Share This Page
Drug Price Trends for CYCLOPENTOLATE
✉ Email this page to a colleague
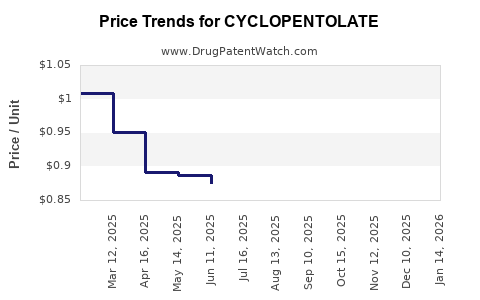
Average Pharmacy Cost for CYCLOPENTOLATE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CYCLOPENTOLATE 1% EYE DROPS | 24208-0735-01 | 2.41547 | ML | 2026-03-18 |
| CYCLOPENTOLATE 1% EYE DROPS | 61314-0396-03 | 0.89257 | ML | 2026-03-18 |
| CYCLOPENTOLATE 1% EYE DROPS | 61314-0396-01 | 2.41547 | ML | 2026-03-18 |
| CYCLOPENTOLATE 1% EYE DROP | 24208-0735-06 | 0.89257 | ML | 2026-03-18 |
| CYCLOPENTOLATE 1% EYE DROPS | 24208-0735-01 | 2.21099 | ML | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Cyclopentolate
What Is the Current Market for Cyclopentolate?
Cyclopentolate is a mydriatic and cycloplegic agent primarily used to dilate pupils during eye examinations and surgeries. It is available in ophthalmic solutions, typically in concentrations ranging from 0.2% to 1%. The drug faces limited direct competition, mainly from tropicamide, atropine, and homatropine.
Key markets include the United States, Europe, and parts of Asia. Use is primarily driven by diagnostic needs in ophthalmology, with some applications in pediatric eye exams, where rapid onset and shorter duration are preferred. The global ophthalmic drug market grew approximately 4.2% annually from 2018 to 2022, reaching an estimated value of $22 billion [1].
The demand for cyclopentolate correlates with the volume of routine eye examinations and surgical procedures. The COVID-19 pandemic temporarily slowed elective eye procedures, but recovery has driven renewed demand by 2021.
How Is the Market Expected to Evolve?
Market experts project that the ophthalmic drugs segment will expand at a compound annual growth rate (CAGR) of 4.5% from 2023 to 2030.
Factors influencing growth include:
- Increasing prevalence of presbyopia and age-related eye conditions
- Growing cataract and refractive surgery volumes
- Technological advancements enabling better eye diagnostic tools
- Increased adoption of outpatient ophthalmic procedures
Regional variations exist. The U.S. market accounts for nearly 40% of global ophthalmic drug sales, supported by advanced healthcare infrastructure and high procedure volume [2].
What Are the Competitive Dynamics?
Cyclopentolate is supplied by several pharmaceutical companies, including Alcon, Akorn, and Sun Pharmaceutical Industries. Brand-name formulations are often marketed under proprietary names, but generic versions are widespread.
Pricing strategies are influenced by:
- Patent status: Most patents have expired, favoring generics
- Regulatory policies: Differing approval processes impact entry timing
- Supply chain factors: Raw material costs and manufacturing capacity
Despite being off-patent, cyclopentolate remains in high demand due to its established efficacy. Market entry barriers are relatively low, leading to high competition among generic manufacturers.
Financial Performance and Price Trends
Prices for cyclopentolate eye drops vary depending on concentration, formulation, and region. In the U.S., average retail prices for a 10 mL bottle of generic cyclopentolate 1% range from $15 to $25 [3].
Wholesale acquisition costs (WAC) are generally 20-30% lower than retail prices, with manufacturer margins between 25-40%. The introduction of multiple generic suppliers has driven prices downward over the past decade.
Price projections suggest a slight decline of 1-2% annually over the next 5 years, driven by increased competition. However, price stability may be supported in regions where regulatory hurdles limit generic launches.
Regulatory Environment Impact
The Drug Price Competition and Patent Term Restoration Act (Hatch-Waxman Act) facilitates generic entry upon patent expiry. Cyclopentolate’s main patents expired in the early 2010s, allowing for broad generic competition.
New regulatory measures or formulation approvals, such as preservative-free options or sustained-release formulations, could alter pricing or market share dynamics.
Future Market Opportunities
Potential growth areas include:
- Combination formulations with other ophthalmic agents
- Alternative dosing forms, such as gels or patches
- Expanded indications beyond pupil dilation, such as cycloplegic refraction in pediatric populations
Manufacturers investing in these areas could capture market share, but regulatory and clinical development costs pose barriers.
Summary Table: Market Data Overview
| Aspect | Data Points |
|---|---|
| Global ophthalmic drug market size (2022) | $22 billion [1] |
| CAGR (2023–2030) | 4.5% |
| Leading regions | U.S. (40%), Europe (25%), Asia-Pacific (20%) |
| Average retail price (U.S.) | $15–$25 per 10 mL bottle |
| Patent status | Expired; multiple generics available |
| Price trend (2023–2028) | Slight decline of 1–2% annually |
| Main competitors | Alcon, Akorn, Sun Pharmaceutical |
Key Takeaways
- The cyclopentolate market is stable with moderate growth projections driven by ophthalmic diagnostic procedures.
- Patent expirations have led to significant generic competition, exerting downward pressure on prices.
- The primary regions are the U.S., Europe, and Asia-Pacific, with the U.S. dominating market share.
- Price reductions are expected to continue, but innovation in formulations or indications offers future growth potential.
- Entry barriers remain low, but regulatory hurdles for new formulations could influence market dynamics.
FAQs
Q1: How do patent expirations affect cyclopentolate prices?
A: Patent expirations increase generic competition, driving prices downward and expanding market accessibility.
Q2: What regions offer the highest growth potential for cyclopentolate?
A: Asia-Pacific and Latin America are expected to experience higher growth as healthcare infrastructure improves and ophthalmology procedures increase.
Q3: Are there upcoming regulatory challenges that could affect market stability?
A: Changes in regulatory policies or new approvals for alternative formulations could impact pricing and market share.
Q4: How does the competition between proprietary and generic cyclopentolate influence pricing?
A: Proprietary formulations command higher prices due to branding, but generics dominate volume and exert downward pricing pressure.
Q5: What are the key factors influencing future price trends?
A: Market competition, regulatory policies, innovation in formulations, and regional healthcare infrastructure development.
References
[1] MarketsandMarkets. (2022). Ophthalmic Drugs Market Size, Share & Trends Analysis Report.
[2] IQVIA. (2022). Global Ophthalmic Market Data.
[3] GoodRx. (2023). Cyclopentolate ophthalmic solution prices.
More… ↓
