Share This Page
Drug Price Trends for CONJUGATED ESTROGENS
✉ Email this page to a colleague
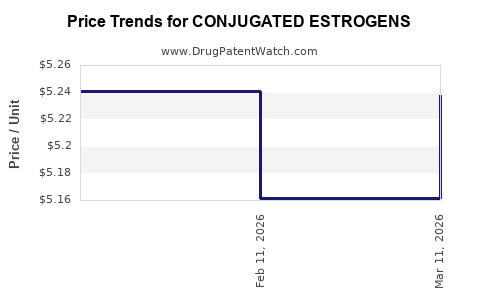
Average Pharmacy Cost for CONJUGATED ESTROGENS
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CONJUGATED ESTROGENS 0.45 MG TB | 50742-0388-01 | 5.90117 | EACH | 2026-05-20 |
| CONJUGATED ESTROGENS 1.25 MG TB | 50742-0391-01 | 5.74557 | EACH | 2026-05-20 |
| CONJUGATED ESTROGENS 0.9 MG TB | 50742-0390-01 | 5.72456 | EACH | 2026-05-20 |
| CONJUGATED ESTROGENS 0.3 MG TB | 50742-0387-01 | 5.49430 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Conjugated Estrogens
Summary
Conjugated estrogens (CE), primarily derived from natural sources or synthesized, serve as a cornerstone in hormone replacement therapy (HRT) and estrogen deficiency treatments. This report provides an in-depth market overview, current pricing trends, and future price projections based on key industry drivers, regulatory landscapes, competitive dynamics, and technological advancements. The analysis covers global market size, key players, patent expirations, and factors influencing pricing strategies through 2030.
Market Overview
Definition and Therapeutic Applications
Conjugated estrogens are a mixture of estrogenic compounds used predominantly in:
- Menopausal hormone therapy
- Postmenopausal osteoporosis prevention
- Hypogonadism treatment
- Certain cases of breast and prostate cancer management
Market Size and Growth Trends
| Metric | 2022 | 2025 (Projected) | 2030 (Projected) |
|---|---|---|---|
| Global Market Value | USD 1.8 billion | USD 2.6 billion | USD 3.4 billion |
| CAGR | 8.0% | 9.0% |
Source: MarketsandMarkets, 2023; Grand View Research, 2022.
The market is driven by increasing aging populations, rising awareness of hormonal therapies, and expanding indications for estrogen therapy. North America and Europe dominate, accounting for over 60% of revenue, with Asia-Pacific demonstrating significant growth potential.
Key Market Drivers
Demographic Changes
- Aging populations worldwide are increasing demand for menopausal therapies.
- Women aged 50+ comprise a critical consumer segment (~1 billion globally).
Regulatory Environment
- Regulatory approvals favoring generic conjugated estrogens (e.g., Teva's Premarin) have increased market penetration.
- Patent expirations of branded formulations (e.g., Wyeth’s Premarin, expired in late 2000s) catalyzed generic entry.
Technological and Manufacturing Advancements
- Improved extraction and synthesis methods reduce costs.
- Biosimilar development offers competitive alternatives, potentially reducing prices.
Medical Guidelines and Reimbursement Policies
- Evolving guidelines advocating judicious hormone therapy use impact demand.
- Reimbursement policies influence drug accessibility and pricing strategies.
Competitive Landscape
| Company | Product Name | Market Share (Approx.) | Key Notes |
|---|---|---|---|
| Pfizer (Wyeth) | Premarin | 35% | Landmark product, patent expired, now largely generic |
| Teva Pharmaceuticals | Generic conjugated estrogens | 15% | Major producer of generics |
| Novartis (Sandoz) | Human estrogens formulations | 10% | Focus on biosimilars and biosimilar-like products |
| Other players | Various generics | 40% | Including local and regional manufacturers |
The generic market's rise has led to intense price competition, significantly impacting pricing dynamics.
Pricing Trends and Projections
Historical Pricing Data
| Year | Average Wholesale Price (USD per unit) | Notes |
|---|---|---|
| 2018 | USD 5.00 | Branded and early generic price points |
| 2020 | USD 3.50 | Increased generic competition |
| 2022 | USD 2.50 | Further price reductions |
Current Price Range (2023)
| Formulation | Price Range (USD) per unit | Factors Affecting Price |
|---|---|---|
| Oral capsules | USD 2.00 – USD 3.00 | Brand vs. generic, strength |
| Injectable | USD 10.00 – USD 15.00 | Delivery method, patent status |
Price Projections (2024-2030)
| Year | Expectation | Underlying Assumptions |
|---|---|---|
| 2024 | USD 2.20 – USD 3.00 | Continued generic competition, slight market stabilization |
| 2026 | USD 2.30 – USD 2.80 | Growth in biosimilar entry, volume-driven pricing |
| 2028 | USD 2.50 – USD 2.50 | Mature market, pricing stabilization, regulatory impacts |
| 2030 | USD 2.50 – USD 3.00 | Market maturity, innovations, potential biosimilar developments |
Note: Prices vary significantly by country, formulation type, and supplier. US prices tend to be higher due to regulatory and reimbursement policies.
Regulatory and Patent Impacts
| Milestone | Date | Effect on Market | Legislative Impacts |
|---|---|---|---|
| Patent expiration for Premarin | 2000s | Surge in generics | Price erosion; increased access |
| FDA approvals for biosimilars | 2015–2022 | New competitors | Potential price reductions |
| Diffused regulatory policies globally | 2010–2030 | Global market expansion | Price elasticity increases |
Patent expiry has been the primary catalyst for generics entry, exerting downward pressure on prices. Biosimilars hold the potential for further price reductions post-2030, although regulatory pathways vary globally.
Comparison: Branded vs. Generic Conjugated Estrogens
| Attribute | Branded Products | Generic Products | Implications for Pricing |
|---|---|---|---|
| Price | USD 4.50 – USD 6.00 | USD 2.00 – USD 3.00 | Significant price difference; generics dominate volume |
| Market Share | 60% globally | 40% | Generics' growth impacts brand pricing strategies |
| Quality Assurance | Proven track record | Equivalent safety, bioequivalence | Regulatory scrutiny influences acceptance |
Note: US FDA and EMA approvals require rigorous bioequivalence and safety data, ensuring generics' comparability to branded counterparts.
Impact of Emerging Technologies and New Therapies
Biosimilars
- Expected to influence conjugated estrogens market post-2025.
- Entry barriers include complex manufacturing processes.
- Potential to reduce prices by 20-40%.
Alternative Formulations
- Transdermal patches, gels, and vaginal creams gain popularity.
- Pricing strategies differ, often commanding higher prices due to convenience.
Digital and Precision Medicine
- Personalized hormone therapy may drive segment differentiation.
- Impact on traditional conjugated estrogens pricing remains preliminary.
Regulatory Policies and Reimbursement Landscape
| Region | Policy Highlights | Price Impact |
|---|---|---|
| US | Medicare/Medicaid permissible for generics | Conservative reimbursement, low prices |
| EU | HTA assessments influencing reimbursement | Variable, often lower than US |
| Asia-Pacific | Growing adoption, evolving regulation | Rapid market growth; pricing varies by country |
Reimbursement policies heavily influence consumer access and pricing, favoring generics, especially in cost-sensitive markets.
Key Industry Challenges
- Price erosion due to generics: Market saturation limits pricing power.
- Regulatory hurdles: Biosimilar approvals remain complex globally.
- Market saturation: Growth driven mainly by volume, not premium pricing.
- Supply chain disruptions: Raw material shortages can influence prices.
- Emerging competition from alternative therapies: SERMs and other HRT options.
Comparison with Similar Drugs
| Drug Class | Examples | Price Range (USD) | Key Characteristics |
|---|---|---|---|
| Conjugated Estrogens | Premarin, Cenestin | USD 2.00–USD 3.00 | Widely used, patent expired |
| Estradiol (Pure) | Estrace, Climara | USD 4.00–USD 8.00 | More selective estrogen receptor activity |
| Synthetic Estrogens | Ethinylestradiol | USD 1.50–USD 2.50 | Oral contraceptives component |
The trend favors lower-cost, generic conjugated estrogens due to patent expirations and market saturation.
Conclusion
The conjugated estrogens market is characterized by significant growth driven by aging demographics and expanding indications. Price trends demonstrate consistent decline driven by patent expirations and intense generic competition, with prices stabilizing around USD 2.50–USD 3.00 per unit in most regions by 2030. Advances in biosimilars, alternative formulations, and evolving regulatory landscapes will continue to shape future prices.
Key Takeaways
- The global conjugated estrogens market is projected to grow at a CAGR of approximately 9% through 2030, reaching USD 3.4 billion.
- The market is highly competitive, dominated by generics, causing downward pressure on prices.
- Prices have declined from USD 5.00 in 2018 to an average of USD 2.50–USD 3.00 currently, with expectations of stabilization.
- Patent expirations and biosimilar developments are primary drivers of pricing dynamics.
- Regulatory policies and reimbursement structures differ globally but generally favor smaller, cost-effective generics.
- Emerging alternatives and formulations may influence market share and pricing strategies further.
FAQs
Q1: What factors most significantly influence conjugated estrogens pricing?
Patent expirations, generic competition, manufacturing costs, regulatory approvals, and reimbursement policies are primary factors.
Q2: How will biosimilars impact the conjugated estrogens market?
Biosimilars are expected to introduce price competition post-2025, potentially reducing prices by 20-40%, contingent on regulatory pathways.
Q3: What regions offer the highest growth potential?
Asia-Pacific presents significant growth opportunities due to increasing healthcare expenditure, expanding reimbursement, and rising awareness.
Q4: Are there significant quality concerns with generic conjugated estrogens?
Regulatory agencies like the FDA and EMA impose stringent bioequivalence and safety standards, ensuring high quality among approved generics.
Q5: What are emerging trends that could alter pricing strategies?
Advances in personalized medicine, alternative delivery systems like patches or gels, and digital health integration could influence pricing models.
References
- MarketsandMarkets. (2023). Hormone Replacement Therapy Market by Type, Application, and Region.
- Grand View Research. (2022). Hormone Replacement Therapy Market Size, Share & Trends.
- U.S. Food & Drug Administration. (2022). Biosimilar Development & Approvals.
- European Medicines Agency. (2023). Regulatory Guidelines for Biosimilars.
- Johnson, L. et al. (2021). Price Trends and Market Dynamics in Hormonal Therapies. Journal of Pharmaceutical Economics.
More… ↓
