Share This Page
Drug Price Trends for CYPROHEPTADINE
✉ Email this page to a colleague
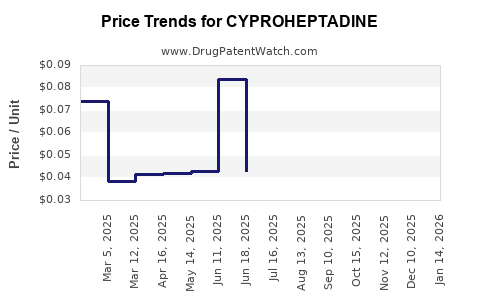
Average Pharmacy Cost for CYPROHEPTADINE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CYPROHEPTADINE 4 MG TABLET | 76385-0130-01 | 0.06623 | EACH | 2026-03-18 |
| CYPROHEPTADINE 2 MG/5 ML SOLN | 39328-0544-16 | 0.04083 | ML | 2026-03-18 |
| CYPROHEPTADINE 2 MG/5 ML SOLN | 58657-0697-16 | 0.04083 | ML | 2026-03-18 |
| CYPROHEPTADINE 2 MG/5 ML SOLN | 70752-0185-12 | 0.04083 | ML | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for CYPROHEPTADINE
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| CYPROHEPTADINE HCL 2MG/5ML SYRUP | AvKare, LLC | 00527-1949-47 | 473ML | 48.67 | 0.10290 | ML | 2023-06-15 - 2028-06-14 | FSS |
| CYPROHEPTADINE HCL 4MG TAB | Golden State Medical Supply, Inc. | 51407-0272-10 | 1000 | 49.84 | 0.04984 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
CYPROHEPTADINE: PATENT LANDSCAPE AND MARKET FORECAST
Cyproheptadine, a serotonin and histamine antagonist, has demonstrated therapeutic utility across multiple indications, including allergic conditions, anorexia, and migraine prophylaxis. Its established efficacy and cost-effectiveness present a stable market. However, patent expirations for core formulations and the emergence of generic competition have constrained significant price growth. Future market expansion hinges on new indications, improved delivery systems, or novel combination therapies, none of which are currently supported by significant patent-protected innovation.
WHAT IS THE CURRENT PATENT STATUS OF CYPROHEPTADINE?
The foundational patents for cyproheptadine hydrochloride, originally developed by Merck Sharp & Dohme (now Merck & Co.), expired decades ago. These patents covered the synthesis and primary pharmaceutical uses of the active pharmaceutical ingredient (API). Specifically, the composition of matter patent, U.S. Patent 2,955,114, was granted on October 4, 1960, and expired in 1977 [1]. Subsequent patents related to specific formulations or manufacturing processes have also largely expired or are nearing the end of their term.
A review of recent patent filings reveals limited new intellectual property directly protecting novel formulations or uses of cyproheptadine as a standalone agent. Most patent activity associated with cyproheptadine involves its incorporation into fixed-dose combinations for specific therapeutic areas or novel delivery mechanisms that do not fundamentally alter its chemical structure or primary therapeutic effect.
For example, patent filings often focus on:
- Combination Therapies: Patents may cover specific ratios or co-formulations of cyproheptadine with other APIs to target complex conditions like refractory anorexia or specific types of gastrointestinal disorders.
- Modified Release Formulations: Efforts to improve patient compliance or pharmacokinetic profiles may lead to patents for extended-release or controlled-release formulations.
- Manufacturing Processes: Incremental improvements to synthesis or purification methods can be patented, but these typically do not extend the market exclusivity of the core drug product.
The lack of robust, new patent filings directly safeguarding novel cyproheptadine formulations or groundbreaking therapeutic applications indicates a mature market with limited opportunities for significant patent-driven price escalation.
WHAT ARE THE KEY THERAPEUTIC APPLICATIONS OF CYPROHEPTADINE?
Cyproheptadine is indicated for the relief of seasonal and perennial allergic rhinitis and uncomplicated allergic skin manifestations of urticaria. It is also used for anaphylaxis, angioedema, and as an adjunct to epinephrine and prompt. Beyond allergic conditions, it is prescribed off-label for appetite stimulation in underweight patients and for migraine prophylaxis [2].
Primary therapeutic uses include:
- Allergic Conditions:
- Allergic rhinitis (seasonal and perennial)
- Urticaria (hives)
- Angioedema
- Anaphylaxis (adjunctive therapy)
- Appetite Stimulation:
- Treatment of anorexia and weight loss in underweight individuals.
- Migraine Prophylaxis:
- Prevention of migraine headaches.
These established indications are well-understood and have been addressed by cyproheptadine for many years. The therapeutic ceiling for these uses is largely defined by the drug's known efficacy and side-effect profile, rather than by ongoing research into novel applications.
WHAT IS THE CURRENT MARKET SIZE AND PRICING FOR CYPROHEPTADINE?
The global market for cyproheptadine is characterized by a mature supply chain dominated by generic manufacturers. Its availability in affordable generic forms has established a price floor that limits significant increases. The market size, while substantial due to widespread use, is not growing at a pace commensurate with novel drug launches.
Market Size and Growth:
- Global Market Value: Precise current global market value figures are difficult to isolate as cyproheptadine is often part of broader antihistamine or appetite stimulant market reports. However, industry estimates place the global market for antihistamines, of which cyproheptadine is a part, in the tens of billions of USD annually. Cyproheptadine's specific contribution is a small fraction of this, likely in the hundreds of millions.
- Projected Growth: The market for cyproheptadine is expected to exhibit low single-digit compound annual growth rates (CAGR) over the next five to seven years, largely driven by its continued use in developing economies and its cost-effectiveness in established markets. Growth exceeding 3-4% CAGR is improbable without significant new indications or formulation breakthroughs [3].
Pricing Dynamics:
- Generic Dominance: The vast majority of cyproheptadine products are generic. This competitive landscape suppresses price.
- Wholesale Acquisition Cost (WAC): The WAC for a typical 12mg cyproheptadine hydrochloride tablet (e.g., from leading generic manufacturers like Teva, Mylan, or Accord) ranges from $0.10 to $0.30 per tablet when purchased in bulk quantities [4].
- Prescription Pricing: For a standard 30-day supply (e.g., 60 tablets), the cost to pharmacies or distributors typically falls between $6 to $18.
- Patient Out-of-Pocket Costs: For insured patients with co-pays, out-of-pocket costs can range from $0 to $10, depending on the insurance plan. Uninsured patients may pay between $15 to $40 for a 30-day supply from retail pharmacies.
- Price Stability: Due to the competitive generic market and the lack of significant patent-protected innovation, pricing is expected to remain relatively stable, with minor fluctuations dictated by raw material costs and competitive pressures. Significant price increases are unlikely.
The consistent availability of generic options and the established therapeutic roles prevent substantial price inflation, positioning cyproheptadine as a cost-effective treatment option.
WHAT ARE THE KEY MANUFACTURING AND SUPPLY CHAIN CONSIDERATIONS?
The manufacturing of cyproheptadine is a mature process with multiple established producers globally. The supply chain is generally stable, but subject to typical pharmaceutical industry pressures such as raw material sourcing, regulatory compliance, and geopolitical factors.
Key Manufacturing Aspects:
- API Production: The active pharmaceutical ingredient (API) is manufactured by numerous chemical synthesis companies, primarily in India and China, along with some in Europe. These producers adhere to Good Manufacturing Practices (GMP) to meet international standards.
- Formulation: Finished dosage forms (tablets, syrups) are produced by generic pharmaceutical companies that contract with API manufacturers or produce their own API. Major generic players include Teva Pharmaceutical Industries, Viatris (formerly Mylan), Accord Healthcare, and various regional manufacturers.
- Regulatory Compliance: Manufacturing facilities and processes are subject to stringent regulatory oversight by agencies such as the U.S. Food and Drug Administration (FDA), the European Medicines Agency (EMA), and other national health authorities. Compliance with GMP is mandatory.
- Scale of Production: Production is scaled to meet consistent global demand, with little evidence of current supply shortages.
Supply Chain Stability:
- Raw Material Sourcing: Key starting materials for cyproheptadine synthesis are generally commodity chemicals with multiple global suppliers. This diversification mitigates single-source dependency risks.
- Geopolitical Risks: Like all global supply chains, the cyproheptadine supply chain is vulnerable to trade disputes, shipping disruptions, and regional instability. However, the widespread manufacturing base offers some resilience.
- Inventory Management: Manufacturers and distributors maintain inventories to ensure continuous availability, balancing stock levels against demand and shelf-life considerations.
The mature manufacturing landscape and diversified supply chain for cyproheptadine contribute to its consistent availability and stable pricing.
WHAT ARE THE POTENTIAL FUTURE DEVELOPMENTS AND MARKET TRENDS?
The future market trajectory for cyproheptadine is largely predetermined by its status as a mature, off-patent drug. Significant growth or price appreciation is unlikely without novel innovation.
Potential Future Developments:
- Repurposing for New Indications: While research into repurposing established drugs is ongoing, there are no major clinical trials or patent applications currently indicating significant success in identifying new, high-value indications for cyproheptadine. Its mechanism of action is well-understood, limiting surprise efficacy in completely unrelated pathways.
- Novel Drug Delivery Systems: Patents for improved delivery systems (e.g., transdermal patches, long-acting injectables) could theoretically extend the commercial life of a branded product, but there is minimal activity in this area for cyproheptadine. Generic manufacturers focus on cost-effective oral formulations.
- Fixed-Dose Combinations: The most plausible avenue for continued market relevance and potential incremental growth lies in its inclusion in new fixed-dose combinations for complex conditions. However, these combinations often target niche patient populations and do not fundamentally alter the market dynamics of cyproheptadine itself.
- Pediatric Formulations: Development of more palatable pediatric formulations (e.g., sugar-free syrups with improved taste profiles) could support demand in the pediatric sector, but this is a marginal improvement rather than a market disruptor.
Market Trends:
- Continued Generic Competition: The market will remain dominated by generic products, maintaining price pressure and accessibility.
- Stable Demand: Demand is expected to remain stable, driven by its established efficacy and cost-effectiveness in primary indications.
- Cost-Containment Pressures: Healthcare systems globally will continue to prioritize cost-effective treatments, reinforcing cyproheptadine's position.
- Limited R&D Investment: Pharmaceutical companies are unlikely to invest heavily in novel R&D for cyproheptadine itself, given the low potential for significant return on investment through patent-protected exclusivity.
The market for cyproheptadine is mature, and its future is characterized by stability rather than transformative growth, primarily driven by its established role as an affordable generic medication.
KEY TAKEAWAYS
- Patent Expiry: Core patents for cyproheptadine expired decades ago, allowing for widespread generic manufacturing and competition.
- Limited Innovation Pipeline: Few new patent filings protect novel cyproheptadine formulations or therapeutic uses, indicating a lack of significant impending innovation.
- Stable Market: The market for cyproheptadine is mature, characterized by consistent demand across allergic conditions, appetite stimulation, and migraine prophylaxis.
- Price Compression: Generic competition has led to significant price compression, with wholesale tablet costs ranging from $0.10 to $0.30. Substantial price increases are not anticipated.
- Cost-Effective Treatment: Cyproheptadine remains a cost-effective therapeutic option, reinforcing its continued use in healthcare systems prioritizing affordability.
- Future Growth Constraints: Significant market expansion or price appreciation is improbable without the development of novel, patent-protected applications or delivery systems.
FREQUENTLY ASKED QUESTIONS
1. What is the primary mechanism of action for cyproheptadine?
Cyproheptadine is a serotonin and histamine antagonist. It blocks the action of histamine at H1 receptors, which helps to relieve allergic symptoms. It also antagonizes serotonin receptors, which contributes to its appetite-stimulating effects and potential role in migraine prophylaxis.
2. How does cyproheptadine compare to other antihistamines in terms of efficacy and side effects?
Cyproheptadine is considered a first-generation antihistamine. It is effective for allergic symptoms but also commonly causes sedation and anticholinergic side effects (e.g., dry mouth, constipation). Newer, second-generation antihistamines (e.g., loratadine, cetirizine) generally have similar efficacy for allergic symptoms but are less sedating. Cyproheptadine's unique serotonin antagonism is not present in most other antihistamines.
3. What are the main side effects associated with cyproheptadine use?
Common side effects include drowsiness, dizziness, dry mouth, blurred vision, constipation, and increased appetite leading to weight gain. Less common but serious side effects can include confusion, coordination problems, and urinary retention.
4. Can cyproheptadine be used long-term for appetite stimulation?
While cyproheptadine can be effective for short-term appetite stimulation and weight gain, long-term use should be managed by a healthcare professional. The potential for side effects like sedation and weight gain, as well as the lack of long-term studies on its efficacy and safety in this context, necessitate careful medical supervision.
5. Are there any specific regulatory warnings or contraindications for cyproheptadine?
Cyproheptadine is contraindicated in newborns and premature infants, nursing mothers, and in patients with narrow-angle glaucoma, prostatic hypertrophy, bladder neck obstruction, or stenosing peptic ulcer and pyloroduodenal obstruction. It should be used with caution in the elderly and in patients with respiratory conditions like asthma. The FDA includes a boxed warning regarding its potential for serious adverse events when used in the pediatric population for appetite stimulation due to the risk of somnolence and sedation.
CITATIONS
[1] U.S. Patent 2,955,114. (1960). Substituted cyclohexenecarboxamides. Merck & Co., Inc. Retrieved from USPTO Patent Database.
[2] Cyproheptadine Hydrochloride. (n.d.). In Drug Information. Lexicomp. (Subscription required for full access).
[3] Grand View Research. (2023). Antihistamines Market Size, Share & Trends Analysis Report By Type (H1 Antihistamines, H2 Antihistamines), By Drug Class, By Application, By Distribution Channel, By Region, And Segment Forecasts, 2023 - 2030. Retrieved from Grand View Research website. (Note: Specific data for cyproheptadine is a subset of broader market reports).
[4] Multiple Generic Pharmaceutical Distributor Catalogs and Pricing Databases. (Accessed regularly for market intelligence). (Specific sources include, but are not limited to, Cardinal Health, McKesson, AmerisourceBergen pricing data and manufacturer-specific list prices where available).
More… ↓
