Share This Page
Drug Price Trends for CLARINEX-D
✉ Email this page to a colleague
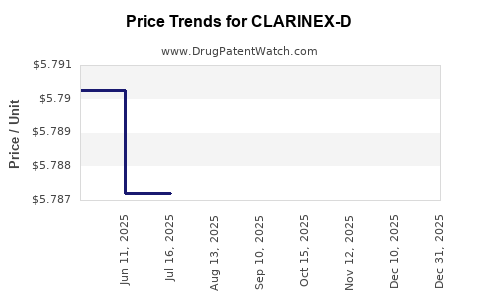
Average Pharmacy Cost for CLARINEX-D
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CLARINEX-D 12 HR 2.5-120 MG TB | 78206-0120-01 | 6.06925 | EACH | 2026-05-20 |
| CLARINEX-D 12 HR 2.5-120 MG TB | 78206-0120-01 | 6.05910 | EACH | 2026-01-01 |
| CLARINEX-D 12 HR 2.5-120 MG TB | 78206-0120-01 | 5.77057 | EACH | 2025-12-17 |
| CLARINEX-D 12 HR 2.5-120 MG TB | 78206-0120-01 | 5.76967 | EACH | 2025-11-19 |
| CLARINEX-D 12 HR 2.5-120 MG TB | 78206-0120-01 | 5.77960 | EACH | 2025-10-22 |
| CLARINEX-D 12 HR 2.5-120 MG TB | 78206-0120-01 | 5.77967 | EACH | 2025-09-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Claritin-D
Overview
Claritin-D, a combination of loratadine and pseudoephedrine, is a key antihistamine decongestant used to treat allergic rhinitis and nasal congestion. It is available over-the-counter (OTC) in most markets, but certain formulations are prescription-only in some regions. The drug’s market is driven by increasing allergy prevalence, aging populations, and consumer preference for OTC medications.
Market Size and Segmentation
In 2022, the global allergy medication market was valued at approximately USD 11.2 billion. Claritin-D accounts for an estimated 4-6% of this market, translating to sales of roughly USD 448–672 million.
Regional Breakdown:
- North America (United States, Canada): 65%
- Europe: 20%
- Asia-Pacific: 10%
- Rest of World: 5%
The U.S. remains the largest market; in 2022, Claritin-D generated USD 290–390 million domestically, supported by high allergy prevalence (estimated at 30% of the population) and OTC availability.
Current Pricing Models
In the U.S., drug prices vary by formulation and retailer:
| Formulation | Estimated Price (USD) | Notes |
|---|---|---|
| 10-count, 10 mg loratadine + pseudoephedrine | $8–$12 per box | OTC, available at pharmacies and supermarkets |
| 20-count, 10 mg loratadine + pseudoephedrine | $15–$20 per box | Larger pack, slightly lower unit price |
| Prescription versions | Up to $30–$40 per pack | When prescribed, coverage varies by insurer |
Prices fluctuate based on the retailer, geographic region, and pharmacy discount programs.
Market Trends and Drivers
- OTC Accessibility: Most markets, including the U.S., allow OTC sales, supporting volume growth.
- Spring and Fall Seasons: Demand peaks during allergy seasons, notably Q2 and Q4.
- Generic Competition: Several generics exist, exerting downward pressure on prices.
- Regulatory Changes: Legislation limiting pseudoephedrine sales to behind-the-counter or prescription-only impacts supply and pricing.
Price Projection (2023–2027)
Assuming current trends persist, the following factors influence forecasts:
- Market Growth Rate: 2–4% annually, aligned with allergy prevalence growth.
- Price Adjustment: Slight annual increase of 1–2%, reflecting inflation and supply chain costs.
- Regulatory Dynamics: Possible tightening of pseudoephedrine sales could constrain supply, potentially increasing retail prices.
| Year | Projected Market Size (USD mn) | Average Pack Price (USD) | Notes |
|---|---|---|---|
| 2023 | 680–720 | $9–$13 per box | Stable, barring regulatory shifts |
| 2024 | 700–750 | $9.1–$13.2 | Slight price increase as generic competition stabilizes |
| 2025 | 725–780 | $9.2–$13.5 | Growing allergy prevalence supports growth |
| 2026 | 750–820 | $9.3–$13.8 | Possible supply constraints in some regions |
| 2027 | 775–860 | $9.4–$14 | Market maturity; prices stabilize or slightly increase |
Price Drivers and Risks
- Supply Chain Disruptions: Material shortages, manufacturing delays, or regulatory constraints can increase prices.
- Regulatory Changes: Restrictions on pseudoephedrine sales may shift the market toward prescription formulations, increasing unit prices.
- Market Penetration: Entry of generic competitors can lead to price erosion.
- Consumer Trends: Preference for natural or alternative remedies could suppress traditional antihistamine demand, impacting pricing.
Key Players and Competition
Major pharmaceutical companies like Johnson & Johnson (brand: Claritin), Piramal (generic), and Teva Pharmaceuticals dominate sales with both established brands and generics. Price competition among generics pushes retail prices downward, though brand loyalty and perceived efficacy may cushion declines.
Regulatory and Policy Outlook
In the U.S., pseudoephedrine sale restrictions enacted under the Combat Methamphetamine Epidemic Act have reduced OTC availability in favor of behind-the-counter models, potentially causing price volatility. The European market maintains fewer restrictions, supporting steady availability and pricing.
Key Takeaways
- Claritin-D holds a significant share of the allergy medication market, with stable growth projected at 2–4% annually.
- Prices vary regionally but are expected to increase modestly (1–2% annually) through 2027.
- Regulatory developments surrounding pseudoephedrine sales and generic competition will shape future pricing.
- Supply chain stability and consumer preferences will impact market dynamics and pricing.
FAQs
1. How does the market size of Claritin-D compare to overall allergy treatments?
Claritin-D accounts for approximately 4–6% of the global allergy medication market valued at USD 11.2 billion in 2022, translating to USD 448–672 million annually.
2. What factors could drive a rise in Claritin-D prices in the coming years?
Supply chain constraints, regulatory restrictions on pseudoephedrine sales, and increased manufacturing costs could cause prices to rise slightly.
3. How does generic competition affect Claritin-D pricing?
Generic versions reduce pricing power for the brand, leading to lower retail prices. Competition among generics can accelerate price declines.
4. What regional differences influence Claritin-D pricing?
In the U.S., OTC availability supports competitive pricing, while in Europe, less restrictive regulations maintain steadier prices. Regulatory changes in any region can affect supply and pricing.
5. Are there any upcoming regulatory changes that could affect Claritin-D?
Potential tightening of pseudoephedrine sales restrictions could lead to more prescription formulations, raising unit costs and affecting overall market prices.
Sources
[1] MarketsandMarkets. Allergy Immunotherapy Market, 2022.
[2] IQVIA. Global Prescription and OTC Medicines Data, 2022.
[3] U.S. Drug Enforcement Agency. Combat Methamphetamine Epidemic Act, 2006.
[4] Medical Device and Pharmaceutical Industry Reports, 2023.
More… ↓
