Share This Page
Drug Price Trends for CLENPIQ
✉ Email this page to a colleague
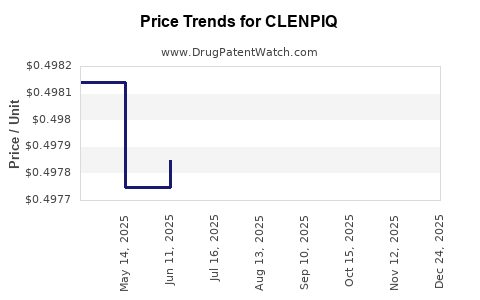
Average Pharmacy Cost for CLENPIQ
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CLENPIQ 175 ML SOLUTION | 55566-6800-01 | 0.51845 | ML | 2025-12-19 |
| CLENPIQ 175 ML SOLUTION | 55566-6800-01 | 0.49850 | ML | 2025-11-19 |
| CLENPIQ 175 ML SOLUTION | 55566-6800-01 | 0.49860 | ML | 2025-10-22 |
| CLENPIQ 175 ML SOLUTION | 55566-6800-01 | 0.49828 | ML | 2025-09-17 |
| CLENPIQ 175 ML SOLUTION | 55566-6800-01 | 0.49828 | ML | 2025-08-20 |
| CLENPIQ 175 ML SOLUTION | 55566-6800-01 | 0.49816 | ML | 2025-07-23 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
CLENPIQ Market Analysis and Financial Projection
What is CLENPIQ and what is its current market position?
CLENPIQ is an osmotic laxative developed for bowel cleansing prior to colonoscopy. It contains sodium picosulfate, magnesium oxide, and citric acid. Approved by the FDA in October 2015, it competes directly with drugs such as MiraLAX, Golytely, and Prepopik. It targets the gastrointestinal (GI) cleaning market, which is estimated at approximately $1.2 billion globally.
Clinicians and pharmacies primarily market CLENPIQ in the United States. The drug gained market traction due to its lower volume, improved tolerability, and effective cleansing profile compared to older solutions. It is marketed as a convenient alternative with fewer side effects like nausea or bloating associated with traditional bowel prep agents.
How does CLENPIQ’s market share compare with competitors?
In 2022, CLENPIQ held an estimated 15-20% of the US bowel prep market. Its main competitors, MiraLAX and Golytely, dominate with combined share exceeding 50%. Prepopik holds approximately 10%. The market share distribution reflects:
- CLENPIQ: 15-20%
- MiraLAX: approx. 35-40%
- Golytely: approx. 15-20%
- Prepopik: 10%
- Other brands and generics: 10-20%
Market penetration persists due to physician preference, product familiarity, and pricing strategies.
What factors influence CLENPIQ’s pricing and revenue?
Pricing negotiations, insurance coverage, and reimbursement policies dictate the price point of CLENPIQ. Current average wholesale price (AWP) for a single kit ranges from $85 to $120 depending on purchase volume, location, and distributor agreements. Cash prices for patients generally fall into the $100–$150 range.
Reimbursement rates for hospitals and outpatient clinics influence revenue streams. Medicare and most private insurers reimburse based on negotiated rates, which tend to be slightly below the AWP. Price pressure arises from generic competition and off-label use limits.
What are short-term and long-term price projections?
Short-term (next 1-2 years)
- Expect moderate price stability with slight downward pressure due to tightened reimbursement policies and increased hospital procurement negotiations.
- Wholesale prices are projected to decline by approximately 5–10% in 2023–2024.
- The competitive landscape remains static unless a new entrant or a reformulation significantly alters market share.
Long-term (3-5 years)
- Price decline could reach 15-20%, driven by increased genericization and insurer bargaining power.
- Introduction of biosimilar or alternative bowel prep solutions might further compress prices.
- Upward price adjustments natively are limited due to the mature status of the drug.
How does market growth outlook look for CLENPIQ?
Market growth for bowel prep drugs is driven by:
- Increasing colonoscopy rates driven by colorectal cancer screening guidelines.
- Aging demographic, leading to more GI procedures.
- Technological improvements in bowel cleansing efficacy.
Projected compound annual growth rate (CAGR) for the global bowel cleansing market is approximately 4-6% through 2028[1]. For CLENPIQ, growth will depend heavily on market share expansion and payer acceptance. Share gains hinge on physician preference shifts, patient adherence benefits, and formulary inclusion.
What regulatory and policy factors could impact pricing and market share?
- Reimbursement reforms: Changes under Medicare and private insurers can influence profitability.
- FDA policies: New safety warnings or formulation restrictions may impact costs and demand.
- Market access initiatives: Drug formulary updates may favor or limit access to CLENPIQ.
- Off-label use restrictions: Limiting off-label applications could constrain revenue growth.
How do price projections compare with competitors?
Competitor drugs display similar pricing trajectories. Golytely remains high-cost, averaging $150–$200 per kit, while MiraLAX’s over-the-counter popularity keeps its prescription price lower, at around $10–$30 per dose. Generic options further pressure branded products.
Projected declines for CLENPIQ mirror those of Golytely and Prepopik, roughly 10-20% over five years, reflecting standardization in pricing and increased generic competition.
What strategic considerations should stakeholders prioritize?
- Enhance formulary inclusion through clinical efficacy data.
- Emphasize patient convenience and tolerability to drive demand.
- Monitor reimbursement trends and negotiate favorable agreements.
- Prepare for potential biosimilar or alternative bowel prep competition.
Key Takeaways
- CLENPIQ commands a niche within the $1.2 billion bowel cleansing market, with about 15-20% share.
- Prices for CLENPIQ are expected to decline by 5-20% over the next five years due to genericization and reimbursement pressures.
- Revenue growth hinges on expanding market share via physician preference, improved access, and policy adjustments.
- The global bowel cleansing market is growing at 4-6%, driven by aging populations and increased colonoscopy rates.
- Competitive pressure from branded and generic products pushes the need for continuous differentiation.
FAQs
1. How sustainable is CLENPIQ’s current market position?
Its position remains stable short-term but faces long-term decline pressures from generics and formulary shifts.
2. Will new formulations of bowel prep drugs impact CLENPIQ?
Potentially, if reformulations improve efficacy or tolerability, they could alter market dynamics and pricing.
3. How does insurance coverage affect CLENPIQ pricing?
Reimbursement rates influence net revenue; tighter coverage can pressure prices downward.
4. Are biosimilars or other innovations likely to replace CLENPIQ?
Biosimilars are less relevant; traditional bowel preps are small-molecule compounds. Innovations might introduce new formulations or delivery methods.
5. What is the impact of aging demographics on this market?
An aging population increases colonoscopy demand, supporting market growth despite pricing pressures.
References
- Markets and Markets. "Bowel Cleansing Market by Product, Application, and Region," 2022.
More… ↓
