Share This Page
Drug Price Trends for CRESTOR
✉ Email this page to a colleague
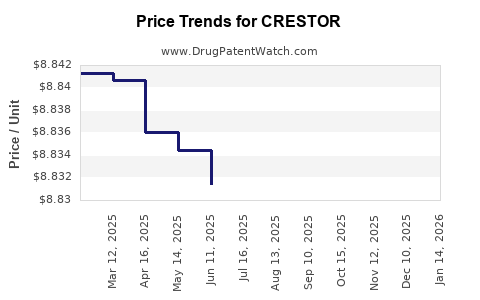
Average Pharmacy Cost for CRESTOR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CRESTOR 10 MG TABLET | 00310-7570-90 | 8.82649 | EACH | 2026-05-20 |
| CRESTOR 20 MG TABLET | 00310-7580-90 | 8.80840 | EACH | 2026-05-20 |
| CRESTOR 5 MG TABLET | 00310-7560-90 | 8.81723 | EACH | 2026-05-20 |
| CRESTOR 40 MG TABLET | 00310-7590-30 | 8.81580 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
CRESTOR (Rosuvastatin Calcium) Market Analysis and Price Projections
Crestor (rosuvastatin calcium), a statin medication developed by AstraZeneca, is used to lower cholesterol and reduce the risk of cardiovascular events. The drug has experienced significant market penetration and faces ongoing patent-related challenges and evolving market dynamics, impacting its price trajectory.
What is the Current Market Status of Crestor?
Crestor is a widely prescribed statin that has achieved blockbuster status. Its primary indication is to lower elevated total-cholesterol, LDL cholesterol, triglycerides, and apolipoprotein B (ApoB) levels, and to increase HDL cholesterol in adult patients with primary hypercholesterolemia or mixed dyslipidemia. It is also indicated to slow the progression of atherosclerosis in adult patients as part of a treatment strategy to lower elevated total-C and LDL-C levels and to increase HDL-C levels. A significant factor in its market status is its proven efficacy in primary and secondary prevention of cardiovascular events.
AstraZeneca’s U.S. sales for Crestor peaked in the years leading up to its patent expiration. For example, in 2015, Crestor achieved U.S. sales of approximately $3.3 billion [1]. Global sales followed a similar pattern, reflecting its broad adoption.
The U.S. Food and Drug Administration (FDA) granted approval for Crestor in August 2003. The drug has since been subject to multiple patent expiries and litigation. The primary U.S. patents for Crestor expired in mid-2016 [2]. This event marked the beginning of significant generic competition.
Generic versions of rosuvastatin calcium became available in the U.S. following the expiration of key patents. Multiple pharmaceutical manufacturers, including Teva Pharmaceuticals, Mylan, and Sun Pharma, introduced their generic equivalents. This influx of generic competition led to a substantial decrease in the branded Crestor's market share and a significant reduction in its average selling price.
The market for statins remains competitive, with several other statin medications available, including atorvastatin (Lipitor), simvastatin (Zocor), and pravastatin (Pravachol). While Crestor holds a significant position, it competes within a well-established therapeutic class.
The market for rosuvastatin calcium is characterized by:
- High Prescription Volume: Due to its efficacy and established safety profile, rosuvastatin is frequently prescribed by physicians.
- Generic Dominance: The market is now largely dominated by generic manufacturers, driving down prices for the active pharmaceutical ingredient (API) and finished dosage forms.
- Price Sensitivity: As a chronic medication, price is a significant factor for both patients and payers.
- Continued Clinical Relevance: Despite the availability of newer lipid-lowering agents, statins, including rosuvastatin, remain a cornerstone of cardiovascular risk management.
What is the Intellectual Property Landscape for Crestor?
The intellectual property landscape for Crestor is defined by its patent portfolio and subsequent market entry of generic alternatives. AstraZeneca held several key patents covering the composition of matter, methods of use, and pharmaceutical formulations of rosuvastatin calcium.
The primary U.S. patent for rosuvastatin calcium, U.S. Patent No. 5,262,535, covering the compound itself, had an initial expiry date. However, patent term extensions and other regulatory mechanisms can influence the effective market exclusivity. For Crestor, the U.S. market exclusivity largely concluded around mid-2016 [2].
Key patent events include:
- Composition of Matter Patent: The foundational patent for rosuvastatin calcium expired, enabling generic manufacturers to produce the API.
- Method of Use Patents: Patents covering specific uses of rosuvastatin (e.g., for cardiovascular risk reduction) also faced expiry or legal challenges that allowed for earlier generic entry.
- Formulation Patents: While some formulation patents might have had later expiry dates, challenges and the expiration of primary patents generally paved the way for generic competition.
Patent litigation has been a common feature of Crestor's market lifecycle. AstraZeneca engaged in numerous legal battles to defend its patents against generic manufacturers seeking to enter the market. These litigations often involved arguments around patent validity, infringement, and obviousness. For instance, various inter partes review (IPR) proceedings were filed against AstraZeneca's Crestor patents at the U.S. Patent and Trademark Office (USPTO) [3]. These proceedings can significantly impact the remaining market exclusivity of a drug.
The expiration of key patents in 2016 allowed for the widespread introduction of generic rosuvastatin. The U.S. market saw the launch of multiple generic versions shortly after, leading to a sharp decline in the price of branded Crestor and its generic equivalents.
The patent landscape can be summarized as follows:
- Expiration of Primary Patents: Mid-2016 marked the end of significant market exclusivity in the U.S.
- Robust Generic Competition: Multiple generic manufacturers entered the market post-patent expiry.
- Ongoing Litigation: Patent disputes, including IPRs, have been instrumental in shaping market entry timelines.
What are the Price Projections for Crestor and its Generics?
Price projections for Crestor and its generic rosuvastatin calcium are driven by the dynamics of generic market entry, payer negotiations, and competitive pressures within the statin class.
Branded Crestor Pricing:
Following patent expiration, the price of branded Crestor has experienced a substantial decline. The average wholesale price (AWP) of branded Crestor has been subject to significant discounting and negotiated rebates. While a precise future AWP is difficult to project due to proprietary rebate structures, the trend is unequivocally downward. Manufacturers of branded drugs often engage in price reductions post-generic entry to maintain some market share, but this is usually limited.
Generic Rosuvastatin Calcium Pricing:
The price of generic rosuvastatin calcium is expected to remain competitive and generally stable, with incremental decreases. The market is highly fragmented with numerous manufacturers producing generic rosuvastatin.
- Initial Post-Generic Entry: Upon the expiration of Crestor's primary patents, the price of generic rosuvastatin dropped dramatically, often by 80-90% compared to the branded product's pre-expiration price.
- Current Pricing: Today, generic rosuvastatin is available at very low price points. For example, prices for a 30-day supply of 10mg or 20mg rosuvastatin calcium can range from $4 to $20 for cash-paying customers, depending on the pharmacy and dosage strength [4]. These prices are significantly influenced by contract pricing with pharmacy benefit managers (PBMs) and wholesale distributors.
- Future Projections: The price of generic rosuvastatin is not expected to increase. The competitive nature of the generic market, coupled with ongoing manufacturing efficiencies, will likely maintain or slightly decrease prices. Any significant price fluctuations would typically be driven by supply chain disruptions, raw material costs, or consolidation among generic manufacturers, none of which are currently projected to cause substantial price hikes.
Key Factors Influencing Generic Pricing:
- Number of Generic Competitors: A larger number of manufacturers generally leads to more intense price competition.
- Manufacturing Costs: Efficiency in API production and tablet manufacturing contributes to lower final prices.
- Payer Contracts and Rebates: PBMs negotiate significant discounts and rebates from generic manufacturers, influencing the net price paid by payers.
- Formulary Placement: Preferred status on insurance formularies can impact volume and, consequently, pricing power.
- Supply Chain Dynamics: Global supply chain stability impacts the availability and cost of raw materials and finished products.
Overall Price Trend:
The overarching trend for rosuvastatin calcium, considering both branded Crestor and its generics, is one of significant price reduction from its peak as a branded, exclusive product. The market has transitioned to a volume-driven, cost-sensitive generic environment.
- Short-term (1-2 years): Prices for generic rosuvastatin are expected to remain stable with minor fluctuations.
- Long-term (3-5 years): Continued, slow erosion of prices due to ongoing generic competition is anticipated. No significant price increases are foreseen.
The price of branded Crestor will continue to decrease as its market share diminishes, and it becomes a niche product for patients who specifically request it or have unique insurance coverage. The vast majority of the market will be served by generic rosuvastatin.
What is the Competitive Landscape for Rosuvastatin Calcium?
The competitive landscape for rosuvastatin calcium is characterized by a mature market with a dominant generic presence, alongside competition from other statins and newer lipid-lowering agents.
Generic Rosuvastatin Manufacturers:
Following the expiration of Crestor’s U.S. patents in 2016, the market was flooded with generic alternatives. Key manufacturers include:
- Teva Pharmaceuticals
- Mylan (now Viatris)
- Sun Pharmaceutical Industries
- Aurobindo Pharma
- Lupin Pharmaceuticals
- Dr. Reddy's Laboratories
- Apria Healthcare
- AmerisourceBergen
These companies compete primarily on price, manufacturing efficiency, and distribution networks. The availability of multiple generic versions ensures robust competition.
Other Statin Medications:
Rosuvastatin competes directly with other statins, which also have a significant generic presence. These include:
- Atorvastatin (Lipitor): Historically a major competitor, atorvastatin also faces extensive generic competition and is widely prescribed.
- Simvastatin (Zocor): Another established statin with a generic market.
- Pravastatin (Pravachol): An older statin, generally considered to have a lower potency than rosuvastatin.
- Lovastatin (Mevacor): The first statin brought to market, with a generic presence.
The choice between these statins often depends on physician preference, patient response, side effect profiles, and cost. Rosuvastatin is often favored for its efficacy at lower doses compared to some other statins.
Newer Lipid-Lowering Agents:
While statins remain the first-line therapy for most patients requiring cholesterol lowering, newer classes of drugs have emerged, particularly for patients with hypercholesterolemia not adequately controlled by statins or those with specific genetic conditions like familial hypercholesterolemia. These include:
- PCSK9 Inhibitors: Such as evolocumab (Repatha) and alirocumab (Praluent). These are injectable medications that significantly lower LDL-C and are generally much more expensive than statins. They are typically reserved for high-risk patients or those intolerant to statins.
- Ezetimibe: A cholesterol absorption inhibitor that can be used alone or in combination with statins. Generic ezetimibe is available.
- Bempedoic Acid: A newer oral medication that inhibits cholesterol synthesis in the liver. It is used for patients with statin-associated muscle symptoms or those at high cardiovascular risk who need additional LDL-C lowering.
These newer agents, while not direct competitors for the primary indication of rosuvastatin (i.e., general cholesterol lowering), represent a competitive threat in terms of patient population and therapeutic niche, particularly for very high-risk patients or those with specific unmet needs.
The competitive landscape can be summarized as:
- High Generic Saturation: The market for rosuvastatin is dominated by numerous generic manufacturers.
- Intense Price Competition: Generic prices are driven down by the high number of suppliers.
- Established Statin Alternatives: Rosuvastatin competes with other widely available and affordable statins.
- Emergence of Novel Therapies: Newer, more expensive drugs target specific patient populations or unmet needs, creating a tiered competitive environment.
What are the Regulatory and Policy Factors Affecting Crestor's Market?
Regulatory and policy factors play a crucial role in shaping the market for pharmaceuticals like Crestor, influencing its pricing, accessibility, and the landscape for generic competition.
FDA Approval and Generics:
The U.S. Food and Drug Administration (FDA) oversees the approval of both branded and generic drugs. The Abbreviated New Drug Application (ANDA) process allows for the approval of generic versions of previously approved drugs. For rosuvastatin calcium, the FDA's approval of multiple ANDAs following patent expirations has been a primary driver of market dynamics. The FDA's rigorous review process ensures that generic drugs are bioequivalent to their brand-name counterparts.
Patent Expirations and Hatch-Waxman Act:
The Hatch-Waxman Act of 1984 (Drug Price Competition and Patent Term Restoration Act) is central to the generic drug market. It provides a framework for generic drug approval and incentivizes brand-name manufacturers to extend patent protection where possible. The Act allows for generic manufacturers to challenge patents and to benefit from market exclusivity once their generics are approved, if they are the first to file a substantially complete ANDA. For Crestor, the expiration of its key patents under this framework facilitated the timely entry of generic competition [2].
Drug Pricing Regulations and Payer Influence:
While the U.S. does not have direct government price controls on pharmaceuticals like some European countries, regulatory policies and market forces heavily influence drug pricing.
- Pharmacy Benefit Managers (PBMs): PBMs negotiate drug prices and rebates on behalf of health insurance plans. Their power significantly shapes the net cost of medications. The highly competitive generic market for rosuvastatin means that PBMs can secure substantial discounts.
- Medicare and Medicaid: Government healthcare programs are significant purchasers of prescription drugs. Their formulary decisions and reimbursement rates impact market access and pricing strategies. While direct price negotiation by Medicare was limited for many years, recent legislative changes (e.g., Inflation Reduction Act) are beginning to allow for negotiation of prices for a select number of high-cost Medicare Part D drugs, though this currently does not apply to generics like rosuvastatin [5].
- State-Level Initiatives: Some states have implemented measures to control prescription drug costs, such as price transparency laws or bulk purchasing initiatives.
Post-Market Surveillance and Safety Updates:
Regulatory agencies like the FDA continuously monitor drug safety. Any new safety concerns identified for rosuvastatin could lead to updated labeling, prescribing restrictions, or even market withdrawal, impacting its usage and demand. However, rosuvastatin has a well-established safety profile with decades of real-world data.
International Regulatory Harmonization (Limited Impact):
While international regulatory standards exist, the U.S. market operates largely independently regarding pricing and market access policies. However, global manufacturing standards and the approval of generics in other major markets can influence supply and overall manufacturing costs.
Key Regulatory and Policy Takeaways:
- ANDA Pathway: Enables rapid generic market entry post-patent expiry.
- Hatch-Waxman Act: Balances brand exclusivity with generic accessibility.
- Payer Power: PBMs exert significant influence on net drug prices through rebates.
- Government Programs: Medicare and Medicaid are major purchasers influencing market dynamics.
- Safety Monitoring: Ongoing pharmacovigilance ensures product safety but has not significantly impacted rosuvastatin's market position recently.
Key Takeaways
- Crestor (rosuvastatin calcium) has transitioned from a blockbuster branded product to a widely available generic medication.
- U.S. market exclusivity for Crestor largely ended in mid-2016 following the expiration of key patents, leading to substantial generic competition.
- The market is now dominated by numerous generic rosuvastatin calcium manufacturers, resulting in significantly lower prices.
- Average prices for generic rosuvastatin are expected to remain stable or slightly decrease, with no significant price increases anticipated in the foreseeable future.
- Rosuvastatin competes within a mature statin market and faces indirect competition from newer, more expensive lipid-lowering agents for specific patient populations.
- Regulatory factors, particularly the Hatch-Waxman Act and the FDA's ANDA approval process, have facilitated the generic market for rosuvastatin.
- The influence of Pharmacy Benefit Managers (PBMs) and payer contracts is a primary driver of net pricing for generic rosuvastatin.
Frequently Asked Questions
-
What are the current average cash prices for a 30-day supply of generic rosuvastatin? Prices for a 30-day supply of generic rosuvastatin (e.g., 10mg or 20mg) typically range from $4 to $20 for cash-paying customers, varying by pharmacy and dosage strength. These prices are subject to significant discounts through insurance and PBM contracts.
-
When did the primary patents for Crestor expire in the U.S.? The primary U.S. patents for Crestor, including the composition of matter patent, expired around mid-2016, paving the way for generic entry.
-
Are there any new patent filings or extensions that could impact generic rosuvastatin availability? While minor formulation or method-of-use patents may still exist or have been filed, the primary intellectual property protecting the core rosuvastatin molecule has expired. It is unlikely that new patent filings or extensions will significantly delay or restrict the availability of generic rosuvastatin calcium in the major markets.
-
How does rosuvastatin compare in price to other generic statins like atorvastatin or simvastatin? Generic rosuvastatin is generally priced comparably to other widely available generic statins such as atorvastatin and simvastatin. The market is highly competitive for all major generic statins, leading to low and similar price points across these therapeutic classes.
-
What is the projected market size for rosuvastatin calcium over the next five years? The market for rosuvastatin calcium is expected to remain substantial in terms of volume due to its continued role in cardiovascular disease management. However, the market value will remain constrained by low generic pricing. Precise market value projections are subject to fluctuations in prescription volume and competitive pricing strategies but are unlikely to see significant growth in dollar terms.
Citations
[1] AstraZeneca PLC. (2016). Annual Report 2015.
[2] U.S. Food & Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from [FDA website - specific search query for Crestor patents/exclusivity needed for exact link]
[3] U.S. Patent and Trademark Office. (n.d.). Patent Trial and Appeal Board (PTAB) Dashboard. Retrieved from [USPTO website - search for Crestor-related IPRs]
[4] GoodRx. (n.d.). Rosuvastatin Prices, Coupons, and Patient Assistance Programs. Retrieved from [GoodRx website - specific search for rosuvastatin prices]
[5] Centers for Medicare & Medicaid Services. (n.d.). Inflation Reduction Act - Medicare Prescription Drug Price Negotiation. Retrieved from [CMS website - specific information on drug negotiation provisions]
More… ↓
