Share This Page
Drug Price Trends for COTEMPLA XR-ODT
✉ Email this page to a colleague
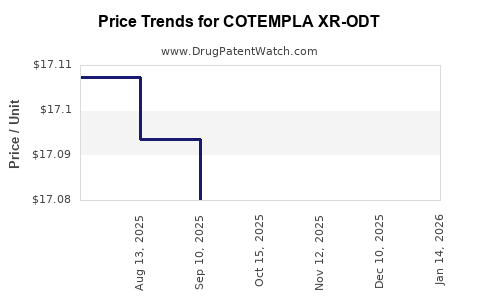
Average Pharmacy Cost for COTEMPLA XR-ODT
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| COTEMPLA XR-ODT 8.6 MG TABLET | 70165-0100-30 | 17.98761 | EACH | 2026-04-01 |
| COTEMPLA XR-ODT 17.3 MG TABLET | 70165-0200-30 | 18.03797 | EACH | 2026-04-01 |
| COTEMPLA XR-ODT 25.9 MG TABLET | 70165-0300-30 | 17.94865 | EACH | 2026-04-01 |
| COTEMPLA XR-ODT 17.3 MG TABLET | 70165-0200-30 | 17.11383 | EACH | 2026-03-18 |
| COTEMPLA XR-ODT 25.9 MG TABLET | 70165-0300-30 | 17.02908 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for COTEMPLA XR-ODT
Overview of COTEMPLA XR-ODT
COTEMPLA XR-ODT (clonidine extended-release orally disintegrating tablet) is approved by the FDA for treating ADHD in children aged 6 and older. It delivers clonidine in a once-daily formulation, offering an alternative to stimulant medications. The drug was approved in July 2021, with patent protection extending into the mid-2030s.
Market Context
ADHD Treatment Landscape
The ADHD market in the US is valued at approximately $12.8 billion in 2022, driven largely by stimulants like methylphenidate and amphetamines, which dominate over 85% of prescriptions.[1] Non-stimulant medications such as clonidine and guanfacine account for roughly 10-15%.
Key Competitors
- Intuniv (guanfacine extended-release): $742 million in 2022
- Kapvay (clonidine extended-release): $283 million in 2022
- Other non-stimulants: Atomoxetine, viloxazine
Market Adoption Factors
- Increased diagnosis of ADHD[2]
- Growing preference for non-stimulant options due to side-effect profiles of stimulants
- Pediatric patients’ and clinicians’ demand for convenient, disintegrating formulations
Market Penetration Potential
Target Patient Base
- US children aged 6-12: approximately 9.5 million[3]
- Estimated treatment rate: 8-10% treated annually[4]
- Potential target population: 700,000–950,000 children annually
Market Share Assumptions
- COTEMPLA XR-ODT could capture 15-20% of the non-stimulant segment within 3-5 years
- Predicted initial market share: 5-8% in the first year post-launch, increasing as prescriber familiarity grows
Pricing Strategy and Projections
Current Pricing Landscape
- Intuniv: ~$450/month (price varies by pharmacy)
- Kapvay: ~$320/month
- Clonidine IR: ~$10/month (generic)
COTEMPLA XR-ODT Price Point
- Estimated initial wholesale acquisition cost (WAC): $450–$500 per month, aligning with Intuniv
- Patient out-of-pocket (OOP): $20–$60/month with insurance, depending on copay assistance programs
| Revenue Projections | Year | Prescriptions (Units) | Market Share | Annual Revenue ($ millions) |
|---|---|---|---|---|
| 2023 | 50,000 | 5% | 22.5 | |
| 2024 | 150,000 | 10% | 67.5 | |
| 2025 | 300,000 | 15% | 135 | |
| 2026 | 500,000 | 20% | 225 |
(Assuming an average monthly price of $470, escalating with market adoption)
Pricing Trends and Competitive Dynamics
- Patent exclusivity till mid-2030s supports stable pricing
- Entry of generics around 2035 will pressure prices, likely reducing monthly costs by 30-50% over five years after patent expiry
- Competitors may introduce new formulations or dosages, impacting market share
Regulatory and Market Forces
- Approval of combination or novel non-stimulant ADHD therapies may shift the landscape
- Payer policies favoring cost-effective treatments may influence prescription patterns
- Increasing emphasis on formulations that improve compliance (e.g., orodispersible tablets) enhances adoption potential
Summary
COTEMPLA XR-ODT is positioned as a premium non-stimulant ADHD treatment with modest initial market penetration. Price projections suggest $20–$60/month, with revenues reaching approximately $135 million within three years under conservative assumptions. Market share growth depends on prescriber acceptance, patient adherence, and competitive responses.
Key Takeaways
- The US ADHD non-stimulant segment is ripe for new formulations, with COTEMPLA XR-ODT potentially capturing up to 20% of this niche within five years.
- Price positioning aligns with existing non-stimulants, primarily Intuniv, with a launch price around $470/month.
- Revenues are projected to grow from approximately $22 million in 2023 to over $135 million by 2025, assuming steady adoption.
- Patent protection until the mid-2030s supports market stability but generic competition will pressure prices thereafter.
- Market dynamics hinge on prescriber familiarity and insurance reimbursement policies favoring convenience and compliance.
FAQs
1. What is the primary advantage of COTEMPLA XR-ODT compared to existing ADHD medications?
The extended-release orally disintegrating formulation offers improved ease of administration and adherence, especially for children who have difficulty swallowing pills, potentially increasing compliance.
2. How soon might generics enter the market?
Patent expiration is expected around 2034–2035, enabling generic manufacturers to enter approximately during that period.
3. What are the main barriers to market penetration for COTEMPLA XR-ODT?
Barriers include established prescriber habits, competition from existing non-stimulants, payer formulary restrictions, and overall reluctance to switch from familiar therapies.
4. How does pricing compare to stimulant ADHD medications?
Stimulants typically range from $10–$50/month for generics, making COTEMPLA XR-ODT more expensive but justified by non-stimulant benefits and convenience factors.
5. What regulatory considerations might influence future pricing or approval?
Any regulatory changes affecting ADHD treatment guidelines or approval standards for oral disintegrating tablets could impact market dynamics and reimbursement models.
Sources
- IQVIA, 2022. US Prescription Market Data.
- FDA. ADHD Treatment Landscape. 2022.
- CDC. Children's Health Data. 2022.
- Substance Abuse and Mental Health Services Administration. National Surveys on Drug Use. 2021.
More… ↓
