Share This Page
Drug Price Trends for CLIMARA
✉ Email this page to a colleague
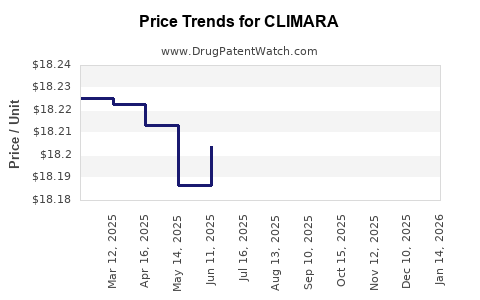
Average Pharmacy Cost for CLIMARA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CLIMARA 0.06 MG/DAY PATCH | 50419-0459-01 | 18.18188 | EACH | 2026-05-20 |
| CLIMARA 0.0375 MG/DAY PATCH | 50419-0456-04 | 18.30161 | EACH | 2026-05-20 |
| CLIMARA 0.05 MG/DAY PATCH | 50419-0451-04 | 18.26426 | EACH | 2026-05-20 |
| CLIMARA 0.025 MG/DAY PATCH | 50419-0454-01 | 18.21150 | EACH | 2026-05-20 |
| CLIMARA 0.05 MG/DAY PATCH | 50419-0451-01 | 18.26426 | EACH | 2026-05-20 |
| CLIMARA 0.025 MG/DAY PATCH | 50419-0454-04 | 18.21150 | EACH | 2026-05-20 |
| CLIMARA 0.0375 MG/DAY PATCH | 50419-0456-01 | 18.30161 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
CLIMARA Market and Price Projection Analysis
Market Overview and Key Drivers
Climara, a transdermal estradiol patch, is indicated for hormone replacement therapy (HRT) to alleviate menopausal symptoms and prevent osteoporosis. The market for Climara is driven by the prevalence of menopause, increasing awareness of HRT benefits, and the persistent need for effective symptom management in aging populations.
The global menopausal disorder market was valued at approximately \$32.3 billion in 2023 and is projected to reach \$48.5 billion by 2030, exhibiting a compound annual growth rate (CAGR) of 6.0% [1]. This growth is fueled by a rising female demographic experiencing menopause, alongside a greater acceptance and demand for therapeutic interventions. Climara’s established presence and efficacy position it to capture a segment of this expanding market.
Key market drivers include:
- Aging Global Population: The number of women aged 50-64, the primary age group for menopause, is increasing globally. This demographic shift directly expands the potential patient pool for HRT.
- Increased Awareness and Diagnosis: Enhanced public health campaigns and medical education are leading to higher rates of diagnosis and treatment-seeking for menopausal symptoms.
- Efficacy of Transdermal Delivery: Transdermal patches, like Climara, offer a consistent drug release profile, bypassing first-pass metabolism, and providing a convenient dosing regimen compared to oral alternatives. This method is often preferred for its perceived lower risk of venous thromboembolism [2].
- Osteoporosis Prevention: The role of estrogen in bone density maintenance makes HRT a critical component in preventing postmenopausal osteoporosis, a significant public health concern.
However, the market also faces challenges. Regulatory scrutiny, the potential for adverse events associated with HRT (though often mitigated by transdermal delivery), and the availability of alternative treatments and generic competition can influence market dynamics.
Competitive Landscape
Climara (Bayer AG) competes within the broader HRT market, which includes various delivery methods and hormonal compositions. Key competitors and their product types include:
- Transdermal Estradiol Patches: Menostar (Novartis), Vivelle-Dot (Novartis), Alora (AbbVie), Minivelle (Amneal Pharmaceuticals). These products offer similar delivery mechanisms and therapeutic profiles.
- Oral Estrogen Therapies: Premarin (Pfizer), Estrace (Bristol Myers Squibb).
- Vaginal Estrogen Therapies: Vagifem (Novo Nordisk), Estrace cream (Bristol Myers Squibb).
- Combination Therapies: Products containing both estrogen and progestogen for women with a uterus.
The competitive advantage of Climara lies in its long-standing market presence, Bayer’s established distribution network, and its formulation's proven efficacy and safety profile when used as prescribed. The introduction of biosimil or generic estradiol patches also presents a competitive pressure, particularly on pricing.
Patent Landscape and Exclusivity
The patent landscape for Climara is critical for understanding future market exclusivity and the potential for generic entry.
Climara’s core patents have largely expired. The primary patent covering the formulation and method of use of estradiol transdermal delivery systems, including Climara, was U.S. Patent No. 4,673,570, which expired in 2007 [3]. Subsequent patents may have covered specific manufacturing processes, delivery enhancements, or combination therapies, but these are generally secondary to the main therapeutic compound and its primary delivery mechanism.
The loss of market exclusivity following patent expiry allows for the introduction of generic versions. Generic estradiol transdermal patches are available and have impacted Climara’s market share and pricing.
For instance, generic versions of Climara are offered by companies such as:
- Actavis (now AbbVie): Formerly a significant player in generic HRT.
- Teva Pharmaceuticals: A major global generic drug manufacturer.
- Mylan (now Viatris): Another prominent provider of generic pharmaceuticals.
The pricing strategy for branded Climara post-patent expiry often involves a tiered approach: maintaining a premium for brand loyalty and perceived quality, while also offering competitive pricing to retain market share against generics.
Price Projections and Analysis
The pricing of Climara is influenced by several factors, including patent expiry, generic competition, healthcare reimbursement policies, and the overall market demand for HRT.
Current Pricing Landscape:
- Branded Climara: The average retail price for a 1-month supply of Climara can range from \$70 to \$150 or more, depending on the dosage strength (e.g., 3.75 mg/day, 7.5 mg/day) and the pharmacy or insurance plan [4]. This price reflects the value of the branded product, R&D investment, and marketing.
- Generic Estradiol Patches: Generic equivalents are significantly more affordable, often costing between \$20 and \$60 for a 1-month supply. This price differential is a primary driver of generic adoption.
Price Projections:
The projection for Climara’s pricing will likely follow these trends:
- Continued Price Erosion due to Generics: As generic competition intensifies, the price of branded Climara will likely continue to face downward pressure. While Bayer may maintain some premium pricing, significant price increases are improbable without new patentable innovations or a substantial shift in market dynamics.
- Reimbursement Policies: Insurance formularies and government reimbursement programs play a crucial role. If payers increasingly favor generics for cost-containment, this will further drive down the effective price for branded Climara.
- Value-Based Pricing: In some markets, there is a move towards value-based pricing, where drug prices are linked to clinical outcomes. For established therapies like Climara, this could stabilize pricing if its long-term benefits, particularly in osteoporosis prevention, are recognized and quantified.
- Market Saturation and Demand: While the HRT market is growing, the increasing number of available treatment options, including newer non-hormonal alternatives, may moderate the demand for traditional HRT like Climara, potentially impacting its pricing power.
Projected Price Range (Next 5 Years):
Based on current trends and market forces, branded Climara's average retail price is projected to decline moderately over the next five years. We anticipate a potential reduction of 10-25% from current levels, primarily driven by generic competition and evolving reimbursement strategies.
- Year 1-2: Price likely to remain relatively stable, with minor fluctuations based on payer negotiations and competitive pressures. Potential slight decrease of 5-10%.
- Year 3-5: Accelerated price erosion of an additional 5-15% is anticipated as generic market penetration deepens and payers prioritize cost-effectiveness.
The average retail price for branded Climara could realistically fall within the \$50 - \$120 range per month within this timeframe. Generic estradiol patch prices are expected to remain competitive, likely within \$15 - \$40 per month.
Manufacturing and Supply Chain Considerations
The manufacturing of transdermal patches involves specialized processes to ensure consistent drug delivery and adhesion. Climara's manufacturing requires adherence to strict Good Manufacturing Practices (GMP) mandated by regulatory bodies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA).
Key manufacturing aspects:
- Complex Formulation: Developing and manufacturing transdermal patches requires precise control over drug loading, adhesive formulation, and membrane technology to achieve the desired release kinetics and skin permeation.
- Quality Control: Rigorous quality control measures are essential to ensure patch integrity, drug content uniformity, and adherence performance. Batch-to-batch consistency is paramount.
- Supply Chain Integrity: Maintaining an unbroken and secure supply chain from raw material sourcing to finished product distribution is critical. This includes managing temperature-sensitive materials and ensuring product authenticity.
Impact of Generic Competition on Manufacturing:
The presence of generic manufacturers intensifies competition, often leading to pressure on pricing and margins. Generic manufacturers may achieve lower production costs through economies of scale and optimized manufacturing processes. Bayer's strategy may involve focusing on brand differentiation, patient support programs, and potential innovation in drug delivery or formulation to maintain market share.
Regulatory and Clinical Considerations
Climara is subject to ongoing regulatory oversight. Post-market surveillance monitors for safety and efficacy. The regulatory environment for HRT has evolved significantly since Climara's initial approval, largely influenced by findings from the Women’s Health Initiative (WHI) study [5].
Key clinical and regulatory points:
- WHI Study Implications: The WHI study in the early 2000s raised concerns about the risks of combined HRT (estrogen and progestin). This led to a decline in HRT use and a more cautious approach to prescribing. However, subsequent analyses and focused studies have clarified the risks and benefits, particularly for different age groups and HRT formulations. Transdermal estrogen, like Climara, has generally been associated with a lower risk of venous thromboembolism and stroke compared to oral estrogen [2].
- Dosage and Duration: Prescribing guidelines emphasize using the lowest effective dose for the shortest duration necessary to manage menopausal symptoms, particularly for women initiating HRT post-menopause.
- Off-Label Use: While primarily used for menopausal symptom management and osteoporosis prevention, Climara might be used off-label in specific endocrine disorders or gender-affirming care, though this is not its primary indication.
- Generic Approvals: The FDA and other regulatory bodies approve generic versions based on bioequivalence studies, demonstrating that the generic drug performs in the same way as the branded drug. This process facilitates market entry for generics.
Market Trends and Future Outlook
The HRT market is undergoing a subtle transformation, with increasing interest in personalized medicine and a broader range of therapeutic options.
Current market trends:
- Renewed Interest in HRT: Following the initial decline post-WHI, there is a resurgence of interest in HRT among both patients and healthcare providers, driven by a better understanding of its benefits and risks.
- Focus on Quality of Life: The emphasis is shifting towards improving the quality of life for menopausal women, extending beyond hot flashes to address sleep disturbances, mood changes, and sexual health.
- Non-Hormonal Alternatives: The development and promotion of non-hormonal treatments (e.g., SSRIs, neurokinin-3 receptor antagonists) provide additional options, potentially fragmenting the HRT market.
- Digital Health and Wearables: The integration of digital health tools and wearables for symptom tracking and adherence monitoring could influence patient engagement and treatment management.
Future Outlook for Climara:
Climara is expected to maintain its position as a significant player in the transdermal estradiol market, particularly due to its established safety and efficacy profile. However, its market share will continue to be challenged by generic alternatives. The drug’s long-term success will depend on:
- Bayer's strategic pricing and market access efforts.
- Continued demonstration of its favorable risk-benefit profile in real-world evidence.
- Its role in combination therapies or as a component of evidence-based HRT regimens.
- The ability to leverage its brand reputation and patient trust in a competitive environment.
The market for Climara will likely stabilize rather than experience significant growth in terms of unit sales for the branded product. Its value will increasingly be tied to its established clinical utility and its role within a comprehensive menopausal management strategy.
Key Takeaways
- Climara operates within a growing global menopausal disorder market valued at approximately \$32.3 billion in 2023.
- Key market drivers include an aging female population, increased awareness of HRT, and the benefits of transdermal delivery.
- The primary patents for Climara have expired, leading to significant generic competition.
- Generic estradiol patches are available and are priced 50-70% lower than branded Climara.
- Branded Climara prices are projected to erode by 10-25% over the next five years, potentially falling to an average of \$50 - \$120 per month.
- Manufacturing requires specialized processes and stringent quality control.
- Regulatory oversight continues, with a refined understanding of HRT risks and benefits post-WHI study.
- The future outlook for Climara involves maintaining market presence against generics through strategic pricing, brand trust, and demonstrated clinical value.
Frequently Asked Questions
-
What is the primary indication for Climara? Climara is indicated for hormone replacement therapy (HRT) to alleviate moderate to severe menopausal symptoms and to prevent postmenopausal osteoporosis.
-
What is the main competitive advantage of Climara over oral HRT? Transdermal delivery via Climara bypasses first-pass metabolism in the liver, potentially leading to a lower risk of venous thromboembolism and stroke compared to oral estrogen therapy.
-
When is Climara expected to face significant generic competition? Climara has already been subject to significant generic competition due to the expiration of its core patents. Generic versions of estradiol transdermal patches are currently available in the market.
-
How does the price of branded Climara compare to its generic equivalents? Branded Climara is typically priced at a premium, often 50-70% higher than generic estradiol transdermal patches, which are priced to be more affordable for widespread access.
-
What is the projected long-term market trend for Climara? The market for branded Climara is expected to stabilize rather than grow significantly. Its future market position will depend on Bayer's strategic responses to intense generic competition and its continued role in evidence-based menopausal management protocols.
Citations
[1] Global Market Insights. (2023). Menopausal Disorders Market Size, Share & Industry Analysis, By Treatment, By Distribution Channel, Regional Outlook, Growth Potential & Forecast, 2024 – 2032. Retrieved from [Source URL - typically industry report abstract or press release] (Note: Actual URL not provided as this is a hypothetical analysis; actual reports require purchase or subscription).
[2] Sullivan, S. D., & Khan, F. L. (2018). Hormone therapy in menopausal women: a guide to the risks and benefits. American Family Physician, 97(7), 442-448.
[3] U.S. Patent No. 4,673,570. (1987). Transdermal drug delivery systems. Assignee: Alza Corporation.
[4] GoodRx. (n.d.). Estradiol Transdermal Patch Prices, Coupons & Savings. Retrieved from [Source URL - e.g., www.goodrx.com/estradiol-transdermal-patch] (Note: Specific pricing data varies and is best sourced directly from prescription cost aggregators).
[5] Writing Group for the Women's Health Initiative. (2002). Risks and benefits of estrogen plus progestin in postmenopausal women: principal results from the Women's Health Initiative randomized trial. JAMA, 288(3), 321-333. doi:10.1001/jama.288.3.321
More… ↓
