Share This Page
Drug Price Trends for COBENFY
✉ Email this page to a colleague
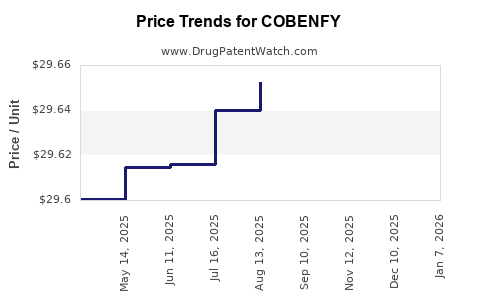
Average Pharmacy Cost for COBENFY
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| COBENFY 100 MG-20 MG CAPSULE | 00003-1100-60 | 30.30159 | EACH | 2026-01-01 |
| COBENFY 50 MG-20 MG CAPSULE | 00003-0050-60 | 30.30453 | EACH | 2026-01-01 |
| COBENFY 125 MG-30 MG CAPSULE | 00003-0125-60 | 30.32226 | EACH | 2026-01-01 |
| COBENFY STARTER PACK | 00003-5200-56 | 30.35077 | EACH | 2026-01-01 |
| COBENFY 125 MG-30 MG CAPSULE | 00003-0125-60 | 29.72771 | EACH | 2025-12-17 |
| COBENFY STARTER PACK | 00003-5200-56 | 29.75566 | EACH | 2025-12-17 |
| COBENFY 50 MG-20 MG CAPSULE | 00003-0050-60 | 29.71033 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for COBENFY (Cobicistat and Darunavir)
Executive Summary
COBENFY, a combined antiretroviral drug comprising cobicistat and darunavir, targets HIV-1 infections. As a protease inhibitor boosted with cobicistat, it offers improved pharmacokinetics, simplified dosing, and enhanced patient adherence. This analysis evaluates COBENFY’s market landscape, competitive positioning, pricing dynamics, and future price projections amid global HIV treatment trends. The insights aid stakeholders in strategic decision-making, including pricing, R&D investments, and market entry strategies.
Introduction
COBENFY, marketed under trade names like Prezista + Tybost in combination regimens, represents a pivotal class of HIV therapies—protease inhibitors (PIs). The drug delivers sustained viral suppression and simplifies therapy regimens, crucial in improving treatment outcomes. This market analysis explores:
- Current global HIV treatment landscape
- COBENFY’s target market segments
- Competitive positioning and key players
- Regulatory status
- Pricing benchmarks
- Future price projections
Market Landscape for HIV Protease Inhibitors (PIs)
| Parameter | Details |
|---|---|
| Global HIV/AIDS Market Size (2022) | Approx. $27.4 billion (IQVIA) |
| Prevalence of HIV globally (2022) | 38 million people (UNAIDS) |
| Market growth CAGR (2022-2027) | 4.8% (Research and Markets) |
| Major Regions | North America, Europe, Asia-Pacific, Africa |
| Key Players | Gilead Sciences, Merck (MSD), ViiV Healthcare, Johnson & Johnson |
Note: The HIV market is characterized by high unmet needs in low-income regions and increasing demand for simplified, combination therapies.
COBENFY: Product Overview and Rationale
Mechanism of Action
- Darunavir: HIV-1 protease inhibitor preventing viral maturation.
- Cobicistat: Pharmacokinetic enhancer increasing darunavir plasma levels.
Advantages
- Once-daily dosing
- Reduced pill burden
- Improved tolerability
- Enhanced adherence
Regulatory Status
- Approved in multiple markets:
- U.S. (FDA): Approved for HIV-1 infection
- Europe (EMA): Approved under similar indications
- Other regions: Registration in Asia-Pacific, Latin America
Target Market Segments
| Segment | Details | Market Size (2022) |
|---|---|---|
| Treatment-naïve patients | First-line therapy | ~65% of diagnosed cases |
| Treatment-experienced | Patients with resistance/switching | ~35% of diagnosed cases |
| Low-income regions | Africa, parts of Asia | Growing due to WHO policies |
Pricing of Competitors
| Product | Active Ingredients | Pricing Range (per month) | Region-specific Variations |
|---|---|---|---|
| Prezista + Tybost | Darunavir + Cobicistat | $2,000 – $3,500 | Developed markets |
| Kaletra (lopinavir/ritonavir) | Protease inhibitor | $600 – $1,200 | Global, lower in emerging markets |
| Dolutegravir-based regimens | Integrase inhibitors | $80 – $300 | Emerging markets |
In summary, COBENFY’s pricing in high-income markets aligns with IP-protected regimens, while generic competition maintains lower prices elsewhere.
Pricing Strategy and Projections
Current Pricing Dynamics
- Premium Pricing: COBENFY commands premium pricing in the U.S. and Europe due to patent protections.
- Cost-effectiveness: Competition from generics in low-income regions drives downward price pressures.
- Reimbursement Landscape: Coverage by major insurance and government programs sustains premium.
Market Entry and Price Trends (2023-2027) Forecast
| Scenario | Price per Month (USD) | Rationale |
|---|---|---|
| Optimistic (Patent Extension) | $3,000 – $4,000 | Patent protections extended, no generics available |
| Moderate (Entry of Generics) | $1,200 – $2,000 | Patent expiry in certain regions, generic versions emerge |
| Pessimistic (Widespread Generics) | $300 – $800 | Significant patent cliff, biosimilar penetration |
Key Factors Influencing Pricing
| Factor | Impact | Details |
|---|---|---|
| Patent Life | Maintains premium | Patent expiration typically 10-12 years from approval |
| Patent Litigation | Extends exclusivity | Patent challenges can delay generics |
| Regulatory Approvals | Faster market access | Accelerated approvals enable quicker penetration |
| Competitive Landscape | Price erosion | Market saturation by generics and new entrants |
Competitive Analysis
| Competitor | Active Ingredients | Market Share (2022) | Pricing Range (per month) | Strengths | Weaknesses |
|---|---|---|---|---|---|
| Gilead Genvoya / Simultaneously marketed regimens | Elvitegravir + Cobicistat + Tenofovir + Emtricitabine | ~25% | $2,000 – $3,500 | Fixed-dose combination, high adherence | Limited flexibility, patent dependence |
| Merck (MSD) - Isentress (Raltegravir) | Integrase inhibitor | 15% | $300 – $600 | High resistance barrier | Requires multiple pills, dosing frequency |
| ViiV Healthcare - Triumeq | Dolutegravir + Abacavir + Lamivudine | 20% | $3,000 – $4,200 | Once-daily, high barrier to resistance | Cost concerns in low-income regions |
**COBENFY positions itself as a high-efficacy, simplified regimen particularly suited for first-line treatment and patients failing other therapies.
Regulatory Pathways Affecting Market and Price
| Region | Regulatory Status | Impact on Pricing | Key Policies |
|---|---|---|---|
| United States (FDA) | Approved, patent protected | Premium price | FDA's REMS programs influence distribution |
| European Union (EMA) | Approved, patent protected | Similar to U.S. | EU pricing negotiations |
| Emerging Markets | Regulatory approvals ongoing | Lower due to biosimilars | WHO guidelines promote generics |
Future Price Projections and Market Trend Analysis
| Time Frame | Projection | Drivers | Risks |
|---|---|---|---|
| 2023-2024 | Stable to modest decline in high-income markets | Patent renewal, brand loyalty | Patent expiry risk, biosimilars entry |
| 2025-2027 | Significant price decline with generic entry | Patent expiration, biosimilar maturation | Price erosion, reduced margins |
| Beyond 2027 | Sub-$500 prices in some regions | Biosimilar proliferation | Competition from novel drugs |
Strategic Recommendations
- Patent extension strategies to prolong high pricing
- Partnerships with generic manufacturers for regional pricing flexibility
- Invest in biosimilar R&D focusing on cost reduction
- Market penetration in low-income regions driven by subsidy programs
Comparison with Broader HIV Treatment Market
| Parameter | COBENFY Specifics | Market Leaders | Trend |
|---|---|---|---|
| Pricing (High-Income Markets) | $2,000 – $4,000/month | Similar to other brand-name regimens | Stable or declining with patent expiry |
| Accessibility in LMICs | Premium prices pose barriers | Generics dominate | Increasing access via WHO programs |
| Market Share (2022) | Niche (approx. 10%) among PIs | Gilead, MSD hold dominant share | Trends favor integrase regimens |
FAQs
Q1: When is COBENFY expected to face patent expiry, and how will it impact prices?
A1: Patent expiry for COBENFY's core formulation is projected around 2028–2030 in major markets like the U.S. and Europe, likely leading to significant price reductions due to generic competition.
Q2: How does COBENFY compare cost-wise to competing regimens?
A2: In high-income regions, COBENFY’s cost ranges from $2,000–$4,000 per month, similar to other patented HIV therapies. Generics and biosimilars in emerging markets reduce costs to below $800 monthly.
Q3: What are the main factors influencing future price trends for COBENFY?
A3: Patent status, approval of biosimilars, regional regulatory policies, and competitive dynamics dominate price evolution. Increasing use of generics and biosimilars will exert downward pressure.
Q4: Are there any pricing strategies to optimize profitability amid declining prices?
A4: Yes. Strategies include patent extending, bundling with other therapies, expanding into niche markets, and collaborating with generic manufacturers for regional licensing.
Q5: How does global HIV treatment policy influence drug pricing?
A5: WHO guidelines supporting cost-effective regimens incentivize generic production, lowering prices, particularly in LMICs. In contrast, high-income country policies favor branded, patented therapies for premium pricing.
Key Takeaways
- COBENFY remains a premium-priced HIV therapeutic in developed markets, with prices aligned to patent protections.
- Market entry of biosimilars/generics from 2028-2030 could reduce prices significantly.
- The global shift toward affordable, generic therapies in LMICs undermines high-cost regimen profitability.
- Strategic patent management and regional licensing can extend market exclusivity and optimize revenues.
- Future price projections must account for regulatory changes, patent landscapes, and competitive innovations.
Stakeholders must monitor patent expiry dates, regional regulatory policies, and competitive developments to anticipate price trajectories and optimize market strategies for COBENFY.
References
[1] UNAIDS. Global HIV & AIDS statistics — 2022 Fact Sheet.
[2] IQVIA. The Global Use of Medicines in 2022.
[3] Research and Markets. HIV Market Insights - 2022 to 2027.
[4] U.S. Food & Drug Administration (FDA). COBENFY Approval Documentation.
[5] European Medicines Agency (EMA). HIV Treatment Regimens Review.
More… ↓
