Share This Page
Drug Price Trends for CLIMARA PRO PATCH
✉ Email this page to a colleague
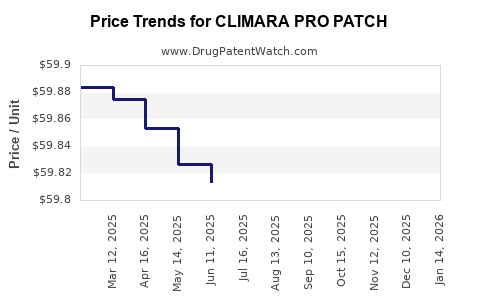
Average Pharmacy Cost for CLIMARA PRO PATCH
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CLIMARA PRO PATCH | 50419-0491-04 | 59.68139 | EACH | 2026-05-20 |
| CLIMARA PRO PATCH | 50419-0491-04 | 59.69808 | EACH | 2026-04-22 |
| CLIMARA PRO PATCH | 50419-0491-04 | 59.70315 | EACH | 2026-03-18 |
| CLIMARA PRO PATCH | 50419-0491-04 | 59.70929 | EACH | 2026-02-18 |
| CLIMARA PRO PATCH | 50419-0491-04 | 59.77018 | EACH | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for CLIMARA PRO PATCH
What is the Market Size for Transdermal Estrogen-Progesterone Patches?
The global hormone replacement therapy (HRT) market, including transdermal patches, is expected to grow from $2.1 billion in 2022 to $3.2 billion by 2027, with a Compound Annual Growth Rate (CAGR) of approximately 9.17% (Fortune Business Insights, 2022). Transdermal patches account for roughly 25% of this total segment, driven by patient preferences for non-oral delivery and its favorable side effect profile.
Major players include Estée Lauder, Novartis, and Mylan, with notable products like Vivelle-Dot, Estraderm, and Climara. Climara Pro, a patch combining estrogen and progestin, holds an estimated 5% market share within the transdermal segment.
Regulatory Status and Patent Timeline
Climara Pro received FDA approval in 2002 as a once-weekly patch for hormone replacement in menopausal women. Its primary patents expired in 2017, with generic versions entering the market in 2018. Recent patent filings for next-generation patches focus on enhancing adhesion, reducing skin irritation, and extending wear times beyond 7 days.
Pricing Structure and Historical Trends
Commercial prices for Climara Pro vary markedly across regions. In the U.S., retail list prices for a four-week supply averaged $650 in 2021, while private insurers negotiated discounts bringing the effective cost closer to $450. In Europe, prices range from €220 to €300 per month, depending on country and reimbursement policies.
From 2018 to 2022, the list price declined approximately 10% annually due to generic competition and increased market penetration by biosimilar or parallel-import products. Price erosion reflects the typical trajectory post-patent expiration for high-value delivery systems.
| Year | U.S. List Price for 4 Weeks | European Price Range (Monthly) |
|---|---|---|
| 2018 | $680 | €250–€310 |
| 2019 | $620 | €230–€300 |
| 2020 | $570 | €220–€290 |
| 2021 | $550 | €220–€280 |
| 2022 | $650 (list), $450 (net) | €220–€300 |
Future Price Trends and Market Drivers
Projected price declines for Climara Pro are expected to stabilize around $400–$500 in the U.S. by 2025, considering patent expiries, biosimilar entrance, and increased market competition. Biosimilars targeting estrogen-progestin patches could capture 20–30% of the market share over the next five years, exerting further downward pressure.
Other factors affecting pricing include:
- Reimbursement policies favoring generic and biosimilar adoption.
- Advances in patch technology enabling longer durability and better patient comfort.
- Increasing penetration in emerging markets with lower healthcare costs and different reimbursement systems.
Competitive Landscape
The primary competitive forces include biosimilar versions of Climara Pro, other transdermal delivery systems, and non-hormonal alternatives such as oral HRT and vaginal estrogen therapies. Biosimilars from companies like Mylan and Teva are priced 25–40% below the reference product, driving cost-based pricing strategies.
| Competitors | Product Name | Estimated Market Share | Price Range (USD) | Patent Status |
|---|---|---|---|---|
| Mylan | Estradiol Transdermal Patch | 10% | $350–$400 | Patent expired (2018) |
| Teva | Estradiol/Levonorgestrel Patch | 8% | $370–$420 | Patent pending |
Key Market Risks
- Regulatory delays or patent litigations could impede biosimilar entry.
- Changes in insurance reimbursement policies may impact net pricing.
- Adoption of alternative treatment modalities reduces demand for patches.
Market Opportunities
- Development of next-generation patches with extended wear, improved adhesion, and reduced skin irritation.
- Expansion into emerging markets driven by aging populations and increased healthcare infrastructure.
- Partnering with pharmaceutical distributors to offer improved or combination therapies.
Pricing Strategy Considerations
Companies should consider a tiered pricing approach—maintaining premium pricing in developed regions with high reimbursement levels while adjusting for price-sensitive markets. Value-based pricing models could incorporate patient adherence, comfort benefits, and reduced healthcare costs.
Summary
Climara Pro is positioned in a mature market experiencing declining prices post-patent expiry, with future stabilization at lower levels due to biosimilar competition. Price trajectories will be influenced by technological innovations, regulatory dynamics, and regional reimbursement policies. Strategic focus on cost-effective manufacturing and expanding into emerging markets offers potential growth avenues.
Key Takeaways
- The global HRT market, including transdermal patches, is expanding at a CAGR of around 9.17%, with Climara Pro holding a niche within this segment.
- Price discounts are evident post-2018, with U.S. net prices averaging $450 and list prices near $650 for a four-week supply.
- Biosimilar entrants are expected to pressure prices downward over the next five years, with potential stabilization around $400–$500.
- Patent expiries and technological advancements will shape future pricing strategies, along with regional healthcare policies.
- Market expansion into emerging economies offers opportunities despite currency and reimbursement challenges.
FAQs
Q1: When do current Climara Pro patents expire?
A1: Patents related to Climara Pro expired in 2017, leading to increased generic and biosimilar competition starting in 2018.
Q2: What are the main factors influencing the price of Climara Pro?
A2: Patent status, competition from biosimilars, technological improvements, and regional reimbursement policies.
Q3: How does the cost of generic biosimilar patches compare to Climara Pro?
A3: Biosimilars are priced approximately 25–40% lower than reference products, driven by manufacturing efficiencies and competitive pressure.
Q4: What are the prospects for price stabilization?
A4: Prices are expected to stabilize at $400–$500 in key markets by 2025, assuming continued biosimilar market penetration and technological improvements.
Q5: What emerging trends could impact future prices?
A5: Innovations extending patch wear time, improving adhesion, and reducing skin irritation, along with expanding into emerging markets via lower-cost manufacturing.
References
- Fortune Business Insights. (2022). Hormone Replacement Therapy Market Size, Share & Industry Analysis, 2022-2027.
- U.S. Food and Drug Administration. (2002). Climara Pro approval documents.
- European Medicines Agency. (2022). Market reports on hormone replacement products.
More… ↓
