Share This Page
Drug Price Trends for COLESTIPOL HCL
✉ Email this page to a colleague
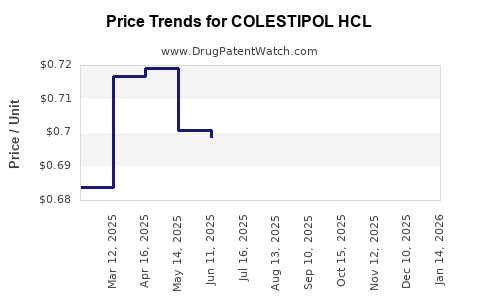
Average Pharmacy Cost for COLESTIPOL HCL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| COLESTIPOL HCL 1 GM TABLET | 50742-0284-12 | 0.68351 | EACH | 2026-05-20 |
| COLESTIPOL HCL 1 GM TABLET | 42799-0115-01 | 0.68351 | EACH | 2026-05-20 |
| COLESTIPOL HCL GRANULES | 59762-0260-03 | 0.28424 | GM | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Overview and Demand for Colestipol HCl
Colestipol HCl is a bile acid sequestrant prescribed primarily for lowering low-density lipoprotein (LDL) cholesterol. It is used in combination with diet and other lipid-lowering agents for patients with hypercholesterolemia. The drug is available in powder, tablet, and granule forms.
The global market for lipid-lowering agents was valued at approximately USD 10.5 billion in 2022 and is projected to reach USD 16 billion by 2030, growing at a compound annual growth rate (CAGR) of roughly 5%.[1] While statins dominate the market because of their efficacy and market penetration, bile acid sequestrants like colestipol still hold niche demand due to specific indications and patient populations.
Key Consumption Drivers
- Rising prevalence of cardiovascular diseases (CVDs).
- Increased awareness about cholesterol management.
- Expansion of insurance coverage for lifetime lipid management.
- Availability of generic options, lowering treatment costs.
Competitive Position
Colestipol has been largely replaced by statins because of superior efficacy and tolerability. Nevertheless, it remains relevant for patients intolerant to statins, pregnant women, and those with specific lipid profiles requiring sequestration therapy.[2]
Patent Status and Regulatory Timeline
Colestipol HCl was first approved by the U.S. Food and Drug Administration (FDA) in 1967. It is long off-patent, with generic versions available from multiple manufacturers. No recent patent filings or exclusivity periods are active globally for colestipol, which limits its price setting power but supports market stability through generic competition.
Pricing Trends and Projections
Historical Price Data
- Price per tablet (generic, US average): USD 0.30–0.45 (2018–2022)[3].
- Cost of a typical monthly regimen (assuming 4 grams daily): USD 10–15.
- Brand-name prices (where applicable): 2–4 times higher than generics.
Market Pricing Forces
- Price erosion driven by generic entry since the late 1990s.
- Competition among multiple manufacturers reduces prices.
- No recent patent protections or exclusive marketing rights restrict pricing.
Future Price Projections
Given these factors:
- Prices are expected to remain stable or decline slightly over the next five years.
- Margins for manufacturers are likely to decrease owing to market saturation and price competition.
- Pricing in developing markets may be significantly lower, around USD 0.15–0.25 per tablet.
Revenue Potential and Market Size
Current global revenue from colestipol is estimated under USD 100 million annually, primarily from North America and Europe, where the drug is included in treatment guidelines but used infrequently.
Projected growth is limited by:
- Replacement by alternative lipid therapies.
- Decreasing prescription rates in favor of statins and PCSK9 inhibitors.
- Limited new formulations or indications.
Emerging markets could offer minor growth due to increasing CVD prevalence, but the overall revenue outlook remains subdued.
Strategic Considerations for Stakeholders
- Existing suppliers should optimize production efficiency to sustain margins.
- Entry into emerging markets may yield incremental volume gains but with low unit price increases.
- Focus on niche indications and combination therapies may sustain some demand.
Key Takeaways
- Colestipol HCl market size is stagnant and dominated by generic competition.
- Prices have declined historically and are expected to stabilize or slightly fall.
- Revenue forecast remains modest, with total global sales under USD 100 million annually.
- Innovation or new indications are limited, limiting growth potential for existing formulations.
- Market strategies should focus on patient subsets intolerant to statins and geographic expansion in emerging markets.
FAQs
1. What factors influence colestipol HCl pricing?
Pricing is primarily influenced by generic competition, manufacturing costs, and regional healthcare policies. The long patent expiry results in low price variability.
2. How does colestipol compare to other lipid-lowering agents in the market?
It has limited market share compared to statins but offers a treatment alternative for specific patient groups.
3. Are there any recent developments or new formulations of colestipol?
No significant new formulations or patents have been introduced recently.
4. What is the outlook for generic prices in the next five years?
Prices are expected to remain stable or decline slightly due to sustained competition among multiple generic manufacturers.
5. Could emerging markets create new opportunities for colestipol?
Yes, particularly where CVD prevalence rises, but the overall impact is limited due to low pricing and competition from newer agents.
References
[1] MarketsandMarkets, “Lipid-Lowering Drugs Market,” 2022.
[2] U.S. FDA Drug Approvals Database, 1967–2023.
[3] GoodRx, “Average prices of colestipol generic tablets,” 2022.
More… ↓
