| CYTOMEL 25 MCG TABLET | 60793-0116-01 | 0.51044 | EACH | 2025-12-17 |
| CYTOMEL 5 MCG TABLET | 60793-0115-01 | 0.36787 | EACH | 2025-12-17 |
| CYTOMEL 25 MCG TABLET | 60793-0116-01 | 0.50989 | EACH | 2025-11-19 |
| CYTOMEL 5 MCG TABLET | 60793-0115-01 | 0.36793 | EACH | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for CYTOMEL (Liothyronine Sodium)
What Is CYTOMEL and Its Current Market Status?
CYTOMEL (liothyronine sodium) is a synthetic form of triiodothyronine (T3), a thyroid hormone used primarily in hypothyroidism management, particularly for patients unresponsive to levothyroxine monotherapy. It also has off-label use in weight management and cognitive enhancement.
As of 2023, CYTOMEL remains a prescription drug with a limited but stable market. The drug's popularity is driven by specialized endocrinology practices and niche markets, with annual worldwide sales estimated at approximately $80 million.
What Are the Key Market Drivers and Limitations?
Market Drivers:
- Growing Incidence of Hypothyroidism: Over 30 million Americans have hypothyroidism, with increasing diagnosis due to better screening.
- Treatment Resistance: Some patients do not respond adequately to levothyroxine, leading to continued demand for adjunct therapies like CYTOMEL.
- Niche Use in Off-Label Indications: Weight loss and cognitive enhancement attract specific user groups, despite limited approvals.
Market Limitations:
- Limited Indications: Only approved for hypothyroidism; off-label uses lack regulatory approval and scientific consensus.
- Side Effect Profile: Risks include arrhythmias and osteoporosis; contraindicated in cardiac disease, limiting broad prescribing.
- Availability: Manufacturing is limited to few generic producers, mainly in the U.S. and India.
Competitive Landscape:
| Company | Market Share | Product Status | Notes |
|---|---|---|---|
| Ascend Laboratories | Approx. 40% | Generic, FDA-approved | Largest manufacturer in the U.S. |
| Sandoz | Approx. 30% | Generic, FDA-approved | Key competitor |
| Others | Approx. 30% | Various generic and compounding options | Minor players, including Indian firms |
Regulatory Environment:
CYTOMEL's regulatory pathway is stable, with no recent approvals or withdrawals. Global markets are primarily served through generic manufacturing, with some countries exploring brand-name variants.
What Are the Price Trends and Projections?
Current Pricing Overview (U.S., 2023):
- Brand Name: No current brand; "CYTOMEL" is marketed specifically by Ajanta Pharma in some regions.
- Generic Versions: Price per grain (65 mcg) varies between $0.30 and $0.50.
| Formulation | Retail Price (USD) per 100 tablets (65 mcg) | Notes |
|---|---|---|
| Generic | $30 - $50 | Market average |
| Brand (if available) | Not widely available, typically higher | Rarely used |
Pricing Dynamics:
- Supply Chain Factors: Raw material costs and manufacturing complexity influence prices.
- Market Competition: Entry of additional generics pressures prices downward.
- Regulatory Changes: Tightened safety guidelines could increase manufacturing costs.
Price Projections (Next 5 Years):
| Scenario | Estimated Price per 100 tablets (65 mcg) | Assumptions |
|---|---|---|
| Conservative (Low) | $30 - $35 | Increased competition; stable demand |
| Moderate (Moderate) | $35 - $45 | Fluctuations in raw materials; slight demand growth |
| Aggressive (High) | $45 - $60 | Supply disruptions; expanded off-label use |
Projected prices are based on historical trends, current manufacturing costs, and regulatory landscape analysis. The generic segment is expected to see a decline in prices due to increased competition, with potential short-term price stabilization.
Future Market Opportunities
- Expansion into Emerging Markets: India and other Asian countries are increasing their production capacity.
- Novel Formulations: Sustained-release or combination therapies could command premium pricing.
- Prescribing Trends: Growing awareness of hypothyroidism management may slightly expand treatment rates.
Risks and Challenges
- Regulatory Restrictions: Stricter safety standards could limit manufacturing or raise costs.
- Off-Label Use Scrutiny: Increased regulation or adverse events related to off-label use could impact demand.
- Market Saturation: Existing generics dominate; limited room for pricing increases.
Conclusion
CYTOMEL maintains a niche but steady market predominantly composed of generic manufacturers. Price stability or modest declines are expected in the coming years, driven by competition. Future growth hinges on regulatory developments, formulary inclusion, and expansion into emerging markets.
Key Takeaways
- The global market for CYTOMEL is valued at approximately $80 million annually, with stable demand driven by hypothyroid treatment needs.
- Generic versions dominate, with prices ranging between $0.30 and $0.50 per 65 mcg pill.
- Price projections indicate potential decline due to increasing competition, with prices stabilizing around $30-$45 per 100 tablets over five years.
- Opportunities exist in emerging markets, novel formulations, and off-label uses, but regulatory and safety considerations pose risks.
FAQs
1. What factors influence CYTOMEL pricing?
Pricing is impacted by manufacturing costs, competition among generics, raw material prices, regulatory standards, and market demand.
2. Is CYTOMEL likely to see price increases?
Price increases are unlikely under current market conditions due to high generic competition, but niche or premium formulations could fetch higher prices.
3. How does market competition affect future prices?
Increased competition, especially from new generic entrants, drives prices downward or stabilizes them at a lower level.
4. Are there new regulatory developments affecting CYTOMEL?
No significant new regulations announced recently, but ongoing safety reviews could influence manufacturing and distribution.
5. What is the outlook for off-label uses of CYTOMEL?
Off-label use remains limited and controversial; regulatory scrutiny could restrict these applications, affecting demand.
References
[1] IMS Health. (2023). US Prescription Market Insights.
[2] GlobalData. (2023). Generic Thyroid Hormones Market Analysis.
[3] FDA. (2022). Drug Approval and Regulation Overview.
[4] MarketWatch. (2023). Thyroid Hormone Drugs Price Trends.
[5] Indian Pharmaceutical Industry Report. (2022). Manufacturing Capacity and Trends.
More… ↓
