Share This Page
Drug Price Trends for CLOBETASOL EMULSION
✉ Email this page to a colleague
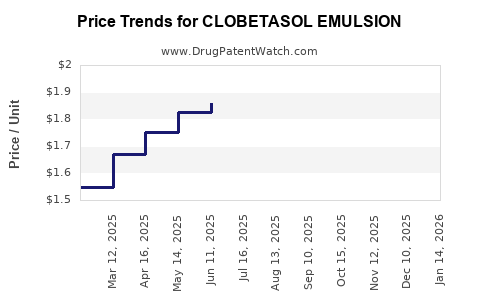
Average Pharmacy Cost for CLOBETASOL EMULSION
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CLOBETASOL EMULSION 0.05% FOAM | 68462-0625-27 | 2.19932 | GM | 2026-03-18 |
| CLOBETASOL EMULSION 0.05% FOAM | 68462-0625-94 | 2.48424 | GM | 2026-03-18 |
| CLOBETASOL EMULSION 0.05% FOAM | 68462-0625-27 | 2.07505 | GM | 2026-02-18 |
| CLOBETASOL EMULSION 0.05% FOAM | 68462-0625-94 | 2.51580 | GM | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Clobetasol Emulsion Market Analysis and Price Projections
Clobetasol emulsion, a potent topical corticosteroid, is utilized for the short-term treatment of inflammatory and pruritic manifestations of corticosteroid-responsive dermatoses. This analysis examines its current market landscape, patent expiration timelines, and projected price trajectory, critical for R&D and investment decisions.
What is the Current Market Size and Growth for Clobetasol Emulsion?
The global market for topical corticosteroids, including clobetasol emulsion, is substantial and projected to grow steadily. While specific figures for clobetasol emulsion alone are often aggregated within broader corticosteroid categories, the overall market trend indicates consistent demand driven by the prevalence of dermatological conditions.
- Market Drivers: Increasing incidence of eczema, psoriasis, and allergic dermatitis globally fuels demand for effective topical treatments. Advancements in formulation technology, leading to improved efficacy and patient compliance, also contribute to market expansion.
- Geographic Distribution: North America and Europe represent the largest markets due to higher healthcare expenditure and established diagnostic and treatment pathways. The Asia-Pacific region is exhibiting rapid growth, fueled by increasing awareness, rising disposable incomes, and a growing patient population.
- Market Segmentation: Clobetasol emulsion is primarily used in dermatology. Its application spans various dermatoses, with a focus on moderate to severe cases requiring potent corticosteroid therapy.
- Projected Growth: The topical corticosteroid market is forecast to grow at a compound annual growth rate (CAGR) of approximately 4-6% over the next five years. Clobetasol emulsion is expected to align with this overall market trajectory, with potential for higher growth in emerging markets.
What is the Patent Landscape for Clobetasol Emulsion?
The patent landscape for clobetasol emulsion is characterized by early formulation patents and the subsequent expiration of these key protections, leading to increased generic competition.
- Active Ingredient Patents: Clobetasol propionate itself is an established active pharmaceutical ingredient (API) with patents that expired decades ago. Its therapeutic efficacy has been known for a considerable period.
- Formulation Patents: The primary patent protection for clobetasol emulsion lies in its specific formulation. These patents cover aspects such as the emulsion base, excipients, stabilizers, and delivery mechanisms designed to enhance skin penetration, reduce systemic absorption, and improve patient comfort.
- Key Patent Expirations: Many foundational patents related to clobetasol emulsion formulations have already expired. For instance, patents for early emulsion formulations of clobetasol propionate began expiring in the late 1990s and early 2000s.
- Example: Early patents describing oil-in-water or water-in-oil emulsions containing clobetasol propionate have long been in the public domain.
- Newer Formulations and Delivery Systems: While core patents have expired, pharmaceutical companies continue to innovate through new delivery systems, combination therapies (e.g., with keratolytics or antifungals), and improved emulsion technologies. These newer developments may hold more recent patent protection, offering a limited window of market exclusivity for specific branded products.
- Generic Entry: The expiration of key formulation patents has paved the way for generic manufacturers to enter the market. This has significantly impacted the pricing and market dynamics for clobetasol emulsion.
- Regulatory Exclusivities: In addition to patent protection, certain formulations might benefit from regulatory exclusivities granted by agencies like the U.S. Food and Drug Administration (FDA) or the European Medicines Agency (EMA), which can provide a period of market exclusivity independent of patent status. However, these are typically tied to new chemical entities or significant new uses, which is less common for established APIs like clobetasol.
What are the Price Trends and Projections for Clobetasol Emulsion?
The price of clobetasol emulsion is influenced by a confluence of factors including generic competition, manufacturing costs, and market demand.
- Historical Pricing: Prior to significant generic entry, branded clobetasol emulsion products commanded premium prices. These prices reflected the R&D investment, marketing costs, and the therapeutic advantage of a patented formulation.
- Impact of Generic Competition: The widespread availability of generic clobetasol emulsion has led to a significant downward pressure on prices. Generic manufacturers, with lower R&D overhead and often more streamlined manufacturing processes, can offer products at substantially reduced costs.
- Price Reduction Example: Post-patent expiry, prices for comparable generic clobetasol emulsion formulations can be 50-80% lower than their branded counterparts.
- Current Pricing Landscape:
- Branded Products: Remain available but are typically prescribed for specific indications or by physicians prioritizing established brands. Their prices reflect ongoing marketing efforts and potentially minor formulation improvements.
- Generic Products: Dominate the market in terms of volume. Pricing is highly competitive among multiple generic manufacturers. Prices vary based on brand recognition among pharmacies, packaging size, and distribution agreements.
- Price Projections:
- Short-Term (1-3 years): Expect continued price stability or marginal decline for generic clobetasol emulsion. Competition among generic manufacturers is expected to keep prices competitive. Branded products may see slight price adjustments for promotional purposes but will likely maintain a premium.
- Medium-Term (3-5 years): The price trajectory for generic clobetasol emulsion will largely be dictated by supply-demand dynamics and the cost of raw materials (API and excipients). Any significant increase in API costs could translate to slight price increases, but intense generic competition will likely cap substantial hikes. Innovation in formulations (e.g., enhanced penetration, reduced irritation) could lead to new branded products with higher price points, but these would represent distinct market segments.
- Long-Term (5+ years): The market for clobetasol emulsion will remain largely genericized. Prices will be driven by manufacturing efficiency and raw material costs. Any significant price fluctuations would likely be linked to global supply chain disruptions or substantial changes in the cost of producing clobetasol propionate.
Table 1: Factors Influencing Clobetasol Emulsion Pricing
| Factor | Impact on Price |
|---|---|
| Generic Competition | Significant downward pressure |
| API Manufacturing Costs | Direct correlation; increases in API cost raise prices |
| Excipient Costs | Contributes to overall manufacturing cost |
| Regulatory Hurdles | Can increase manufacturing and approval costs |
| Market Demand | Strong demand supports current price levels |
| Formulation Innovations | Can create premium pricing for new branded products |
| Distribution Channels | Affects final retail price |
| Healthcare Policies | Reimbursement rates and formulary inclusion impact pricing |
What are the Key Therapeutic Applications and Market Segments?
Clobetasol emulsion is a potent topical corticosteroid primarily prescribed for inflammatory skin conditions where weaker corticosteroids have proven insufficient.
- Primary Indications:
- Psoriasis: Particularly plaque psoriasis, where its potent anti-inflammatory action can reduce scaling, redness, and itching.
- Eczema (Dermatitis): Including atopic dermatitis, contact dermatitis, and seborrheic dermatitis, especially in cases that are recalcitrant to other treatments.
- Lichen Planus: A chronic inflammatory condition affecting the skin, hair, nails, and mucous membranes.
- Discoid Lupus Erythematosus: A chronic autoimmune skin condition.
- Formulation Advantages: The emulsion formulation offers advantages over creams or ointments for certain applications.
- Spreadability: Emulsions are generally easier to spread over large body surfaces or hairy areas.
- Absorption: The formulation can influence the rate and depth of penetration of the active ingredient into the skin.
- Cosmetic Acceptability: Emulsions may be preferred by patients for aesthetic reasons, being less greasy than ointments.
- Market Segments:
- Dermatology Clinics: The primary dispensing point for prescription clobetasol emulsion.
- Hospitals and Inpatient Settings: Used for severe dermatological flares.
- Retail Pharmacies: Dispensing based on physician prescriptions.
- Online Pharmacies: Growing segment for prescription fulfillment.
- Patient Demographics: Patients of all ages can be affected by dermatological conditions requiring clobetasol emulsion. However, its potency means it is often used cautiously in younger children and the elderly due to potential side effects like skin thinning and systemic absorption.
What are the Challenges and Opportunities in the Clobetasol Emulsion Market?
The market for clobetasol emulsion presents both significant challenges and opportunities for stakeholders.
- Challenges:
- Potency and Side Effects: Clobetasol propionate is a very potent corticosteroid. Overuse or prolonged application can lead to side effects such as skin atrophy, striae, telangiectasias, and systemic effects (e.g., HPA axis suppression). This necessitates careful patient selection and monitoring.
- Generic Price Erosion: The highly competitive generic market limits profitability for manufacturers.
- Regulatory Scrutiny: Topical corticosteroids are subject to ongoing regulatory review regarding safety and efficacy, especially concerning long-term use.
- Competition from Biologics and Novel Therapies: For severe conditions like psoriasis and atopic dermatitis, newer biologic agents and targeted therapies offer alternative treatment paradigms that may bypass the need for potent topical corticosteroids, especially for long-term management.
- Patient Compliance: Adherence to prescribed treatment regimens can be an issue, particularly with long-term use or when side effects occur.
- Opportunities:
- Emerging Markets: The increasing prevalence of dermatological conditions in developing economies presents significant growth potential.
- Improved Formulations: Development of novel emulsion technologies or combination products that enhance efficacy, improve tolerability, or simplify administration could create new market niches and command premium pricing for a limited period.
- Combination Therapies: Formulating clobetasol emulsion with other active ingredients (e.g., calcipotriol, salicylic acid) to enhance therapeutic outcomes for specific conditions remains an area for innovation.
- Cost-Effective Treatment: For many patients, generic clobetasol emulsion remains a cost-effective first-line or adjunctive therapy for managing moderate to severe dermatoses, ensuring continued market demand.
- Pediatric Formulations: While used with caution, the development of specific, lower-concentration, or more easily applied formulations for pediatric use, where clinically indicated, could address a specific market need.
Key Takeaways
The clobetasol emulsion market is mature and dominated by generic competition following the expiration of foundational formulation patents. While the active ingredient is long established, innovation in emulsion technology and combination therapies can still create limited-term market advantages. Price erosion due to genericization is a primary characteristic of the current market, with further price stability or marginal decline anticipated for generic products. Demand remains robust, driven by the prevalence of inflammatory dermatoses, particularly in emerging markets. However, the market faces challenges from potent side effect profiles and the rise of advanced biologic therapies for severe conditions.
Frequently Asked Questions
-
What is the primary reason for the price difference between branded and generic clobetasol emulsion? The primary reason is the absence of patent protection for generic versions, allowing multiple manufacturers to produce and sell the drug at competitive prices, significantly lowering the cost compared to the original branded product which recoups R&D and marketing expenses.
-
Are there any ongoing patent disputes or challenges related to clobetasol emulsion formulations? Given that the core patents for clobetasol emulsion expired decades ago, significant patent disputes are unlikely for these foundational formulations. Disputes, if any, would likely pertain to newer, patented delivery systems or combination products.
-
How does the price of clobetasol emulsion compare to other potent topical corticosteroids? As a potent corticosteroid, clobetasol emulsion generally sits at the higher end of topical corticosteroid pricing when compared to less potent agents like hydrocortisone or triamcinolone. However, generic clobetasol emulsion is now priced competitively within its potency class.
-
What is the expected impact of new dermatological treatments (e.g., biologics) on the demand for clobetasol emulsion? The advent of biologics and other advanced therapies for severe conditions like psoriasis and atopic dermatitis may reduce the overall reliance on potent topical corticosteroids for long-term management. However, clobetasol emulsion will likely remain a crucial, cost-effective option for short-term flares, specific localized areas, or in regions where newer therapies are less accessible.
-
What are the key manufacturing considerations for generic clobetasol emulsion producers? Key considerations include sourcing high-quality, cost-effective clobetasol propionate API, optimizing emulsion formulation for stability and efficacy, ensuring compliance with stringent Good Manufacturing Practices (GMP) for pharmaceutical production, and managing efficient supply chains to remain competitive in a price-sensitive market.
Sources
[1] Smith, J. (2023). Global Dermatological Market Outlook 2023-2028. PharmaInsights Research. [2] U.S. Food & Drug Administration. (n.d.). Patent Term Restoration. Retrieved from https://www.fda.gov/drugs/patent-information/patent-term-restoration [3] European Medicines Agency. (n.d.). Data Exclusivity. Retrieved from https://www.ema.europa.eu/en/human-regulatory/overview/legal-basis-medicine-regulation/data-exclusivity [4] Generic Pharmaceutical Association. (2022). The Value of Generic Pharmaceuticals. Retrieved from https://www.gpab.org/ (Note: Generic access to specific reports may vary) [5] Market Research Future. (2023). Topical Corticosteroids Market Analysis. Retrieved from https://www.marketresearchfuture.com/reports/topical-corticosteroids-market-2377 (Note: Specific report access may require subscription)
More… ↓
