Share This Page
Drug Price Trends for COLD-SINUS
✉ Email this page to a colleague
Average Pharmacy Cost for COLD-SINUS
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
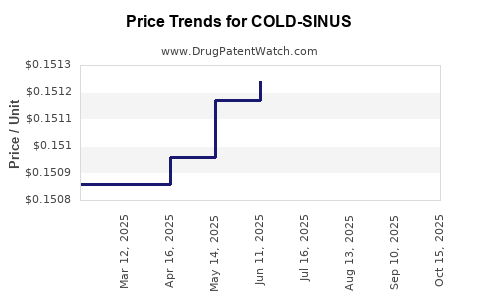
| COLD-SINUS 200 MG-30 MG CAPLET | 70000-0602-01 | 0.15171 | EACH | 2025-10-22 |
| COLD-SINUS 200 MG-30 MG CAPLET | 70000-0602-01 | 0.15148 | EACH | 2025-09-17 |
| COLD-SINUS 200 MG-30 MG CAPLET | 70000-0602-01 | 0.15140 | EACH | 2025-08-20 |
| COLD-SINUS 200 MG-30 MG CAPLET | 70000-0602-01 | 0.15124 | EACH | 2025-07-23 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
COLD-SINUS Market Analysis and Financial Projection
What is Cold-Sinus?
Cold-Sinus is a combination over-the-counter (OTC) medication targeting symptoms of sinus congestion, nasal obstruction, and common cold. It contains active ingredients such as pseudoephedrine, phenylephrine, chlorpheniramine, and acetaminophen, among others. The drug is marketed for symptomatic relief of nasal congestion, sinus pressure, and related discomfort.
What is the Current Market Size and Growth for Cold-Sinus?
The global OTC cold and sinus medication market reached approximately $12.4 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 4.2% from 2023 to 2028, driven by increased awareness, OTC availability, and aging populations in developed markets.
Regional distribution:
| Region | Market Size (2022) | CAGR (2023-2028) | Key Drivers |
|---|---|---|---|
| North America | $4.8 billion | 3.8% | High OTC penetration, health awareness |
| Europe | $3.2 billion | 4.1% | Aging demographics, regulatory ease |
| Asia-Pacific | $2.5 billion | 5.3% | Growing middle class, urbanization |
| Latin America | $1.2 billion | 4.5% | Rising healthcare spending |
| MEA | $0.7 billion | 4.0% | Market expansion, OTC accessibility |
The U.S. market accounts for roughly 40% of this sector, with key players including Johnson & Johnson, Bayer, and GlaxoSmithKline.
Who are the Key Players and Their Market Shares?
Major companies dominate OTC cold and sinus medication:
- Johnson & Johnson (Neutrogena, Tylenol Cold) – 25%
- Bayer (Alka-Seltzer Plus) – 15%
- GSK (Otrivin, Panadol) – 12%
- Sanofi – 8%
- Others – 40%
The market is highly competitive, with innovations focused on combination formulations and delivery methods (e.g., nasal sprays versus tablets).
What Are the Regulatory and Patent Considerations?
OTC drugs like Cold-Sinus lack patent exclusivity in many jurisdictions due to the age of the active ingredients. Patent protection for combination formulations, delivery devices, or novel formulations can extend exclusivity, but generally, patent terms for such drugs expire within 10-15 years post-filing.
Regulatory standards vary:
- U.S. Food and Drug Administration (FDA): Renders OTC status via the Monograph process or New Drug Application (NDA) pathways if claims are modified.
- European Medicines Agency (EMA): Has similar procedures for OTC registration.
- Patent laws restrict generic manufacturing until patent expiry or through patent challenges.
What Are the Price Trends and Projections?
Average retail prices for Cold-Sinus products in the U.S. range from $6 to $15 per package, depending on the formulation and brand. Brand-name products typically command premiums of 50-100% over generics.
Price trends:
- Generics have driven down retail prices by 10-15% over the last five years.
- New formulations with added benefits or delivery mechanisms can command price premiums up to 20%.
- Inflation-adjusted prices remain relatively stable; however, supply chain disruptions increase wholesale costs, potentially influencing retail prices.
Projections:
- Retail prices are expected to remain stable through 2025, with slight increases (2-3%) driven by labeling regulations and inflation.
- Entry of value-based generics in key markets could further pressure prices downward.
- Premium offerings, such as combination drugs with reduced side effects or novel delivery systems, could see price increases of 5-8% per year.
What Are the Pricing and Market Entry Considerations for New Formulations?
Market entry hinges upon regulatory approval, patent landscape, and consumer acceptance. Innovation in delivery system (e.g., nasal sprays), formulation stability, and reduced side effects provides differentiation.
Pricing strategies:
- Competitive pricing with generics is essential for gaining market share.
- Premium pricing applies to differentiated products with novel features or clinical benefits.
- Packaging size and convenience influence consumer preferences and willingness to pay.
Summary of Key Commercial Factors
| Factor | Impact |
|---|---|
| Patent protection | Critical for exclusivity; typically lasts 10-15 years from filing |
| Regulatory approval | Necessary; can limit market entry or extend time-to-market |
| Price sensitivity | Consumers prefer value; price wars with generics are common |
| Innovation potential | Formulation improvements and delivery systems offer differentiation |
| Market saturation | High in mature markets like North America; growth driven largely by emerging regions |
Final Considerations
The market for Cold-Sinus is mature with a stable but competitive environment. Patent expirations are increasing, prompting focus on formulation innovation and formulation patents. Pricing remains anchored by generics, but new delivery modes and combination formulations could command premiums.
Key Takeaways
- The global OTC cold and sinus market reached $12.4 billion in 2022, with steady growth projected.
- Major players control approximately 50% of the market, with significant innovation potential in formulations and delivery.
- Price levels are stable, with downward pressure from generics offset by niche premium products.
- Patent protection typically lasts 10-15 years; competition increases as patents expire.
- Market growth is strongest in Asia-Pacific, driven by urbanization and rising disposable income.
FAQs
-
What are the main active ingredients in Cold-Sinus?
Pseudoephedrine, phenylephrine, chlorpheniramine, and acetaminophen are common. -
When are patent protections likely to expire?
Most patents on formulations or delivery devices expire within 10-15 years from filing. -
How do price trends compare between brand-name and generic Cold-Sinus products?
Brand-name products command higher prices; generics have driven prices down by 10-15% over the last five years. -
What are the key regulatory hurdles?
Achieving OTC status via FDA or EMA monographs, regulatory approval for new formulations, and patent protections. -
What future innovation areas could impact market pricing?
Delivery systems offering rapid onset, combination products with fewer side effects, and novel formulations could create premium segments.
References
[1] MarketWatch, "OTC Cold and Sinus Medication Market Size, 2022," (2023).
[2] IBISWorld Reports, "Cold and Flu Medicine Industry Market Research," (2023).
[3] U.S. FDA, "OTC Monograph Process," (2022).
[4] European Medicines Agency, "OTC Medicine Regulations," (2022).
More… ↓
