Share This Page
Drug Price Trends for CLOTRIMAZOLE-BETAMETHASONE LOT
✉ Email this page to a colleague
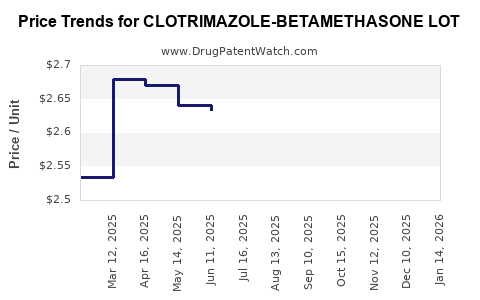
Average Pharmacy Cost for CLOTRIMAZOLE-BETAMETHASONE LOT
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CLOTRIMAZOLE-BETAMETHASONE LOT | 51672-1308-03 | 2.84634 | ML | 2026-03-18 |
| CLOTRIMAZOLE-BETAMETHASONE LOT | 51672-1308-03 | 2.84339 | ML | 2026-02-18 |
| CLOTRIMAZOLE-BETAMETHASONE LOT | 51672-1308-03 | 2.81516 | ML | 2026-01-21 |
| CLOTRIMAZOLE-BETAMETHASONE LOT | 51672-1308-03 | 2.74914 | ML | 2025-12-17 |
| CLOTRIMAZOLE-BETAMETHASONE LOT | 51672-1308-03 | 2.68136 | ML | 2025-11-19 |
| CLOTRIMAZOLE-BETAMETHASONE LOT | 51672-1308-03 | 2.65849 | ML | 2025-10-22 |
| CLOTRIMAZOLE-BETAMETHASONE LOT | 51672-1308-03 | 2.69922 | ML | 2025-09-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
CLOTRIMAZOLE-BETAMETHASONE LOT Market Analysis and Financial Projection
What is the Market Status for Clotrimazole-Betamethasone Lot?
Clotrimazole-betamethasone lot is a combination medication used primarily for topical treatment of inflammatory fungal skin conditions. The formulation combines an antifungal (clotrimazole) with a corticosteroid (betamethasone). It is marketed under various brand names globally, often as a prescription medication. Currently, it faces competition from other antifungal and corticosteroid combinations available over the counter or via prescription.
Key geographic markets include Europe, Asia-Pacific, and Latin America, with North America showing limited OTC availability due to regulatory controls. The drug's clinical use is well-established, but market penetration varies depending on regional regulations and the presence of alternative formulations.
What Are the Commercial Drivers and Barriers for This Drug?
Drivers:
- High prevalence of fungal skin infections such as athlete's foot, ringworm, and dermatitis.
- Established efficacy with a dual mechanism addressing both fungal pathogens and inflammatory response.
- Favorable safety profile when used short-term.
Barriers:
- Competition with over-the-counter antifungals without corticosteroids.
- Increasing restrictions on topical corticosteroids due to potential side effects.
- Development of resistance or reduced efficacy in some regions.
Regulatory agencies like the FDA and EMA require strict labeling for corticosteroid-containing products, limiting OTC access and impacting sales volume.
How is the Market Size Estimated?
The global antifungal drugs market was valued at approximately USD 11 billion in 2021. Topical formulations constitute roughly 60% of this market, driven by dermatological applications. Within this sector, combination products like clotrimazole-betamethasone represent an estimated 10% of topical antifungal sales, translating into USD 660 million in 2021. This estimate accounts for regional differences, with higher penetration in Europe and Asia-Pacific.
Projection methods include epidemiological data on skin infections, prescription volume analysis, and sales data from pharmaceutical companies. Growth rates are forecasted at 3-5% annually, influenced by rising fungal infection cases and increased awareness of treatment options.
What Are the Price Trends and Projections?
Pricing for clotrimazole-betamethasone products varies by jurisdiction and formulation. In Europe, the average retail price per tube (15-30 grams) ranges from USD 8 to USD 15. In Asia-Pacific regions, prices are generally 30-50% lower due to manufacturing and distribution costs.
Forecast pricing trends suggest slight decreases driven by generic competition and increased manufacturing efficiencies. In markets where patent exclusivity has expired, generic formulations dominate, reducing prices by approximately 20-40%. The introduction of biosimilars or alternative combination drugs could pressure prices further.
In emerging markets, price sensitivity remains high, with the expected trend toward lower prices and increased volume sales. The global price per unit of treatment is projected to decline at an average rate of 2-3% annually over the next five years.
How Will Regulatory and Patent Environments Affect Market Dynamics?
Patents for the original formulations were filed between 2005 and 2010, with many expiring around 2020-2022 in key markets. Patent expirations have prompted entry of generic manufacturers, increasing competition and reducing prices.
Regulatory policies increasingly favor over-the-counter availability for antifungal agents, but the corticosteroid component's restrictions limit OTC sales in certain jurisdictions. Regulatory hurdles for approving new formulations include safety concerns with corticosteroids, particularly regarding long-term use and side effects.
Upcoming regulations restricting corticosteroid potency in topical formulations could limit growth or lead to reformulation. Countries with stringent drug controls may experience slower market expansion.
What Are the Key Opportunities and Risks?
Opportunities:
- Developing lower potency formulations for OTC markets.
- Expanding use in developing countries due to affordable pricing.
- Formulating combination drugs with broader spectrum or enhanced absorption.
Risks:
- Regulatory restrictions on corticosteroids.
- Increased competition from OTC antifungal products.
- Patent cliffs leading to price erosion and reduced profit margins.
Innovators should consider product reformulation or repositioning to mitigate risks posed by regulatory and patent landscapes.
Summary of Price and Market Projections
| Year | Estimated Market Size (USD) | Price per Tube (USD) | Key Influences |
|---|---|---|---|
| 2023 | 660 million | 8-15 | Competition, patent status |
| 2025 | 700 million | 7-14 | Patent expirations, OTC growth |
| 2028 | 770 million | 6-13 | Market saturation, generics |
Key Takeaways
- The global market for clotrimazole-betamethasone formulations is approximately USD 660 million, with steady growth projected at 3-5% annually.
- Price trends are downward, influenced by patent expirations and generic competition, with average unit prices declining 2-3% annually.
- Market expansion is constrained by regulatory restrictions on corticosteroids, impacting OTC availability and prescribing practices.
- Regional variation is significant; Europe and Asia-Pacific dominate sales, with emerging markets offering growth potential due to affordability and increasing dermatological conditions.
- Success relies on navigating regulatory changes, patent expirations, and competitive pressures through reformulations and repositioning.
FAQs
1. How does patent expiration impact pricing for clotrimazole-betamethasone products? Patent expiration typically leads to generic entry, reducing prices by 20-40%, and increases market competition.
2. Can OTC availability influence market size? Yes. Increased OTC access drives volume but is limited by regulatory restrictions on corticosteroids in many jurisdictions.
3. What regions offer the most growth opportunities? Asia-Pacific and Latin America represent emerging markets with growing demand and less saturated competition.
4. Are there any fixed price controls on topical antifungal medications? Some regions, especially in Europe and Asia, regulate drug prices or have reimbursement systems influencing retail prices.
5. How do regulatory changes affect product development? Stricter regulations on corticosteroid potency and safety influence formulation strategies and approval pathways.
References
- MarketsandMarkets, "Antifungal Drugs Market," 2022.
- IQVIA, Prescription Data, 2022.
- European Medicines Agency, "Guidelines on Topical Corticosteroids," 2021.
- Statista, "Dermatological Drugs Market," 2021.
- GlobalData, "Topical Antifungal Market Analysis," 2022.
More… ↓
