Share This Page
Drug Price Trends for CLOZAPINE
✉ Email this page to a colleague
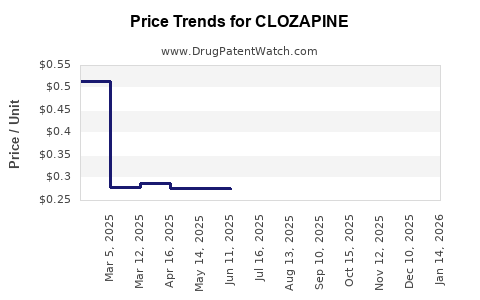
Average Pharmacy Cost for CLOZAPINE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CLOZAPINE ODT 25 MG TABLET | 59651-0261-01 | 1.64454 | EACH | 2026-03-18 |
| CLOZAPINE 100 MG TABLET | 00378-0860-01 | 0.62842 | EACH | 2026-03-18 |
| CLOZAPINE 100 MG TABLET | 00378-0860-05 | 0.62842 | EACH | 2026-03-18 |
| CLOZAPINE 100 MG TABLET | 16729-0142-01 | 0.62842 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for CLOZAPINE
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| CLOZARIL 100MG TAB | HLS Therapeutics (USA), Inc. | 69809-0127-05 | 100 | 1498.88 | 14.98880 | EACH | 2023-01-01 - 2027-06-30 | FSS |
| CLOZARIL 25MG TAB | HLS Therapeutics (USA), Inc. | 69809-0126-05 | 100 | 578.53 | 5.78530 | EACH | 2023-01-01 - 2027-06-30 | FSS |
| CLOZARIL 100MG TAB | HLS Therapeutics (USA), Inc. | 69809-0127-05 | 100 | 1151.06 | 11.51060 | EACH | 2024-01-01 - 2027-06-30 | Big4 |
| CLOZARIL 25MG TAB | HLS Therapeutics (USA), Inc. | 69809-0126-05 | 100 | 342.81 | 3.42810 | EACH | 2024-01-01 - 2027-06-30 | Big4 |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Clozapine Market Analysis and Price Projections
Clozapine, an atypical antipsychotic, holds a significant position in the treatment of treatment-resistant schizophrenia. Its efficacy, particularly in reducing suicidal behavior, has solidified its market presence despite its adverse effect profile. The market is characterized by a stable demand, driven by the chronic nature of schizophrenia and the drug's established treatment protocol. Key factors influencing market dynamics include generic competition, regulatory landscapes, and the development of biosimil or alternative therapies. Price projections indicate continued stability, with minor fluctuations driven by manufacturing costs and market access initiatives.
What is the current global market size and projected growth rate for Clozapine?
The global clozapine market size was valued at approximately $850 million in 2023. Projections indicate a compound annual growth rate (CAGR) of 3.5% to 4.0% over the next five years, reaching an estimated $1.05 billion to $1.10 billion by 2028. This growth is primarily sustained by the persistent need for effective treatment options for treatment-resistant schizophrenia.
Key Market Drivers
- Prevalence of Treatment-Resistant Schizophrenia: Schizophrenia affects an estimated 24 million people worldwide. A significant subset of these patients, estimated to be 15% to 30%, do not respond adequately to standard antipsychotic treatments, making clozapine a critical therapeutic option.
- Established Efficacy Profile: Clozapine demonstrates superior efficacy in reducing positive and negative symptoms of schizophrenia, and notably, it is the only antipsychotic with proven effectiveness in reducing suicidal ideation and behavior in patients with schizophrenia.
- Global Access and Reimbursement: In many developed and developing nations, clozapine is a covered medication, facilitating consistent patient access. Reimbursement policies, while varying, generally support its use in indicated patient populations.
- Continuous Clinical Use: The drug’s long history of clinical use has established it as a reliable, albeit monitored, treatment. Healthcare providers are familiar with its dosing, administration, and management of side effects.
Market Restraints
- Adverse Effect Profile: Clozapine's significant side effect profile, including agranulocytosis, myocarditis, and metabolic disturbances, necessitates rigorous patient monitoring. This requires specialized healthcare infrastructure and patient adherence programs, which can limit its accessibility in resource-constrained settings.
- Generic Competition: The market is largely dominated by generic manufacturers following patent expiries. This intense generic competition places downward pressure on pricing.
- Limited New Entrants: The high regulatory hurdles and the specialized monitoring required for clozapine limit the entry of new companies into the primary manufacturing space.
What is the competitive landscape for Clozapine?
The clozapine market is characterized by a fragmented supply chain primarily driven by generic manufacturers. Key players focus on cost-effective production and efficient distribution.
Major Manufacturers and Generic Suppliers:
- Teva Pharmaceutical Industries: A significant global producer of generic clozapine.
- Sun Pharmaceutical Industries: A major Indian pharmaceutical company with a substantial presence in generic antipsychotic markets.
- Mylan N.V. (now Viatris): A prominent supplier of generic clozapine formulations.
- Lannett Company: Another key player in the U.S. generic clozapine market.
- Other Regional Manufacturers: Numerous smaller manufacturers operate in specific geographic regions, supplying localized markets.
Product Formulations:
The primary oral dosage forms include tablets and orally disintegrating tablets (ODTs). ODTs offer improved patient compliance, particularly for individuals who have difficulty swallowing pills.
Therapeutic Alternatives:
While clozapine remains the gold standard for treatment-resistant schizophrenia, research into alternative therapies is ongoing. These include:
- Investigational Drugs: Several novel compounds are in various stages of clinical trials for treatment-resistant schizophrenia, aiming for improved efficacy or a better safety profile.
- Adjunctive Therapies: Combinations of existing antipsychotics with other medications are explored to enhance treatment outcomes.
What are the key pricing trends and factors influencing Clozapine prices?
Clozapine pricing is predominantly influenced by generic competition, manufacturing costs, and market access strategies. The market has stabilized due to the mature generic landscape.
Average Wholesale Price (AWP) Trends:
- Historical Trend: Over the past decade, the AWP for generic clozapine has seen a gradual decline due to increased competition among manufacturers.
- Current Pricing: In the United States, the average AWP for a 100mg clozapine tablet ranges from $1.00 to $3.00, depending on the manufacturer and volume. For a 30-day supply, this translates to an average of $90 to $270.
- Global Variations: Prices vary significantly by region, influenced by local manufacturing capabilities, import duties, and national healthcare pricing regulations. Developing countries often have lower prices due to local generic production and government procurement policies.
Key Pricing Factors:
- Manufacturing Costs: The cost of raw materials, synthesis, formulation, and quality control are fundamental to pricing. Companies with optimized supply chains and efficient manufacturing processes can offer more competitive pricing.
- Generic Competition: The presence of multiple generic manufacturers creates price pressure. Manufacturers must remain competitive to retain market share.
- Regulatory Compliance Costs: Adhering to stringent Good Manufacturing Practices (GMP) and the specific regulatory requirements for clozapine, including patient monitoring programs, adds to manufacturing and distribution costs.
- Reimbursement Policies: Payer coverage and formulary placements significantly impact patient access and, indirectly, pricing strategies. Manufacturers may offer rebates or discounts to secure preferred formulary status.
- Supply Chain Dynamics: Disruptions in the supply of active pharmaceutical ingredients (APIs) or finished products can lead to temporary price fluctuations.
- Patient Assistance Programs: To improve access for uninsured or underinsured patients, manufacturers may offer patient assistance programs, which can influence net pricing after discounts and rebates.
What are the regulatory considerations and their impact on the Clozapine market?
Regulatory oversight for clozapine is exceptionally stringent due to its severe adverse effect profile. These regulations directly impact manufacturing, distribution, and patient access.
Key Regulatory Aspects:
- Risk Evaluation and Mitigation Strategies (REMS): In the United States, the Clozapine REMS program is a mandatory federal program designed to mitigate the risk of agranulocytosis and other serious adverse events. This program requires healthcare providers to be certified, patients to be enrolled, and regular blood monitoring to be conducted.
- Program Components: Prescribers must be certified to prescribe clozapine, and pharmacies must be certified to dispense it.
- Monitoring Frequency: Typically, initial monitoring involves weekly complete blood counts (CBCs) for the first six months, followed by bi-weekly CBCs for the next six months, and then monthly CBCs thereafter, provided no significant abnormalities are detected.
- Impact: The REMS program adds significant administrative burden and cost for healthcare systems and manufacturers, but it is essential for ensuring patient safety and maintaining market access.
- International Regulatory Bodies: Similar stringent oversight exists in other major markets, enforced by agencies like the European Medicines Agency (EMA) and Health Canada. These bodies mandate specific pharmacovigilance and patient safety protocols.
- Manufacturing Standards: Manufacturers must adhere to rigorous GMP standards to ensure product quality, safety, and efficacy. This includes strict quality control measures for API sourcing and finished product manufacturing.
- Labeling and Prescribing Information: Regulatory agencies require detailed labeling that clearly outlines the drug's risks, benefits, and monitoring requirements. This information is crucial for healthcare professionals prescribing the medication.
Impact on the Market:
- Barriers to Entry: The complexity and cost associated with complying with REMS and other safety regulations create significant barriers for new manufacturers.
- Increased Operational Costs: For existing manufacturers and healthcare providers, ongoing compliance with monitoring and reporting requirements increases operational expenses.
- Controlled Market Access: While essential for safety, stringent regulations can sometimes limit the speed and ease of patient access, particularly in regions with less robust healthcare infrastructure.
- Focus on Quality and Safety: The regulatory environment reinforces a market where quality and safety compliance are paramount, differentiating manufacturers based on their ability to consistently meet these standards.
What are the future outlook and potential disruptors for the Clozapine market?
The future of the clozapine market is expected to be stable, with growth driven by its established efficacy. However, several potential disruptors could influence its trajectory.
Future Outlook:
- Sustained Demand: The unmet need in treatment-resistant schizophrenia will continue to support a consistent demand for clozapine.
- Incremental Improvements: Manufacturers may focus on process optimization and cost efficiencies to maintain competitiveness within the generic market.
- Geographic Expansion: Increased adoption in emerging markets, as healthcare infrastructure and access to monitoring improve, could contribute to growth.
Potential Disruptors:
- Development of Novel Therapies:
- New Chemical Entities (NCEs): Breakthroughs in drug discovery for schizophrenia that offer comparable or superior efficacy with a significantly improved safety profile could displace clozapine for some patient segments.
- Biologics and Gene Therapies: While in early research stages for schizophrenia, these modalities represent a long-term potential disruption.
- Advancements in Monitoring Technology:
- Point-of-Care Diagnostics: Development of rapid, reliable point-of-care blood tests for monitoring blood counts could streamline the REMS process, making clozapine treatment more accessible and less burdensome.
- Digital Health Solutions: Telemedicine platforms and digital adherence tools could improve patient engagement and monitoring compliance, potentially expanding clozapine's reach.
- Policy Shifts:
- Changes in Reimbursement: Significant shifts in government or private payer reimbursement policies could alter market dynamics, either by expanding or restricting coverage.
- Regulatory Streamlining: While unlikely to compromise safety, any future streamlining of REMS processes could reduce administrative burdens and potentially lower costs.
- Alternative Treatment Modalities:
- Neuromodulation Techniques: Techniques like Transcranial Magnetic Stimulation (TMS) are being explored as adjuncts or alternatives for treatment-resistant psychiatric conditions. Their efficacy in schizophrenia requires further validation.
The market will likely remain dominated by generic clozapine for the foreseeable future, with growth tied to disease prevalence and access improvements. Disruptions would require significant advancements in therapeutic alternatives or a fundamental shift in treatment paradigms.
Key Takeaways
- The global clozapine market is valued at approximately $850 million and projected to grow at a CAGR of 3.5-4.0% to $1.05-$1.10 billion by 2028, driven by its critical role in treatment-resistant schizophrenia.
- The market is highly competitive, characterized by numerous generic manufacturers such as Teva, Sun Pharma, and Viatris, focusing on cost-effective production.
- Clozapine pricing is influenced by generic competition, manufacturing costs, and stringent regulatory compliance, with average wholesale prices for a 100mg tablet ranging from $1.00 to $3.00 in the U.S.
- Strict regulatory oversight, particularly the REMS program in the U.S., is a significant factor, increasing operational costs and creating barriers to entry while ensuring patient safety.
- Future growth will likely be stable, with potential disruptions arising from novel therapeutic alternatives, advancements in monitoring technology, and shifts in healthcare policy.
Frequently Asked Questions
- What is the primary reason for clozapine's high regulatory scrutiny compared to other antipsychotics? Clozapine's association with potentially fatal agranulocytosis, requiring intensive blood monitoring, mandates the highest level of regulatory oversight, including mandatory REMS programs in many jurisdictions.
- How does the cost of clozapine monitoring impact its overall accessibility? The mandatory and frequent blood monitoring associated with clozapine adds significant costs to patient care, influencing accessibility, particularly in healthcare systems with limited resources.
- Are there any patented, branded versions of clozapine still available, or is the market exclusively generic? While the original patents have long expired, leading to a predominantly generic market, some manufacturers might offer branded generic formulations or have specific market exclusivity periods for new dosage forms or delivery systems. However, the market is largely driven by generic competition.
- What is the typical duration of clozapine treatment for patients with treatment-resistant schizophrenia? Clozapine treatment is generally considered long-term, often lifelong, for patients who respond positively and tolerate the medication, as schizophrenia is a chronic condition and clozapine is a foundational treatment for severe, resistant cases.
- Can advancements in genetic testing potentially influence clozapine prescribing or monitoring protocols in the future? While not currently standard practice for routine clozapine prescribing, research into pharmacogenomics is ongoing. Future advancements in genetic testing could potentially identify patients at higher risk for specific adverse effects, leading to more personalized monitoring strategies.
More… ↓
