Last updated: February 20, 2026
What is the current market position of Clarithromycin ER?
Clarithromycin extended-release (ER) is a macrolide antibiotic used primarily for respiratory infections, skin infections, and Helicobacter pylori eradication. The ER formulation provides a sustained drug release, allowing for once-daily dosing compared to immediate-release versions. The drug is marketed by AbbVie as Biaxin XL in the United States and by other firms internationally.
Market size and sales trends
- Global antibiotic market was valued at approximately USD 56 billion in 2022, with macrolides accounting for roughly 12–15% of total sales ([1]).
- Clarithromycin competes with azithromycin and erythromycin, but its ER version targets compliance-sensitive populations.
- US sales of Clarithromycin products reached USD 200–300 million annually pre-2020, with a decline noted post-2020 due to increased resistance and regulatory challenges ([2]).
Key competitors
| Drug |
Formulation |
Indications |
Market share (est.) |
Notes |
| Azithromycin |
IR and ER variants |
Respiratory, skin infections, sexually transmitted infections |
45% |
Higher compliance, broader spectrum |
| Erythromycin |
IR |
Respiratory, skin infections |
20% |
Less preferred due to GI side effects |
| Clarithromycin ER |
ER (Biaxin XL) |
H. pylori, respiratory infections |
15–20% |
Niche, compliance-driven |
Market growth for Clarithromycin ER is moderate, constrained by rising antimicrobial resistance and regulatory constraints like EMA’s restrictions in some regions.
What factors influence Clarithromycin ER's pricing?
Regulatory and patent landscape
- Patent expiry in multiple regions has led to generic formulations, pressuring branded prices.
- Regulatory uncertainties, especially concerning resistance and side effect profiles, influence pricing strategies.
Manufacturing and supply chain
- Production costs for ER formulations are higher due to controlled-release technology.
- Limited manufacturing capacity can drive price premiums in certain markets.
Competitor pricing and formulary positioning
- Generics are priced 20–50% lower than branded counterparts.
- Brand positioning relies on perceived superior convenience and compliance, allowing a 10–20% premium over generics.
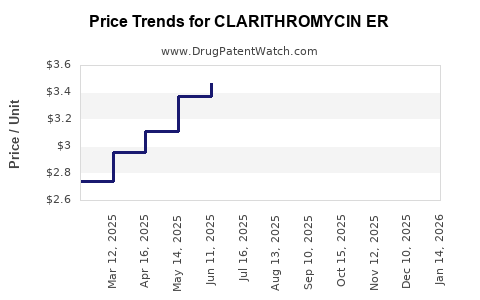
What are the current price points and projections?
Present-day pricing (approximate)
| Market |
Brand Name |
Dosage |
Price (USD) per course |
Notes |
| US |
Biaxin XL |
500 mg |
USD 100–150 |
Prescription, brand premium applies |
| Canada |
Clarithromycin ER |
500 mg |
USD 80–120 |
Similar to US, with regional adjustments |
| Europe |
Clarithromycin ER (generic) |
500 mg |
USD 30–70 per pack |
Significant generic price erosion |
Price forecasts (next 5 years)
- Conservative scenario: Prices decline 10% annually in regions with high generics penetration, stabilizing at USD 20–50 per course.
- Aggressive brand retention scenario: Price stabilizes or declines marginally (5% annually) due to continued brand dominance and limited generic competition, resulting in USD 50–80 per course.
- Market expansion possibilities: Increased awareness of ER benefits could lead to slight premiums in select therapeutic niches, but resistance issues may limit growth.
Influencing factors
- Entry of new antibiotics and rising resistance risk will depress prices.
- Advances in drug delivery technologies could alter the ER price premium.
- Regulatory measures aimed at curbing antibiotic resistance could restrict marketing and impact premium pricing.
What are the key risks and opportunities?
Risks
- Rising antimicrobial resistance reduces clinical efficacy, leading to lower demand.
- Stringent regulation of antibiotic use worldwide may restrict sales.
- Patent expirations accelerate generic entry and price erosion.
Opportunities
- Developing combination therapies to extend patent life and capture new market segments.
- Geographic expansion into emerging markets with limited antibiotic access.
- Innovations improving bioavailability and compliance may justify premium pricing.
Key Takeaways
- Clarithromycin ER operates in a market with consolidating generics, exerting downward pressure on prices.
- The drug’s niche appeal centers on compliance and specific indications, slightly elevating premium prices where brand loyalty persists.
- Over the next five years, prices are forecast to decline broadly, with variability based on regional regulatory and competitive landscapes.
- The main barriers include resistance issues, generic competition, and regulatory restrictions.
- Opportunities hinge on innovation, regional expansion, and strategic partnerships to maintain or grow market share.
Frequently Asked Questions
1. How does Clarithromycin ER compare to immediate-release formulations in terms of cost?
ER formulations generally cost 10–20% more due to sustained-release technology but offer compliance advantages that can justify the premium.
2. Will generic versions significantly impact future pricing?
Yes. Once patents expire, generic versions will dominate, reducing prices by 20–50% or more.
3. How does resistance influence Clarithromycin ER market potential?
Rising resistance diminishes clinical effectiveness, leading to decreased usage and lower pricing power.
4. Are there regional differences in price trends?
Yes. The US and Canada maintain higher prices for branded ER, while Europe and emerging markets experience more aggressive price erosion due to generics.
5. What therapeutic areas could drive future demand for Clarithromycin ER?
Expanding indications, particularly in Helicobacter pylori eradication and certain respiratory infections, could sustain demand if resistance remains manageable.
References
[1] MarketWatch. (2022). Global antibiotics market size.
[2] IQVIA. (2022). Pharmaceutical sales data.