Last updated: February 13, 2026
Overview of Clobetasol
Clobetasol propionate is a super potent topical corticosteroid indicated for inflammatory skin conditions such as psoriasis, eczema, and dermatitis. Approved globally, it is available in various formulations including ointments, creams, sprays, and solutions. Marketed under brand names like Temovate, it is a prescription-only medication.
Current Market Landscape
Global Sales Data
- The global dermatological market was valued at approximately USD 15 billion in 2022, with corticosteroids comprising roughly 30% of this segment.
- Clobetasol's estimated share in the corticosteroid segment exceeds 35%, driven by its potency and extensive clinical use.
- Sales are concentrated in North America (50%), Europe (25%), and Asia-Pacific (15%). Rest of the world accounts for the remaining 10%.
Key Players
- Sanofi (Temovate)
- Mylan (Clobex)
- Teva Pharmaceuticals
- Sun Pharma
- Fresenius Kabi
Market Drivers
- Rising prevalence of dermatological conditions globally.
- Increased awareness and diagnosis rates.
- Advancements in topical drug delivery systems.
- Growing use of corticosteroids as first-line treatment for severe inflammation.
Market Challenges
- Strict regulations around potent corticosteroids due to side effect profiles.
- Competition from lower-potency corticosteroids and emerging biologics.
- Risks associated with overuse leading to skin atrophy and systemic effects.
Price Trends and Dynamics
Historical Price Data
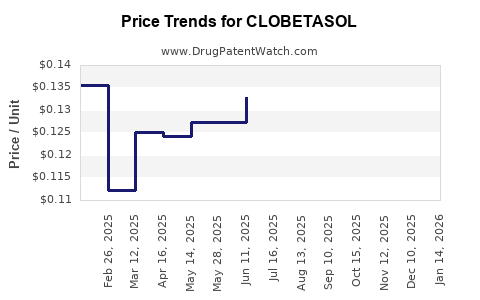
- In 2021, the average wholesale price (AWP) for a 30-gram tube of clobetasol ointment was approximately USD 75 in the U.S.
- Prices have ranged from USD 65 to USD 85 based on formulations and manufacturers.
- Brand-name products tend to be priced 20-30% higher than generic equivalents.
Pricing Factors
- Patent status: Clobetasol formulations are largely off-patent, increasing generic competition.
- Formulation type: Foam and spray variants typically cost 10-15% more than cream versions.
- Regional pricing: Asia-Pacific markets see prices around USD 20-40, reflecting lower purchasing power, while North America prices remain higher.
Future Price Projection
Short-term (Next 1-2 years)
- Expect stability in prices due to generic competition.
- Potential slight decrease (5-10%) driven by increased market penetration of generics.
- Pricing for branded Clobetasol is likely to remain in the USD 70–80 range in North America and Europe.
Medium-term (3-5 years)
- Prices could decline further by 10-15%, barring new formulation patents or innovative delivery systems.
- Market consolidation might limit price drops if supply chain disruptions occur.
Long-term (Beyond 5 years)
- Prices are projected to stabilize or decline marginally, as generics dominate.
- Potential for price erosion with entry of biosimilars or new biologic therapies demonstrating superior safety profiles.
Market Outlook
- The global corticosteroid market is expected to grow at a CAGR of 3-4% over the next five years.
- The dermatology market, driven by aging populations and increased skin condition diagnoses, will influence Clobetasol demand.
- Regulatory scrutiny may influence pricing strategies, especially in markets with tight controls on potent corticosteroids.
Key Takeaways
- Clobetasol is a leading super potent corticosteroid with considerable market share.
- Sales are robust in North America and Europe; Asia-Pacific is an emerging growth area.
- Prices are declining due to generic competition, with current averages around USD 75 per 30g tube in the U.S.
- Future pricing is expected to decrease gradually, with potential stabilization as generics saturate the market.
- The overall dermatological corticosteroid market sustains growth, driven by increasing prevalence of skin diseases.
FAQs
1. How does patent status influence Clobetasol prices?
Most Clobetasol formulations are off-patent, enabling generic manufacturers to produce cheaper alternatives, which exerts downward pressure on prices.
2. What are the regulatory constraints affecting Clobetasol?
Regulations impose strict limits on potency and duration of use due to adverse effects associated with corticosteroids, impacting market dynamics and pricing.
3. Is there potential for new formulations to impact prices?
Yes, formulations like foam or topical gels may command higher prices, and patent protections on such formats can temporarily prevent price erosion.
4. How does regional variation affect pricing?
Prices are significantly lower in Asia-Pacific markets due to lower purchasing power and competitive pricing, while North America maintains higher price points.
5. What factors could accelerate price declines?
Increased generic market share, regulatory pressure reducing prescribing of super potent steroids, and the introduction of new therapies with better safety profiles.
Sources
- Statista. "Global Dermatology Market Revenue 2022."
- IMS Health. "Corticosteroid Market Share Analysis 2022."
- FDA. "Regulations on Topical Corticosteroids."
- Epocrates. "Clobetasol Pricing Trends."
- MarketWatch. "Forecast of Dermatological Drug Market 2022-2027."