Drug Price Trends for CLINDACIN
✉ Email this page to a colleague
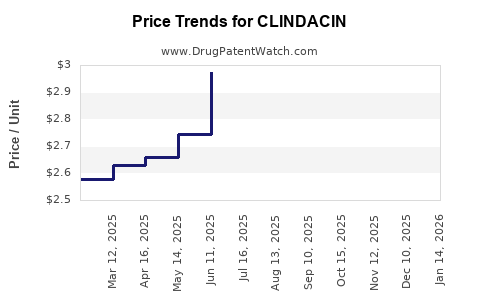
Average Pharmacy Cost for CLINDACIN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CLINDACIN 1% FOAM | 43538-0179-10 | 2.30626 | GM | 2026-05-20 |
| CLINDACIN 1% FOAM | 43538-0179-10 | 2.47430 | GM | 2026-04-22 |
| CLINDACIN 1% FOAM | 43538-0179-10 | 2.63462 | GM | 2026-03-18 |
| CLINDACIN 1% FOAM | 43538-0179-10 | 2.74859 | GM | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
