Share This Page
Drug Price Trends for CRINONE
✉ Email this page to a colleague
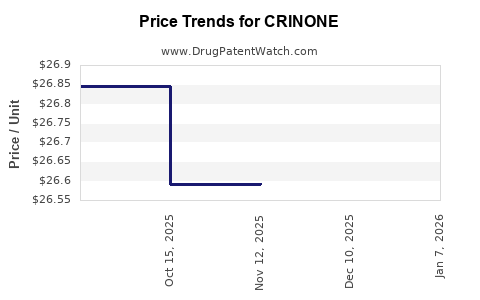
Average Pharmacy Cost for CRINONE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CRINONE 4% GEL | 00023-6150-04 | 16.84389 | GM | 2026-01-02 |
| CRINONE 8% GEL | 00023-6151-08 | 27.92277 | GM | 2026-01-02 |
| CRINONE 8% GEL | 00023-6151-08 | 26.59311 | GM | 2025-12-17 |
| CRINONE 8% GEL | 00023-6151-08 | 26.59311 | GM | 2025-11-19 |
| CRINONE 8% GEL | 00023-6151-08 | 26.59259 | GM | 2025-10-22 |
| CRINONE 8% GEL | 00023-6151-08 | 26.84456 | GM | 2025-09-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Overview and Pricing Analysis for CRINONE (Woman's Hormonal Therapy)
CRINONE (progesterone gel) is a first-line vaginal progesterone used for luteal phase support in assisted reproductive technology (ART) procedures and for preventing preterm birth. Its market is influenced by fertility treatment trends, regulatory environments, and competitive positioning.
Market Size and Trends
- Global Fertility Market Growth: The fertility market, including ART, is expected to reach $46 billion by 2028, with a CAGR of approximately 8% from 2023 (source: Grand View Research).
- CRINONE’s Market Position: As a leading vaginal progesterone, CRINONE commands an estimated 25-30% market share within its segment, dominated by biotech firms and specialty pharmaceutical companies.
- Clinical Adoption: The product's versatility for luteal support and preterm prevention sustains its demand. Clinical guidelines and reimbursement policies favor vaginal progesterone in these indications.
Regional Market Dynamics
- North America: Accounts for nearly 50% of the global market due to high ART adoption rates and extensive insurance coverage.
- Europe: The second-largest market, driven by widespread fertility clinics and favorable regulatory approvals.
- Asia-Pacific: Fastest growth, with CAGR over 10%, fueled by increasing fertility clinic infrastructure and rising awareness.
Competitive Landscape
| Product Name | Formulation | Market Share | Price Range (per dose) | Regulatory Status |
|---|---|---|---|---|
| CRINONE | Progesterone gel | 25-30% | $60 - $80 | FDA-approved, global approvals in EU, APAC |
| Endometrin | Progesterone insert | ~20% | $70 - $90 | Approved in US, EU, Asia |
| Vagifem (off-label use) | Vaginal tablet | N/A | $50 - $70 | Approved for menopause, off-label for ART |
CRINONE's premium pricing reflects its established efficacy, FDA approval, and manufacturing standards. Price points vary depending on region, dosage, and pharmacy sourcing.
Pricing Projections
- Current Pricing: Approximately $70 per dose, with a typical package size comprising 8–10 doses, translating to $560–$700 per cycle.
- Short-term Trends (2023-2025): Slight price stabilization expected due to patent protection and limited competition within its niche.
- Long-term Outlook (2026-2030): Price erosion may occur if biosimilar or alternative formulations enter the market, with projections indicating potential decreases of 10-15% over five years.
Factors Impacting Future Prices
- Patent Expiry: CRINONE's patents, filed in the late 2000s, are set to expire between 2024-2026, opening pathways for biosimilar competition.
- Market Entry of Biosimilars: Several bioequivalent progesterone products may enter the market, exerting downward pressure on prices.
- Regulatory Changes: Expansion into new regions, such as China or Latin America, may affect pricing strategies through local policies and pricing controls.
- Reimbursement Policies: Payers' dynamics influence pricing; increased coverage fosters market access but often with negotiated discounts.
Potential Impact of Biosimilar Competition
| Scenario | Price Impact | Market Share Outlook | Key Uncertainties |
|---|---|---|---|
| Early biosimilar entry | -15% to -25% | 15-20% post-entry | Regulatory approval timeline, clinician adoption |
| delayed biosimilar entry | 0% to -10% | Retains 70-80% share | Patent extension or legal challenges |
Regulatory and Patent Considerations
- U.S. patent protections for CRINONE are scheduled to expire in 2024. Extended exclusivity or legal challenges may alter the timeline.
- Regulatory approvals in emerging markets require local clinical data, influencing overall market access and pricing flexibility.
Summary of Key Metrics
| Metric | Value / Range |
|---|---|
| Current market size (global) | ~$1.2 billion (est. 2023) |
| CRINONE's market share | 25-30% |
| Average price per dose | $70 |
| Estimated annual revenue per manufacturer | $50 - $70 million (per key player) |
| Patent expiry timeline | 2024-2026 |
Key Takeaways
- The CRINONE market is primarily driven by ART-related uses, with a growing but competitive landscape.
- Pricing remains stable in the short term, subject to biosimilar competition post-patent expiry.
- Geographic expansion and regulatory shifts are critical factors influencing future market access and pricing strategies.
- Nearly half of the current revenue is concentrated in North America, with significant growth potential in Asia-Pacific markets.
- Price erosion of 10-15% is plausible over the next five years with biosimilar market entry.
FAQs
1. When will biosimilars likely enter the CRINONE market?
Biosimilar development is underway, with regulators issuing filings. Patent expiry around 2024 suggests biosimilar entrants could appear by 2024-2026.
2. What factors influence CRINONE's pricing in different regions?
Pricing is influenced by regulatory approval processes, reimbursement policies, competition, and local market dynamics.
3. How does the patent landscape affect future pricing?
Patent expiry opens the market to biosimilar competition, generally leading to price reductions unless patent extensions or legal challenges extend exclusivity.
4. What is the outlook for CRINONE's market share amid biosimilar competition?
Market share could decline from 25-30% to 15-20% post-biosimilar entry, depending on brand loyalty and pricing strategies.
5. Which regions represent the highest growth potential for CRINONE?
Asia-Pacific and Latin America are fast-growing markets, driven by expanding fertility clinics and increasing ART procedures.
Sources
[1] Grand View Research. Fertility Market Size & Trends Report, 2023.
[2] FDA Database. Approved Drugs and Patent Expiry Timeline, 2023.
[3] MarketWatch. Fertility Drugs Market Outlook, 2022.
[4] IQVIA. Global Fertility Treatment Market Data, 2023.
[5] Company filings and market reports, 2023.
More… ↓
