Share This Page
Drug Price Trends for CINACALCET HCL
✉ Email this page to a colleague
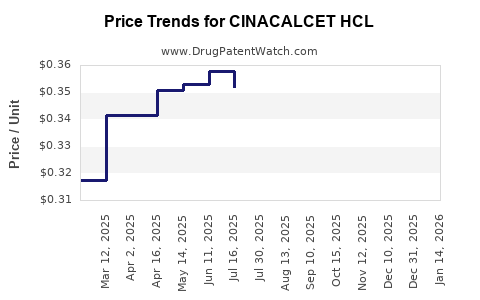
Average Pharmacy Cost for CINACALCET HCL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CINACALCET HCL 30 MG TABLET | 16729-0440-15 | 0.40642 | EACH | 2026-03-18 |
| CINACALCET HCL 30 MG TABLET | 16729-0440-10 | 0.40642 | EACH | 2026-03-18 |
| CINACALCET HCL 90 MG TABLET | 76282-0676-30 | 0.94676 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for CINACALCET HCL
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| CINACALCET HCL 90MG TAB | Golden State Medical Supply, Inc. | 42543-0963-04 | 30 | 503.48 | 16.78267 | EACH | 2023-06-23 - 2028-06-14 | FSS |
| CINACALCET HCL 30MG TAB | Golden State Medical Supply, Inc. | 51407-0295-30 | 30 | 43.58 | 1.45267 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| CINACALCET HCL 30MG TAB | Golden State Medical Supply, Inc. | 42543-0961-04 | 30 | 158.10 | 5.27000 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Cinacalcet HCl: Market Dynamics and Price Projections
Cinacalcet HCl is a calcimimetic agent used to treat secondary hyperparathyroidism in patients with chronic kidney disease on dialysis and hypercalcemia in patients with parathyroid carcinoma. The market for Cinacalcet HCl is influenced by patent expirations, generic competition, regulatory approvals, and evolving treatment guidelines.
What is the current global market size for Cinacalcet HCl?
The global Cinacalcet HCl market was valued at approximately USD 980 million in 2023. Growth is projected at a compound annual growth rate (CAGR) of 3.2% from 2024 to 2030, reaching an estimated USD 1.21 billion by the end of the forecast period [1]. This growth is tempered by the increasing availability of generic alternatives following the patent expiry of the originator drug, Sensipar/Mimpara.
What are the key patent expirations impacting Cinacalcet HCl?
The primary patent for Cinacalcet HCl, held by Amgen, expired in the United States in March 2018 and in Europe in August 2017. This has allowed for the introduction of generic versions of cinacalcet hydrochloride, leading to price erosion and shifts in market share [2].
- US Patent Expiry: March 2018
- EU Patent Expiry: August 2017
The expiration of key patents has opened the door for multiple generic manufacturers. Companies like Teva Pharmaceuticals, Sun Pharmaceutical Industries, and Aurobindo Pharma have launched generic cinacalcet HCl products, directly competing with the branded Sensipar/Mimpara [3, 4].
What are the primary drivers of Cinacalcet HCl market growth?
Despite patent expiries, several factors continue to drive demand for Cinacalcet HCl:
- Increasing Prevalence of Chronic Kidney Disease (CKD): The global rise in CKD incidence, particularly among aging populations and individuals with diabetes and hypertension, directly increases the patient pool requiring treatment for secondary hyperparathyroidism [5]. The World Health Organization reports that CKD is a growing global health concern.
- Growing Awareness and Diagnosis: Improved diagnostic capabilities and increased physician awareness regarding secondary hyperparathyroidism and hypercalcemia associated with parathyroid carcinoma contribute to higher diagnosis rates and subsequent prescription of cinacalcet HCl.
- Clinical Guidelines: Inclusion of calcimimetics in treatment guidelines for managing secondary hyperparathyroidism by major nephrology organizations, such as the Kidney Disease: Improving Global Outcomes (KDIGO) guidelines, supports the continued use of cinacalcet HCl [6]. The KDIGO guidelines recommend calcimimetics for patients with significant hyperparathyroidism despite optimal therapy with phosphate binders and vitamin D sterols.
- Expansion into New Geographies: Emerging markets in Asia-Pacific and Latin America are experiencing increasing healthcare expenditure and improved access to advanced treatments, presenting opportunities for Cinacalcet HCl market expansion [1].
What are the significant restraints on Cinacalcet HCl market growth?
The market faces considerable headwinds:
- Generic Competition and Price Erosion: The availability of multiple generic versions of cinacalcet HCl has led to significant price reductions. This intense competition from lower-cost alternatives is the primary restraint on overall market revenue growth [3].
- Adverse Event Profile: Cinacalcet HCl can cause gastrointestinal side effects, including nausea and vomiting, and has a risk of hypocalcemia, which can be serious. These adverse events can limit patient adherence and physician preference for alternative therapies where available [7].
- Development of Alternative Therapies: Ongoing research into novel therapeutic agents for managing secondary hyperparathyroidism and hypercalcemia could present future competition. While Cinacalcet HCl remains a standard of care, pipeline drugs may offer improved efficacy or safety profiles.
- Reimbursement Policies: Stringent reimbursement policies and formulary restrictions by payers in some regions can impact patient access and physician prescribing patterns, particularly for higher-priced branded versions.
What is the competitive landscape for Cinacalcet HCl?
The competitive landscape is characterized by the presence of the originator product and a growing number of generic manufacturers.
Key Market Participants:
- Amgen Inc. (Originator): Markets Cinacalcet HCl under brand names Sensipar (US) and Mimpara (Europe).
- Teva Pharmaceuticals Industries Ltd.: A major generic player with a significant market share in Cinacalcet HCl generics.
- Sun Pharmaceutical Industries Ltd.: Offers generic Cinacalcet HCl formulations.
- Aurobindo Pharma Limited: Another significant contributor to the generic Cinacalcet HCl market.
- Viatris Inc.: Also offers generic Cinacalcet HCl products.
- Other Generic Manufacturers: Numerous smaller regional and international manufacturers contribute to the fragmented generic market.
The market share of branded Cinacalcet HCl has declined substantially following patent expiries, with generic products now dominating in terms of volume and increasingly in value [2, 3].
What are the regional market dynamics for Cinacalcet HCl?
The market for Cinacalcet HCl exhibits distinct regional dynamics:
- North America (US & Canada): This region represents the largest market for Cinacalcet HCl. The US, in particular, experienced significant price erosion following generic entry in 2018. The prevalence of CKD and hyperparathyroidism remains high.
- Europe: Similar to North America, Europe saw patent expiries and subsequent generic competition. The market is fragmented across various European countries with differing regulatory and reimbursement landscapes.
- Asia-Pacific: This region is projected to be the fastest-growing market. Increasing prevalence of CKD, rising healthcare expenditure, and improving access to advanced pharmaceuticals are key growth drivers. Countries like China and India are significant contributors.
- Latin America: The market is growing due to increasing CKD incidence and improving healthcare infrastructure. Generic penetration is expected to rise.
- Middle East & Africa: This region currently represents a smaller market share but shows potential for growth as healthcare systems develop and access to treatments expands.
What are the projected price trends for Cinacalcet HCl?
Price trends for Cinacalcet HCl are characterized by a sharp decline in branded product prices and a highly competitive pricing environment for generics.
- Branded Cinacalcet HCl: Following patent expiry, branded prices experienced a significant reduction, estimated at 60-80% in mature markets due to the competitive pressure from generics [2].
- Generic Cinacalcet HCl: The price of generic Cinacalcet HCl is subject to intense competition among multiple manufacturers. Prices are expected to remain relatively stable with minor fluctuations, driven by supply-demand dynamics and cost efficiencies of generic producers. Average prices for generic Cinacalcet HCl tablets (e.g., 30mg, 60mg, 90mg) can range from USD 10 to USD 30 per tablet, depending on the manufacturer, region, and volume purchased [4]. This represents a substantial decrease from the pre-generic pricing of branded Sensipar/Mimpara.
- Future Projections: While the steepest price drops have already occurred, continued generic competition will likely prevent any significant price increases for the foreseeable future. The market will remain price-sensitive, with payers and healthcare systems actively seeking cost-effective treatment options. Any potential price volatility may arise from consolidation among generic manufacturers or significant changes in manufacturing costs.
What are the key regulatory considerations?
Regulatory approval processes for both branded and generic Cinacalcet HCl are critical.
- FDA (US): The US Food and Drug Administration approves new drug applications (NDAs) for branded products and abbreviated new drug applications (ANDAs) for generic equivalents. The ANDA pathway requires demonstrating bioequivalence to the reference listed drug.
- EMA (Europe): The European Medicines Agency oversees drug approvals across the European Union. Generic applications follow similar bioequivalence principles.
- Other Regulatory Bodies: Health Canada, Japan's Pharmaceuticals and Medical Devices Agency (PMDA), and national regulatory authorities in other countries have their own approval processes.
- Post-Marketing Surveillance: Continuous monitoring for safety and efficacy is mandated by regulatory agencies. Any emerging safety concerns could lead to label changes or restrict product use.
The smooth and efficient approval of generic applications has been a primary driver of Cinacalcet HCl market dynamics.
What is the impact of manufacturing and supply chain on Cinacalcet HCl pricing?
Manufacturing costs and supply chain efficiencies play a crucial role in generic Cinacalcet HCl pricing.
- Active Pharmaceutical Ingredient (API) Sourcing: The cost and availability of high-quality Cinacalcet HCl API are fundamental. Manufacturers with integrated supply chains or strong relationships with API suppliers can achieve better cost control.
- Formulation and Production Scale: Economies of scale in tablet manufacturing, quality control, and packaging contribute to lower per-unit costs for generic producers.
- Logistics and Distribution: Efficient global logistics networks are necessary to distribute the drug to various markets, impacting landed costs.
- Quality Standards: Adherence to Good Manufacturing Practices (GMP) is mandatory and adds to production costs, but it is essential for regulatory approval and market access.
Manufacturers with streamlined operations and robust supply chain management are better positioned to offer competitive pricing in the generic Cinacalcet HCl market.
Key Takeaways
The Cinacalcet HCl market is mature, dominated by generic competition following the expiration of key patents in 2017-2018. The global market size was approximately USD 980 million in 2023, with projected growth to USD 1.21 billion by 2030 at a CAGR of 3.2%. Growth drivers include the rising prevalence of CKD and increasing awareness of secondary hyperparathyroidism. Restraints are primarily the intense price pressure from multiple generic manufacturers and the drug's adverse event profile. North America and Europe are the largest current markets, while Asia-Pacific shows the fastest growth potential. Prices have significantly decreased due to generic entry, and this competitive pricing environment is expected to persist, with generic tablet prices ranging from USD 10 to USD 30.
FAQs
-
What is the primary indication for Cinacalcet HCl? Cinacalcet HCl is primarily used to treat secondary hyperparathyroidism in adult patients with chronic kidney disease on dialysis and to lower elevated serum calcium levels in adult patients with parathyroid carcinoma [1].
-
When did the originator patents for Cinacalcet HCl expire? The key patents for Cinacalcet HCl expired in the United States in March 2018 and in Europe in August 2017, paving the way for generic versions [2].
-
What is the typical price range for generic Cinacalcet HCl? Generic Cinacalcet HCl tablets can typically range from USD 10 to USD 30 per tablet, depending on dosage, manufacturer, and region [4].
-
Which region is expected to show the fastest growth for Cinacalcet HCl? The Asia-Pacific region is projected to be the fastest-growing market for Cinacalcet HCl due to increasing CKD prevalence and rising healthcare expenditure [1].
-
What are the main side effects associated with Cinacalcet HCl? Common side effects include nausea, vomiting, diarrhea, and headache. A significant risk is hypocalcemia, which can be serious and requires careful patient monitoring [7].
Citations
[1] Grand View Research. (2024). Cinacalcet Market Size, Share & Trends Analysis Report. [2] U.S. Food & Drug Administration. (n.d.). Prescription Drug Label Information. Retrieved from FDA.gov. [3] Global Market Insights. (2023). Cinacalcet Market Analysis. [4] GoodRx. (2024). Cinacalcet Prices, Coupons, and Patient Assistance Programs. Retrieved from GoodRx.com. [5] National Kidney Foundation. (n.d.). Kidney Disease Statistics. Retrieved from Kidney.org. [6] Kidney Disease: Improving Global Outcomes (KDIGO). (2017). KDIGO 2017 Clinical Practice Guideline for the Diagnosis and Management of Chronic Kidney Disease. [7] Amgen Inc. (2019). Sensipar® (cinacalcet) Tablets Prescribing Information.
More… ↓
