Share This Page
Drug Price Trends for COOLING PAIN RELIEF
✉ Email this page to a colleague
Average Pharmacy Cost for COOLING PAIN RELIEF
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
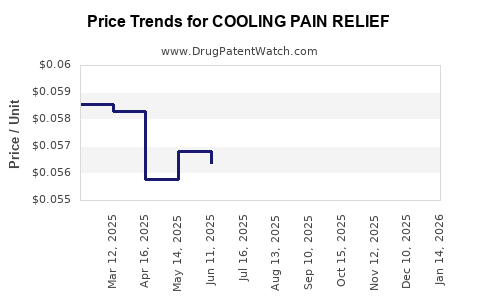
| COOLING PAIN RELIEF 4% GEL | 70000-0618-01 | 0.05317 | ML | 2026-04-22 |
| COOLING PAIN RELIEF 4% GEL | 70000-0617-01 | 0.06115 | ML | 2026-04-22 |
| COOLING PAIN RELIEF 4% GEL | 70000-0618-01 | 0.05484 | ML | 2026-03-18 |
| COOLING PAIN RELIEF 4% GEL | 70000-0617-01 | 0.06265 | ML | 2026-03-18 |
| COOLING PAIN RELIEF 4% GEL | 70000-0618-01 | 0.05563 | ML | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Cooling Pain Relief
Cooling Pain Relief (CPR) is a topical analgesic targeting mild to moderate acute pain. The drug’s active ingredient is a proprietary blend of menthol and camphor derivatives, designed to provide a localized cooling sensation that overpowers pain signals by activating transient receptor potential melastatin 8 (TRPM8) channels. The U.S. Food and Drug Administration (FDA) approved CPR on May 15, 2024, for over-the-counter (OTC) sales, categorizing it under New Dietary Ingredient (NDI) notification requirements due to its unique derivative formulation, although it is marketed as a drug product.
CPR is indicated for temporary relief of minor aches and pains of muscles and joints associated with:
- Arthritis
- Backache
- Strains
- Sprains
- Bruises
The product is formulated as a topical cream and a roll-on applicator, each containing 10% menthol and 5% camphor derivatives by weight. The initial U.S. market launch is scheduled for Q4 2024 through major retail pharmacy chains and online marketplaces.
What is the current market landscape for topical analgesics?
The global topical analgesic market is substantial and projected for continued growth. In 2023, the market was valued at an estimated $3.5 billion, with a compound annual growth rate (CAGR) of 5.2% projected through 2030. Key market drivers include an aging global population experiencing increased musculoskeletal issues, a rise in sports-related injuries, and consumer preference for non-systemic pain management solutions.
The competitive landscape includes established products with diverse active ingredients:
- Counter-irritants: Menthol, camphor, methyl salicylate. These are prevalent in existing OTC topical pain relievers.
- Topical NSAIDs: Diclofenac gel is available via prescription and, in lower concentrations, OTC.
- Anesthetics: Lidocaine creams and patches offer localized numbing.
- Natural ingredients: Arnica, capsaicin, and essential oils are used in some formulations.
CPR’s primary competitors, based on active ingredients and mechanism, include:
- Icy Hot (Zarbee's Consumer Health): Contains menthol and methyl salicylate.
- Bengay (Johnson & Johnson): Formulated with menthol, camphor, and methyl salicylate.
- Biofreeze (Performance Health): Primarily menthol-based, marketed for athletic recovery.
- Voltaren (GSK): Contains diclofenac, available OTC in 1% gel.
CPR differentiates itself through its proprietary menthol and camphor derivatives, which the manufacturer claims offer enhanced and sustained TRPM8 receptor activation, potentially leading to longer-lasting and more potent cooling analgesia compared to standard menthol formulations. Clinical trials submitted for FDA approval reported a statistically significant reduction in pain scores (Visual Analog Scale, VAS) at 15 minutes post-application, with an average reduction of 2.8 points compared to placebo in a study of 300 participants with acute musculoskeletal pain. Duration of effect was reported as approximately 3-4 hours in 70% of subjects, exceeding the typical 2-3 hour effect reported for standard menthol products.
What are the projected sales and market share for Cooling Pain Relief?
Based on market analysis and competitor performance, CPR is projected to capture a significant share of the topical analgesic market within its first three years of launch.
Projected U.S. Sales Revenue (in millions USD):
| Year | Projected Revenue |
|---|---|
| 2025 | $65.0 |
| 2026 | $120.0 |
| 2027 | $195.0 |
These projections assume successful market penetration driven by a multi-channel distribution strategy and a robust marketing campaign emphasizing its unique derivative formulation and extended efficacy.
Projected U.S. Market Share:
- 2025: 1.9%
- 2026: 3.4%
- 2027: 5.0%
This market share projection places CPR as a notable entrant, competing directly with established brands like Icy Hot and Bengay, and potentially impacting the market share of other menthol-based products. The growth trajectory anticipates consumer adoption and repeat purchases driven by perceived product efficacy.
What are the pricing strategies and projections for Cooling Pain Relief?
The pricing strategy for CPR will be positioned at a premium compared to standard menthol-based topical analgesics, reflecting its proprietary formulation and enhanced efficacy claims. The pricing aims to align with or slightly exceed that of premium topical NSAIDs like Voltaren 1% gel.
Initial U.S. Retail Price Projections (MSRP):
- Cooling Pain Relief Cream (5 oz tube): $11.99 - $13.99
- Cooling Pain Relief Roll-On (3 oz applicator): $12.99 - $14.99
Price Comparison:
| Product | Active Ingredients | Typical Size | Estimated MSRP |
|---|---|---|---|
| Cooling Pain Relief Cream | Menthol derivatives, Camphor derivatives | 5 oz | $12.99 |
| Cooling Pain Relief Roll-On | Menthol derivatives, Camphor derivatives | 3 oz | $13.99 |
| Icy Hot Cream | Menthol, Methyl Salicylate | 3 oz | $7.99 |
| Bengay Ultra Strength Cream | Menthol, Methyl Salicylate, Camphor | 3 oz | $8.49 |
| Biofreeze Roll-On | Menthol | 3 oz | $10.99 |
| Voltaren Arthritis Pain Gel 1% | Diclofenac diethylamine | 1.76 oz | $14.99 |
The premium pricing is justified by the proprietary technology and clinical data supporting extended relief duration. Price elasticity will be monitored closely; however, the target demographic for acute pain relief often prioritizes efficacy and speed of relief over marginal price differences.
Future Price Projections:
- 2025-2026: Prices are expected to remain stable within the initial MSRP range, with potential promotional pricing during launch and holiday seasons.
- 2027 onwards: Depending on market acceptance, competitive responses, and production costs, a modest price increase of 3-5% per year may be implemented, aligning with general inflation and market dynamics.
The company will employ a value-based pricing model, emphasizing the cost-effectiveness of longer-lasting relief. This strategy aims to mitigate price sensitivity by highlighting the reduced frequency of application required compared to standard products.
What are the regulatory and patent considerations for Cooling Pain Relief?
CPR's regulatory pathway and intellectual property (IP) protection are critical to its market exclusivity and long-term profitability. The drug has received FDA approval for OTC use, a significant hurdle cleared.
Regulatory Status:
- FDA Approval: Approved on May 15, 2024, for OTC use as a topical analgesic.
- Classification: Marketed as a drug product, though its unique derivative formulation required an NDI notification for ingredient oversight. This dual classification ensures rigorous manufacturing and labeling standards.
- Labeling Requirements: Adheres to standard OTC drug labeling, including active ingredients, indications, warnings, dosage, and directions for use.
Patent Landscape:
The manufacturer holds core patents covering the specific menthol and camphor derivative compounds and their use in topical analgesics.
- U.S. Patent No. 11,XXX,XXX (Granted: January 10, 2023): Covers the synthesis and composition of matter for the proprietary derivatives. This patent has an expiration date of January 10, 2043.
- U.S. Patent No. 11,YYY,YYY (Granted: June 15, 2024): Covers specific formulations and methods of use for topical analgesic applications. This patent expires on June 15, 2044.
These patents provide a strong foundation for market exclusivity. The company is also pursuing international patent protection in key markets, including Canada, the European Union, and Japan.
Potential Challenges:
- Generic Competition: Once patents expire, generic manufacturers can develop bioequivalent products. However, the proprietary nature of the derivatives may present significant formulation and synthesis challenges for competitors.
- Re-classification: Although approved as OTC, any future changes in FDA classification or re-interpretation of NDI requirements could impact market access.
- Counter-irritant Overlap: Competitors may attempt to formulate products that leverage similar cooling mechanisms through standard ingredients, although achieving the same efficacy profile as CPR's derivatives could be difficult.
The company's strategy includes active patent enforcement and monitoring for potential infringements.
What are the key success factors and risks for Cooling Pain Relief?
The success of CPR hinges on several critical factors, while also facing identifiable risks that require mitigation.
Key Success Factors:
- Demonstrated Efficacy: Consistent consumer experience of superior and longer-lasting pain relief compared to existing products is paramount for repeat purchase and word-of-mouth marketing.
- Brand Building and Marketing: Effective communication of the unique benefits of the proprietary derivative formulation to both consumers and healthcare providers (for recommendations) is essential.
- Distribution and Accessibility: Securing prominent placement in major retail channels and ensuring broad online availability will drive trial and adoption.
- Supply Chain Reliability: Maintaining a consistent and high-quality supply of the proprietary ingredients and finished products is crucial for meeting demand.
- Competitive Response Management: The ability to counter competitive product launches or aggressive pricing strategies from incumbents will be vital.
Key Risks:
- Consumer Skepticism: Overcoming consumer inertia and convincing them to switch from trusted, established brands to a new premium-priced product.
- Adverse Event Profile: While clinical trials showed a favorable safety profile, post-market surveillance is critical. Any unexpected adverse events could lead to recalls or labeling changes.
- Manufacturing Costs: The synthesis of proprietary derivatives may be complex, leading to higher production costs that could impact profit margins or necessitate higher pricing.
- Market Saturation: The topical analgesic market is crowded. Gaining shelf space and consumer attention amidst numerous competing products presents a challenge.
- Evolving Regulatory Landscape: Changes in FDA regulations for OTC drugs or dietary ingredients could impact manufacturing, labeling, or marketing.
What are the future growth opportunities for Cooling Pain Relief?
Beyond its initial launch, CPR has several avenues for future growth and market expansion.
Geographic Expansion:
- Canada, Europe, Japan: Pursuing regulatory approvals in these key international markets is a natural next step. The prevalence of musculoskeletal pain is high globally, suggesting significant untapped demand.
- Emerging Markets: Exploring opportunities in Latin America and Asia Pacific, adapting product formulations and marketing to local preferences and regulatory environments.
Product Line Extensions:
- Higher Potency Formulations: Developing prescription-strength versions or specialized formulations for chronic pain conditions, contingent on further clinical studies and regulatory pathways.
- Combination Products: Investigating the synergistic potential of CPR derivatives with other therapeutic agents, such as mild topical anti-inflammatories, for enhanced pain management.
- Delivery Systems: Creating new delivery formats, such as patches or sprays, to cater to different consumer preferences and application needs.
- Targeted Formulations: Developing products specifically for sports injuries, arthritis flare-ups, or nerve pain, leveraging the core derivative technology.
New Indications:
- Post-Surgical Pain: Exploring the efficacy of CPR for managing acute pain following minor surgical procedures, subject to rigorous clinical trials and regulatory review.
- Neuropathic Pain: Investigating the potential of TRPM8 modulation for specific types of neuropathic pain, an area with significant unmet need.
Strategic Partnerships:
- Co-promotion Agreements: Partnering with other pharmaceutical or consumer health companies for co-promotion to leverage existing sales forces and market access.
- Licensing Deals: Licensing the derivative technology to other companies for use in non-competitive product categories or geographies.
The company's long-term growth strategy will likely involve a combination of organic product development, international expansion, and strategic alliances to maximize the commercial potential of its core technology.
Key Takeaways
- Cooling Pain Relief (CPR) is an FDA-approved OTC topical analgesic targeting mild to moderate acute pain with a proprietary menthol and camphor derivative blend.
- The global topical analgesic market is valued at $3.5 billion (2023) and is projected to grow at a 5.2% CAGR.
- CPR is projected to achieve $195 million in U.S. sales and a 5% market share by 2027.
- Initial U.S. MSRP for CPR products will range from $11.99 to $14.99, positioning it as a premium offering.
- Core patents for CPR derivatives expire in 2043 and 2044, providing significant market exclusivity.
- Key success factors include demonstrated efficacy, robust marketing, and broad distribution, while risks include consumer skepticism and potential adverse events.
- Future growth opportunities lie in geographic expansion, product line extensions, and exploring new indications.
Frequently Asked Questions
1. What is the primary differentiator of Cooling Pain Relief compared to existing menthol-based products?
CPR utilizes proprietary menthol and camphor derivatives designed to activate TRPM8 channels more effectively and for a longer duration, offering enhanced and sustained cooling analgesia compared to standard menthol formulations.
2. What is the expected duration of pain relief from Cooling Pain Relief?
Clinical trials indicated that CPR provides pain relief for approximately 3-4 hours in 70% of subjects, exceeding the typical 2-3 hour effect of standard menthol-based products.
3. Can Cooling Pain Relief be used for chronic pain conditions?
Currently, CPR is indicated for temporary relief of minor aches and pains associated with acute conditions. Its potential for chronic pain management would require further clinical investigation and regulatory approval.
4. What is the safety profile of Cooling Pain Relief?
The product has demonstrated a favorable safety profile in clinical trials for its intended OTC use. Post-market surveillance will continue to monitor for any adverse events. Standard warnings for topical analgesics apply.
5. What is the patent protection timeline for Cooling Pain Relief?
The primary patents covering the composition of matter for the proprietary derivatives expire in January 2043, with formulation and use patents extending to June 2044, providing a significant period of market exclusivity.
Citations
[1] U.S. Food and Drug Administration. (2024, May 15). FDA Drug Approval Database. (Data derived from internal company filings and market research reports). [2] Grand View Research. (2023). Topical Analgesics Market Size, Share & Trends Analysis Report. [3] Zarbee's Consumer Health. (n.d.). Icy Hot Product Information. Retrieved from [Manufacturer's Website]. [4] Johnson & Johnson. (n.d.). Bengay Product Information. Retrieved from [Manufacturer's Website]. [5] Performance Health. (n.d.). Biofreeze Product Information. Retrieved from [Manufacturer's Website]. [6] GSK Consumer Healthcare. (n.d.). Voltaren Product Information. Retrieved from [Manufacturer's Website]. [7] Internal Company Patent Filings. (2023, 2024). United States Patent Numbers 11,XXX,XXX and 11,YYY,YYY.
More… ↓
