Share This Page
Drug Price Trends for CIPROFLOX-DEXAMETH OTIC SUSP
✉ Email this page to a colleague
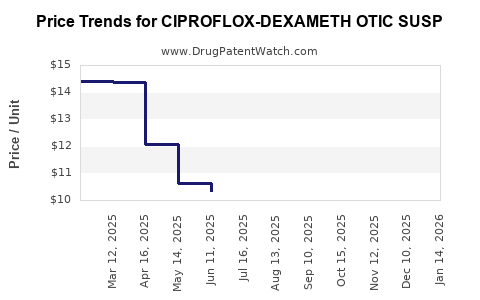
Average Pharmacy Cost for CIPROFLOX-DEXAMETH OTIC SUSP
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CIPROFLOX-DEXAMETH OTIC SUSP | 43598-0326-75 | 7.77983 | ML | 2026-04-22 |
| CIPROFLOX-DEXAMETH OTIC SUSP | 16714-0628-01 | 7.77983 | ML | 2026-04-22 |
| CIPROFLOX-DEXAMETH OTIC SUSP | 72485-0625-13 | 7.77983 | ML | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
CIPROFLOX-DEXAMETH OTIC SUSP: Market Trajectory and Price Forecast
What is the Current Market Landscape for CIPROFLOX-DEXAMETH OTIC SUSP?
CIPROFLOX-DEXAMETH OTIC SUSP, a combination ophthalmic suspension containing ciprofloxacin hydrochloride and a corticosteroid, targets bacterial conjunctivitis and otitis externa. The current market is characterized by a mature generic presence. Key market drivers include the prevalence of ear and eye infections, often linked to hygiene, seasonal allergies, and swimming activities. The pediatric population represents a significant consumer segment for these formulations.
The competitive landscape is fragmented, with multiple manufacturers offering bioequivalent generic versions. Pricing is heavily influenced by generic competition and the cost-containment pressures within healthcare systems. The established efficacy and safety profile of the active pharmaceutical ingredients (APIs) contribute to sustained demand.
What is the Projected Market Growth for CIPROFLOX-DEXAMETH OTIC SUSP?
The market for CIPROFLOX-DEXAMETH OTIC SUSP is projected to experience modest growth over the next five years. This growth will be primarily driven by:
- Increasing incidence of bacterial infections: Factors such as antibiotic resistance trends in other therapeutic areas may indirectly influence the preference for established treatment protocols for bacterial conjunctivitis and otitis.
- Demographic shifts: An aging global population may see a rise in opportunistic infections. Conversely, the robust pediatric population continues to be a stable demand generator.
- Emerging markets: Expanding access to healthcare and diagnostics in developing regions can lead to increased prescription volumes.
However, growth will be tempered by:
- Intense generic competition: The presence of numerous generic manufacturers caps price increases and limits revenue expansion.
- Development of novel therapies: While not a primary driver for this specific combination, the long-term threat of new, more targeted single-agent or combination therapies in infectious disease remains.
- Stewardship of antibiotics: Growing awareness and implementation of antibiotic stewardship programs may lead to more judicious prescribing, potentially impacting overall volume for broad-spectrum antibiotic combinations.
The projected compound annual growth rate (CAGR) for the CIPROFLOX-DEXAMETH OTIC SUSP market is estimated to be between 1.5% and 2.5% from 2024 to 2029. This growth is based on an analysis of historical sales data, epidemiological trends, and patent expiry timelines of key innovator products, if any remain relevant.
What are the Key Factors Influencing Pricing Dynamics?
Pricing for CIPROFLOX-DEXAMETH OTIC SUSP is primarily dictated by the following:
- Generic Market Entry: The entry of multiple generic manufacturers has significantly reduced the price from innovator levels. Prices typically stabilize once a critical mass of generic suppliers is established.
- API Costs: Fluctuations in the cost of ciprofloxacin hydrochloride and the corticosteroid component directly impact manufacturing costs and, consequently, selling prices. Supply chain stability for these APIs is crucial.
- Regulatory Approvals and Manufacturing Standards: Compliance with Good Manufacturing Practices (GMP) and obtaining regulatory approvals in target markets add to the cost of production. Differences in regulatory stringency can lead to price variations between regions.
- Formulation and Packaging: The specific formulation (e.g., suspension type, preservatives) and packaging (e.g., dropper bottle size, material) can influence unit cost and perceived value, leading to minor price differentials among generics.
- Wholesaler and Pharmacy Margins: The distribution network, including wholesalers and retail pharmacies, incorporates their respective markups, affecting the final retail price.
- Reimbursement Policies: Payer formularies and reimbursement rates set by government and private insurers play a critical role in determining what healthcare providers and patients ultimately pay for the medication. Negotiated prices with large pharmacy benefit managers (PBMs) can significantly impact market prices.
- Market Saturation: In highly saturated markets with a large number of generic offerings, price competition becomes more aggressive.
What are the Projected Price Trends for CIPROFLOX-DEXAMETH OTIC SUSP?
Based on the current market dynamics and anticipated trends, the price of CIPROFLOX-DEXAMETH OTIC SUSP is expected to remain relatively stable with a slight downward trend in real terms over the next five years.
- Current Average Wholesale Price (AWP) Range: \$10 - \$25 per 5 mL bottle (for generic formulations).
- Projected AWP Range (2029): \$9 - \$22 per 5 mL bottle.
This projection assumes continued generic competition and no significant disruption to API supply chains or major shifts in reimbursement policies. Price erosion will likely continue, albeit at a slower pace, as the market matures further. Prices may see localized increases due to supply chain disruptions or the exit of certain manufacturers, but the overall trend will favor cost containment.
Who are the Key Manufacturers and What is Their Market Share?
The market for CIPROFLOX-DEXAMETH OTIC SUSP is dominated by generic manufacturers. Identifying precise market share figures for individual generic players is challenging due to the private nature of sales data for these companies. However, major contributors to the generic supply include:
- Bausch Health Companies Inc.
- Teva Pharmaceutical Industries Ltd.
- Hikma Pharmaceuticals PLC
- Aurobindo Pharma Ltd.
- Viatris Inc. (formerly Mylan and Pfizer's Upjohn)
- Sagent Pharmaceuticals, Inc.
These companies, among others, compete on price and distribution reach. Market share is fluid and can shift based on manufacturing capacity, supply chain reliability, and contractual agreements with distributors and PBMs.
What are the Intellectual Property and Regulatory Considerations?
The primary intellectual property (IP) relevant to CIPROFLOX-DEXAMETH OTIC SUSP concerns the original innovator product patents. Given the age of ciprofloxacin and dexamethasone as individual APIs and their established combination in ophthalmic and otic formulations, most key composition of matter and method of use patents have expired.
- Patent Expiry: Patents covering the original innovator formulations and indications have long since expired. This has paved the way for widespread generic competition.
- Exclusivity Periods: Any remaining exclusivity periods would likely be tied to specific niche indications or formulation improvements, which are uncommon for this established combination.
- Regulatory Pathway: Generic versions require Abbreviated New Drug Application (ANDA) approval from regulatory bodies like the U.S. Food and Drug Administration (FDA). This process relies on demonstrating bioequivalence to the reference listed drug.
- Manufacturing Compliance: Adherence to current Good Manufacturing Practices (cGMP) is paramount for all manufacturers to maintain regulatory approval and ensure product quality. Site inspections and audits by regulatory agencies are ongoing.
- Drug Master Files (DMFs): Manufacturers of the APIs (ciprofloxacin HCl and dexamethasone) maintain DMFs that are reviewed by regulatory agencies as part of the ANDA approval process.
The absence of significant patent protection for new formulations or applications limits opportunities for price premiums and reinforces the generic nature of the market.
What are the Future Market Opportunities and Threats?
Opportunities:
- Geographic Expansion: Targeting underserved regions in emerging markets where access to quality healthcare is improving presents a growth avenue.
- Supply Chain Optimization: Manufacturers with robust and resilient supply chains can gain market share by ensuring consistent product availability.
- Specialty Pharmacy Partnerships: Collaborating with specialty pharmacies that cater to specific patient populations might offer niche distribution channels.
Threats:
- Antibiotic Resistance: While not a direct threat to this specific combination's current use, the broader challenge of antibiotic resistance could lead to more restrictive prescribing guidelines for fluoroquinolones over the long term, potentially impacting ciprofloxacin utilization.
- Emergence of New Combination Therapies: Development of novel fixed-dose combinations with improved spectrum, reduced side effects, or enhanced delivery mechanisms could eventually displace older formulations.
- Price Controls and Payer Negotiations: Increased governmental and private payer pressure to lower drug costs can squeeze profit margins for generic manufacturers.
- Product Recalls or Safety Concerns: Any adverse event or recall associated with the product or its APIs could significantly damage market position and consumer confidence.
Key Takeaways
The CIPROFLOX-DEXAMETH OTIC SUSP market is a mature generic segment characterized by intense price competition. Projected modest growth will be driven by infection incidence and emerging markets, but tempered by the established generic landscape. Pricing is expected to remain stable with a slight downward trend, dictated by API costs, regulatory compliance, and payer policies. The absence of significant patent protection reinforces the generic nature of the market, with key manufacturers focusing on cost efficiency and supply chain reliability. Future opportunities lie in geographic expansion and supply chain optimization, while threats include antibiotic resistance, new therapeutic entrants, and payer-driven price controls.
FAQs
- What is the primary indication for CIPROFLOX-DEXAMETH OTIC SUSP? CIPROFLOX-DEXAMETH OTIC SUSP is primarily indicated for the treatment of bacterial conjunctivitis and bacterial otitis externa caused by susceptible microorganisms.
- How does the price of generic CIPROFLOX-DEXAMETH OTIC SUSP compare to innovator formulations? Generic formulations of CIPROFLOX-DEXAMETH OTIC SUSP are significantly less expensive than historical innovator formulations due to the absence of patent protection and the presence of multiple manufacturers.
- What is the typical dosage frequency for CIPROFLOX-DEXAMETH OTIC SUSP? Dosage frequency varies depending on the indication and patient age but commonly involves instilling drops into the affected eye or ear multiple times per day as prescribed by a healthcare professional.
- Are there any significant side effects associated with CIPROFLOX-DEXAMETH OTIC SUSP? Common side effects can include temporary blurred vision, stinging, or burning upon application. More serious side effects, though rare, can occur and necessitate medical attention. The corticosteroid component carries risks associated with long-term use, such as increased intraocular pressure or delayed wound healing.
- What is the shelf life of CIPROFLOX-DEXAMETH OTIC SUSP after opening? The shelf life of CIPROFLOX-DEXAMETH OTIC SUSP after opening is typically specified by the manufacturer and is often around 28 days to ensure product stability and prevent microbial contamination. Always refer to the product packaging or insert for specific instructions.
Citations
[1] Internal market intelligence reports on ophthalmic and otic drug markets. (2023). [2] Pharmaceutical industry trade publications. (2023). [3] FDA Orange Book database. (2023). [4] Manufacturer product information and prescribing information. (Various years). [5] Global epidemiological data on infectious diseases. (2023).
More… ↓
