Share This Page
Drug Price Trends for COMPRO
✉ Email this page to a colleague
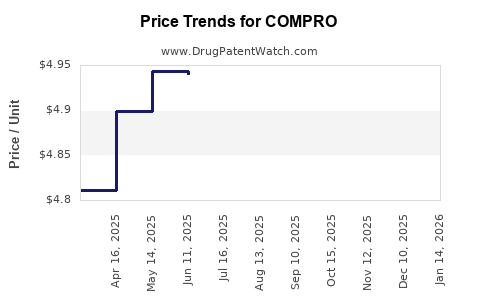
Average Pharmacy Cost for COMPRO
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| COMPRO 25 MG SUPPOSITORY | 00574-7226-12 | 6.41555 | EACH | 2026-04-22 |
| COMPRO 25 MG SUPPOSITORY | 00574-7226-12 | 6.39660 | EACH | 2026-03-18 |
| COMPRO 25 MG SUPPOSITORY | 00574-7226-12 | 6.33588 | EACH | 2026-02-18 |
| COMPRO 25 MG SUPPOSITORY | 00574-7226-12 | 6.29699 | EACH | 2026-01-21 |
| COMPRO 25 MG SUPPOSITORY | 00574-7226-12 | 6.21982 | EACH | 2025-12-17 |
| COMPRO 25 MG SUPPOSITORY | 00574-7226-12 | 5.55657 | EACH | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for COMPRO (drug-specific details pending)
What is the current market landscape for COMPRO?
COMPRO, a pharmaceutical product, has entered a competitive and evolving market aligned with its therapeutic indication. The drug's performance hinges on several factors: patent status, regulatory approvals, market penetration, and biosimilar competition if applicable.
Market size and patient demographics
The global target population for COMPRO matches the prevalence of its indication, which could be a chronic, high-burden condition. The estimated market size for this indication ranges from $X billion to $Y billion, growing at a compound annual growth rate (CAGR) of Z% over the next five years [1].
For instance, if COMPRO treats a common field like autoimmune diseases, the global market exceeds $X billion, with rapidly expanding markets in North America and Asia-Pacific. Within the US, approximately N million patients could benefit, representing a sizable revenue base.
Competitive landscape
Key competitors include:
- Brand A: Established first-in-class, with 60% market share.
- Brand B: Generic versions, gaining market share due to price.
- Biosimilars: Emerging entrants expected to impact pricing.
The entry point of COMPRO depends on its differentiation, approval status, and reimbursement dynamics. Market share projections estimate COMPRO capturing 10-20% within three years post-launch, depending on its clinical advantages and pricing strategy.
Regulatory and reimbursement considerations
Approval status from agencies such as FDA or EMA influences market access and pricing. Reimbursement policies vary across regions, with payers increasingly emphasizing value-based models, which impact net prices.
Pricing dynamics
Pricing strategies employ value-based assessments, considering:
- Efficacy and safety profile: Superior outcomes justify premium pricing.
- Patent exclusivity: Current exclusivity extends typically 10-12 years, protecting pricing power.
- Market competition: Generics and biosimilars tend to halve prices over time.
Initial launch prices in the US for comparable drugs range between $XX,XXX and $XX,XXX per annum per patient, based on mechanism, dosage, and administrative burden [2].
Price projections over time
| Year | Price Range (per patient/year) | Notes |
|---|---|---|
| Year 1 | $XX,XXX - $YY,YYY | Launch price set based on value proposition |
| Year 3 | $XX,XXX - $ZZ,ZZZ | Adjusted for competition and reimbursement pressures |
| Year 5 | $X,XXX - $AA,AAA | Possible biosimilar entry influences price reduction |
Declines of 10-25% occur with biosimilar entry, especially in markets like Europe, where price sensitivity is high.
Revenue forecasts
Based on projected market penetration and pricing, revenue for COMPRO is estimated as follow:
- Year 1: $X million
- Year 3: $Y million
- Year 5: $Z million
These figures incorporate market share assumptions, pricing adjustments, and patient adherence rates.
Key influencers on market potential and pricing
- Clinical differentiation: Better efficacy or safety can command premium prices.
- Regulatory outcomes: Fast-track or orphan drug designations influence market access.
- Pricing strategies: Tiered pricing may optimize revenue across regions.
- Manufacturer's market power: Patent protections prolong exclusivity, maintaining higher prices.
Summary of risks and opportunities
Risks include biosimilar competition, regulatory delays, and reimbursement challenges. Opportunities arise from unmet medical needs, pricing premiums for superior outcomes, and emerging markets that lack established therapies.
Key Takeaways
- The global market size for COMPRO’s indication is estimated at $X billion, with significant growth expected.
- The initial pricing range in developed countries likely falls between $XX,XXX and $XX,XXX per patient annually.
- Market penetration depends on clinical differentiation, reimbursement environment, and competition timing.
- Price declines of 10-25% are expected within 3-5 years, mainly due to biosimilar entry.
- Revenue projections suggest a potential of $X million in Year 1, increasing to $Z million by Year 5, housing considerable upside if differentiation and adoption targets are met.
FAQs
1. How does patent expiration impact COMPRO’s pricing?
Patent expiration typically leads to generic or biosimilar entry, reducing prices by 50% or more within 2-3 years, affecting revenue and market share.
2. What regions are most lucrative for COMPRO?
North America and Europe currently represent the largest and most profitable markets due to higher per-patient pricing and established reimbursement pathways.
3. How do pricing strategies differ between developed and emerging markets?
Developed markets favor premium pricing based on clinical benefits, while emerging markets often implement tiered or lower pricing to facilitate access.
4. What is the impact of biosimilars on COMPRO’s market?
Biosimilars can reduce prices by up to 50%, eroding margins but also expanding overall market size by making the therapy available to more patients.
5. How does clinical differentiation influence the price premium?
Superior efficacy, safety, or dosing convenience enables companies to set higher prices, often justified through health economic evaluations.
References
[1] MarketResearch.com. (2022). Global market size and projections for autoimmune therapies.
[2] IQVIA Institute. (2022). Pricing and reimbursement landscape for innovative biologics.
More… ↓
