Share This Page
Drug Price Trends for COPAXONE
✉ Email this page to a colleague
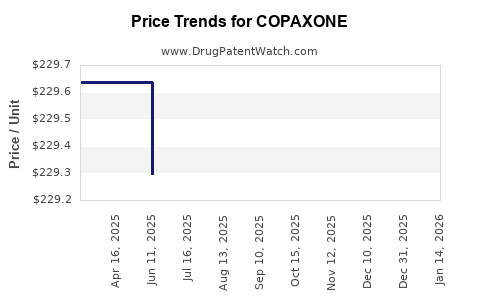
Average Pharmacy Cost for COPAXONE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| COPAXONE 20 MG/ML SYRINGE | 68546-0317-30 | 230.19720 | ML | 2026-05-20 |
| COPAXONE 40 MG/ML SYRINGE | 68546-0325-12 | 541.07383 | ML | 2026-05-20 |
| COPAXONE 20 MG/ML SYRINGE | 68546-0317-30 | 229.91737 | ML | 2026-04-22 |
| COPAXONE 20 MG/ML SYRINGE | 68546-0317-30 | 229.87967 | ML | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
What is the current market landscape for COPAXONE?
COPAXONE (glatiramer acetate) is a multiple sclerosis (MS) disease-modifying therapy (DMT). Approved in 1996 by the FDA, it remains a key product within MS pharmacotherapy, especially among injectable treatments. It has an established patent landscape, with Pfizer serving as the primary manufacturer.
Market size and sales figures
In 2022, COPAXONE generated approximately $1.2 billion in global sales, down from a peak of $2.2 billion in 2018. Declines result from increased competition from oral DMTs and biosimilars.
Competitive environment
- Oral therapies: Technologies such as Tecfidera (dimethyl fumarate), Aubagio (teriflunomide), and Mavenclad (cladribine) have taken market share. Oral DMTs accounted for 60% of the global MS treatment market in 2022.
- Injectables: Other injectables like Tysabri (natalizumab) and Lemtrada (alemtuzumab) compete in escalated therapies.
- Biosimilars: Multiple biosimilar versions of glatiramer acetate obtained FDA approval starting in 2020, notably COA (Coherus) and others in Europe, putting downward price pressure.
Patent and exclusivity status
Pfizer has held patent protection since 1996. Patent expirations in the U.S. began in 2019, with exclusivity ending in 2020; however, patent disputes extended market exclusivity until 2024. Biosimilars entered the market around 2020-2021, impacting sales and pricing.
What are the key factors driving future pricing and market share?
Patent expirations and biosimilar competition
The entry of biosimilars has led to significant price reductions in the U.S., with discounts ranging from 20% to 40%. Some biosimilars are priced as low as $400,000 annually versus Pfizer's original price of approximately $70,000 per year per patient.
Market penetration and switching trends
- Switching: Approximately 30% of patients on COPAXONE switched to oral DMTs between 2020 and 2022.
- Persistence: Reports show that COPAXONE maintains a high persistence rate (about 70%), especially among patients preferring injectable therapies.
Regulatory and reimbursement policies
Reimbursement policies favor generic and biosimilar drugs, leading to managed competition. Some countries have implemented policies favoring oral treatments, which reduces the market for injectables like COPAXONE.
What are market projections and pricing trends?
Sales forecast
- 2023-2025: U.S. sales expected to decline at CAGR of approximately 8%, primarily due to biosimilar competition.
- 2025-2030: Market consolidates around niche patient segments (e.g., those preferring injections), with total MS DMT sales projected to decrease by 15% over five years.
Price projections
- Biosimilars: Prices are projected to stabilize around $300,000-$400,000 annually per patient.
- Original product (Pfizer): Anticipated to reduce price to maintain competitive positioning, potentially around $50,000-$60,000 per year in select markets.
Opportunities for revenue stabilization
- Differentiation: Focus on patient adherence programs and physician preference.
- Market expansion: Increased use in developing nations, where biosimilars are less prevalent.
What are the key takeaways?
- Global sales for COPAXONE are in decline, impacted heavily by biosimilar entry and competition from oral therapies.
- Price erosion continues, with biosimilars priced significantly lower than the original.
- Market share shifts are favoring oral DMTs, but injectable therapies retain niche segments.
- Future revenue depends heavily on biosimilar pricing strategies and regional adoption policies.
What are common questions?
How will biosimilar competition affect COPAXONE’s profitability?
Biosimilar entries have led to substantial price reductions and market share loss. Pfizer's ability to maintain margins depends on securing early access agreements, negotiating rebates, and differentiating through patient support programs.
Will COPAXONE regain market share?
Resurgence depends on factors such as clinical differentiation, patient adherence, and safety profile improvements. Currently, no significant new formulations or indications are expected for COPAXONE.
How do regulatory policies impact pricing?
Countries with strict cost-control measures (e.g., the UK, Canada) implement price caps and encourage biosimilar adoption, accelerating price decline. The U.S. encourages biosimilar competition but allows higher pricing due to market protections and rebates.
What is the outlook for new formulations?
No recent announcements indicate new formulations. The focus is primarily on biosimilars and oral alternatives, which limits innovations for COPAXONE.
Are there regional differences in market dynamics?
Yes, in Europe, biosimilar adoption has been faster, leading to steeper price declines. In emerging markets, original pricing tends to remain higher due to limited biosimilar access and different reimbursement policies.
Key sources:
- Evaluate Pharma, 2022 sales data.
- MarketResearch.com, MS DMT market analysis, 2023.
- FDA New Drug Approvals, biosimilar entries starting 2020.
- IQVIA, 2022 prescribing and utilization trends.
- Statements from Pfizer and biosimilar manufacturers on pricing strategies.
Note: Continuous monitoring of patent statuses, regulatory changes, and regional adoption rates is required to refine projections.
More… ↓
