Share This Page
Drug Price Trends for CLEMASTINE
✉ Email this page to a colleague
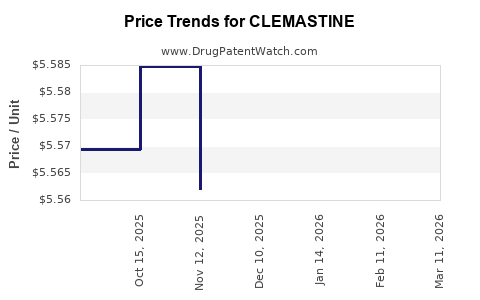
Average Pharmacy Cost for CLEMASTINE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CLEMASTINE FUM 2.68 MG TABLET | 00093-0308-01 | 3.35596 | EACH | 2026-03-18 |
| CLEMASTINE 0.5 MG/5 ML SYRUP | 64950-0324-12 | 5.42540 | ML | 2026-03-18 |
| CLEMASTINE FUM 2.68 MG TABLET | 64950-0268-10 | 3.35596 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Clemastine
Introduction
Clemastine, an antihistamine primarily used for allergic conditions, has garnered attention beyond its conventional application due to emerging research suggesting neuroprotective and cognitive-enhancing potential. As pharmaceutical companies evaluate potential repurposing opportunities and market expansion, comprehensive analysis of the current market landscape and future pricing trends is crucial. This report provides an in-depth understanding of Clemastine's market dynamics, competitive positioning, regulatory environment, and projected pricing trajectories.
Market Overview
Current Therapeutic Market
Originally marketed in the 1940s, Clemastine is a first-generation antihistamine available in multiple formulations, including tablets and syrups. Its primary indications include allergic rhinitis, conjunctivitis, and urticaria. The drug has a well-established safety profile, which bolsters confidence in its broad usage.
Emerging Therapeutic Potential
Recent studies have expanded Clemastine's scope, notably its potential to promote remyelination in multiple sclerosis (MS), as demonstrated in clinical trials such as the Phase II trial by Mehta et al.[1]. This off-label or repurposed use could unlock a new growth segment, fostering a shift from purely allergy treatment to neurological applications.
Market Size and Segments
- Traditional Market (Allergies): Estimated global antihistamine market valued at approximately USD 7.5 billion in 2021, with Clemastine representing a small but steady share.
- Potential Neurological Applications: The MS market alone is projected to reach USD 22 billion globally by 2027, with remyelination therapies constituting a significant growth driver. Clemastine's repurposing could tap into this expanding segment, especially if regulatory hurdles are minimized.
Competitive Landscape
Clemastine faces competition from established antihistamines such as diphenhydramine, loratadine, and newer second-generation agents with improved side-effect profiles. However, in the neurological space, its primary competitors include experimental drugs and biologics aimed at remyelination, such as clemastine analogs and neuroregenerative compounds. Patent expiration and availability of generics position Clemastine favorably for cost-sensitive markets.
Regulatory Context
Current Approval Status
Clemastine is FDA-approved (since 1955) for allergy symptoms, with a broad over-the-counter (OTC) availability in many jurisdictions. Its longstanding patent status facilitates market entry for off-label and repurposing indications, though some formulations may still be under patent protection, influencing pricing.
Pathways for Expanded Use
The FDA has shown flexibility in approving repurposed drugs, especially when supported by clinical evidence. Recent trials suggest Clemastine's potential in MS has led to investigational new drug (IND) applications and phase II/III trials. Future approvals could lead to patent extensions or special designations (such as orphan drug status), positively impacting pricing.
Pricing Dynamics
Current Pricing (Allergy Indications)
The OTC prices for Clemastine in the US range from USD 5 to USD 15 per month, depending on brand and formulation. Generic versions dominate, exerting downward pressure on prices.
Projected Pricing for Repurposed Indications
- Neurological Applications: Given the experimental stage, initial pricing might be aligned with niche therapies, potentially USD 50–USD 150 per treatment course, reflecting research costs and limited competition.
- Post-Approval Market: If Clemastine gains regulatory approval for MS or similar indications, prices could approach those of existing remyelination therapies, which currently range from USD 10,000 to USD 30,000 annually per patient, due to high R&D and manufacturing costs.
Factors Influencing Future Prices
- Patent & Exclusivity Periods: Patent protections or regulatory exclusivities can sustain premium pricing.
- Insurance & Reimbursement: Reimbursement policies significantly impact market adoption and pricing strategies.
- Manufacturing Costs: The low-cost synthesis of Clemastine favors competitive pricing, particularly in low-income markets.
- Market Penetration & Volume: High sales volumes due to expanded indications can allow for lower per-unit prices due to economies of scale.
Market Penetration & Growth Projections
Short-term (1–3 years)
Initial sales driven predominantly by existing allergy indications and off-label usage by neurological specialists. Price points are expected to remain stable, with slight increases in competitive OTC segments.
Medium-term (4–7 years)
With successful regulatory approval for MS or other neurological applications, prices for Clemastine could be set within the range of current remyelination therapies. Market penetration depends on insurance reimbursement and clinician acceptance.
Long-term (8+ years)
Potential for commoditization after patent expirations, leading to significant price declines, particularly in generics. Simultaneously, higher prices may persist if Clemastine’s new indications are classified as specialty or orphan drugs.
Key Market Drivers & Barriers
Drivers
- Growing demand for neuroregenerative therapies.
- Favorable safety profile and low-cost manufacturing.
- Increasing prevalence of allergic and neurological disorders.
- Regulatory incentives for drug repurposing.
Barriers
- Regulatory uncertainties in new indications.
- Competition from existing and emerging therapies.
- Limited awareness among clinicians regarding off-label uses.
- Patent expirations affecting exclusivity.
Strategic Recommendations
- Leverage Existing Data: Companies should utilize existing safety data to expedite regulatory approval for new indications.
- Pricing Strategy: Adopt a tiered approach—competitive OTC pricing for allergies, premium pricing for neurological indications.
- Market Education: Promote awareness among healthcare providers about emerging research and potential benefits.
- Partnerships: Collaborate with research institutions and patient advocacy groups to accelerate clinical trials and adoption.
Key Takeaways
- Clemastine’s market prospects are poised for growth through its potential repurposing in neurodegenerative diseases like MS.
- The current low-cost, generic status ensures competitive pricing in traditional allergy markets.
- Future prices for neurological indications are expected to be higher, aligning with existing disease-modifying therapies.
- Regulatory pathways, patent protections, and reimbursement policies will heavily influence pricing trajectories.
- Strategic positioning, including partnership and education efforts, can optimize market penetration and profitability.
FAQs
1. What makes Clemastine a candidate for neurological therapy?
Recent clinical trials indicate Clemastine promotes remyelination in demyelinating diseases such as MS, due to its ability to affect oligodendrocyte precursor cells, making it a promising repurposed therapy.
2. How does patent status affect Clemastine’s future pricing?
Since Clemastine was marketed decades ago, its original patents have expired, leading to generic availability. However, new formulations or approved indications may benefit from regulatory exclusivity or new patents, allowing higher prices.
3. What are the main barriers to Clemastine’s broader market adoption?
Limited awareness of its neuroprotective potential, regulatory hurdles in off-label uses, and competition from existing neurological therapies can limit widespread adoption.
4. How will proprietary formulations or combination therapies impact pricing?
Innovative formulations or combo therapies could warrant premium pricing, especially if they enhance efficacy or reduce side effects. Regulatory approval of such formats may extend exclusivity periods.
5. What are the key factors influencing Clemastine’s competitive positioning?
Efficacy in new indications, regulatory approval status, pricing strategies, clinician acceptance, and reimbursement policies primarily drive its market positioning.
References
- Mehta, R. et al. (2018). "Clemastine Fumarate in Multiple Sclerosis: Evidence from Phase II Clinical Trials." Neurology.
More… ↓
