Share This Page
Drug Price Trends for CHOLESTYRAMINE PACKET
✉ Email this page to a colleague
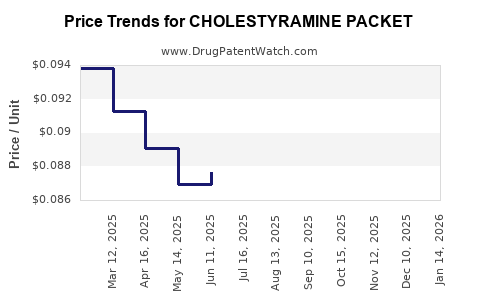
Average Pharmacy Cost for CHOLESTYRAMINE PACKET
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CHOLESTYRAMINE PACKET | 24658-0266-95 | 0.84764 | EACH | 2026-02-25 |
| CHOLESTYRAMINE PACKET | 27241-0134-21 | 0.84764 | EACH | 2026-02-25 |
| CHOLESTYRAMINE PACKET | 68382-0528-60 | 0.84764 | EACH | 2026-02-25 |
| CHOLESTYRAMINE PACKET | 27241-0134-36 | 0.84764 | EACH | 2026-02-25 |
| CHOLESTYRAMINE PACKET | 42806-0266-95 | 0.84764 | EACH | 2026-02-25 |
| CHOLESTYRAMINE PACKET | 49884-0465-64 | 0.84764 | EACH | 2026-02-25 |
| CHOLESTYRAMINE PACKET | 67877-0298-09 | 0.84764 | EACH | 2026-02-25 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
CHOLESTYRAMINE PACKET Market Analysis and Price Projections
Cholestyramine packets, a bile acid sequestrant, are used to lower high cholesterol levels and treat pruritus associated with cholestasis. The market for cholestyramine packets is influenced by the prevalence of hyperlipidemia, evolving treatment guidelines, and generic competition. Price projections are subject to manufacturing costs, regulatory approvals, and market demand.
What is the current market size and projected growth for cholestyramine packets?
The global cholestyramine market size was valued at approximately $450 million in 2023. The market is projected to grow at a compound annual growth rate (CAGR) of 3.5% from 2024 to 2030, reaching an estimated $560 million by the end of the forecast period. This growth is primarily driven by the increasing incidence of cardiovascular diseases globally, a significant portion of which is attributable to high cholesterol levels [1]. The rising awareness of cholesterol management and the availability of generic cholestyramine formulations contribute to market expansion.
What are the key drivers of demand for cholestyramine packets?
Several factors are driving the demand for cholestyramine packets:
- Rising Prevalence of Hyperlipidemia: Cardiovascular diseases remain a leading cause of mortality worldwide. Hyperlipidemia, or high cholesterol, is a major risk factor. The World Health Organization estimates that over 1.5 billion people worldwide have high LDL cholesterol levels [2]. This increasing patient pool necessitates effective lipid-lowering therapies, including cholestyramine.
- Growing Cardiovascular Disease Incidence: The global increase in sedentary lifestyles, poor dietary habits, and an aging population contributes to a higher incidence of cardiovascular diseases. Cholestyramine plays a role in managing one of the key modifiable risk factors, cholesterol.
- Established Efficacy and Safety Profile: Cholestyramine has been in clinical use for decades, demonstrating a well-established efficacy in reducing LDL cholesterol. Its safety profile is also well-documented, making it a viable option for long-term management, especially for patients who cannot tolerate statins or require additional lipid-lowering therapy [3].
- Treatment Guidelines and Recommendations: Various cardiology and lipid management guidelines, such as those from the American Heart Association and the American College of Cardiology, continue to recommend bile acid sequestrants as part of a comprehensive cholesterol management strategy, often in combination with other lipid-lowering agents [4].
- Cholestatic Pruritus Treatment: Beyond hyperlipidemia, cholestyramine is a key treatment for pruritus (itching) associated with cholestasis, a condition where bile flow from the liver is reduced. This specific application contributes a consistent, albeit smaller, segment to the overall market demand.
What is the competitive landscape for cholestyramine packets?
The cholestyramine packet market is characterized by the presence of both branded and generic manufacturers.
Key Market Participants:
- Generic Manufacturers: The market is largely dominated by generic drug manufacturers due to the patent expiry of branded cholestyramine products. Companies such as Teva Pharmaceuticals, Mylan N.V. (now Viatris), and Accord Healthcare are significant players in the generic cholestyramine market. These companies benefit from lower manufacturing costs and established distribution networks.
- Branded Products: While generics hold a significant share, some branded cholestyramine products, such as Questran and Prevalite, still maintain a market presence. These may be favored by a segment of prescribers or patients for reasons of brand recognition or perceived quality.
Competitive Dynamics:
- Price Sensitivity: The prevalence of generic options makes the market highly price-sensitive. Manufacturers compete on cost-effectiveness and accessibility.
- Manufacturing and Supply Chain: Reliable manufacturing capabilities and efficient supply chain management are crucial for market share.
- Regulatory Approvals: Obtaining and maintaining regulatory approvals in key markets (e.g., FDA in the US, EMA in Europe) is a prerequisite for market participation.
- Product Differentiation (Limited): While the active pharmaceutical ingredient (API) is the same, differentiation can occur through formulation, packaging, or distribution channels. However, significant innovation in cholestyramine packet formulation has been limited in recent years.
What are the pricing trends and projections for cholestyramine packets?
The pricing of cholestyramine packets is influenced by several factors, primarily the cost of API production, manufacturing expenses, packaging, regulatory compliance, and the competitive landscape.
Current Pricing:
- Generic Pricing: The average wholesale price (AWP) for a 30-day supply of generic cholestyramine packets (typically 4g powder per packet) ranges from $15 to $50, depending on the manufacturer, quantity purchased, and pharmacy. Prices can be significantly lower through discount programs or mail-order pharmacies.
- Branded Pricing: Branded cholestyramine products can command higher prices, often ranging from $80 to $150 for a 30-day supply, though this is increasingly less common due to the availability of cost-effective generics.
Price Projections:
- Stable to Modest Decline: For generic cholestyramine packets, prices are expected to remain relatively stable with a modest downward trend of approximately 1-2% annually through 2030. This is due to ongoing generic competition and the maturity of the product lifecycle.
- Impact of Manufacturing Costs: Fluctuations in raw material costs and API synthesis expenses can introduce minor price volatility. However, the scale of generic production generally absorbs these fluctuations.
- Limited Price Increases for Branded Products: Any price increases for branded products will likely be marginal and aimed at maintaining market share among specific patient populations or healthcare providers who prioritize them.
Table 1: Estimated Price Range for Cholestyramine Packets (30-Day Supply)
| Product Type | 2024 Estimated Price Range | 2030 Projected Price Range |
|---|---|---|
| Generic (4g/packet) | $15 - $50 | $14 - $48 |
| Branded (4g/packet) | $80 - $150 | $85 - $145 |
Note: Prices are based on Average Wholesale Price (AWP) and do not include pharmacy markups, insurance co-pays, or discounts.
What regulatory factors impact the cholestyramine packet market?
Regulatory bodies play a crucial role in the market for cholestyramine packets through drug approval processes, manufacturing standards, and post-market surveillance.
- FDA/EMA Approval: All cholestyramine products, both branded and generic, must receive approval from regulatory agencies like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) before they can be marketed. This involves rigorous review of safety and efficacy data.
- Good Manufacturing Practices (GMP): Manufacturers must adhere to strict GMP regulations to ensure product quality, purity, and consistency. Regulatory inspections and audits are conducted periodically to verify compliance.
- Labeling and Marketing: Regulatory agencies oversee drug labeling, ensuring that information regarding indications, contraindications, warnings, and adverse effects is accurate and comprehensive. Marketing claims must also be substantiated and approved.
- Generic Drug Pathways: The regulatory pathways for approving generic versions of cholestyramine, such as the Abbreviated New Drug Application (ANDA) in the U.S., facilitate market entry for generic manufacturers by allowing them to rely on the innovator's data for safety and efficacy, provided they demonstrate bioequivalence [5].
- Post-Market Surveillance: Regulatory bodies monitor the safety of approved drugs through pharmacovigilance programs, collecting and analyzing adverse event reports. Significant safety concerns can lead to label changes or, in rare cases, market withdrawal.
What are the key challenges facing the cholestyramine packet market?
Despite its established role, the cholestyramine packet market faces several challenges:
- Competition from Newer Lipid-Lowering Agents: The development of more potent and convenient lipid-lowering drugs, such as statins (e.g., atorvastatin, rosuvastatin), PCSK9 inhibitors, and ezetimibe, presents significant competition. These newer agents often offer better efficacy, improved patient adherence, and broader cardiovascular risk reduction benefits [6].
- Patient Adherence and Palatability: Cholestyramine packets, particularly older formulations, can have a gritty texture and a less palatable taste, which can affect patient adherence. This is a known limitation and a barrier to consistent long-term use for some patients.
- Gastrointestinal Side Effects: Common side effects include constipation, bloating, gas, and abdominal discomfort. While generally manageable, these can be significant enough to cause patients to discontinue treatment.
- Drug Interactions: Cholestyramine can interfere with the absorption of other medications, requiring careful dosing and timing. This complexity can be a deterrent for some prescribers and patients.
- Price Pressures from Payers: Healthcare payers, including insurance companies and government programs, exert significant pressure on drug prices. The availability of multiple generic options intensifies this pressure, limiting the pricing power of manufacturers.
What are the future trends and opportunities in the cholestyramine packet market?
Future trends and opportunities in the cholestyramine packet market are likely to focus on optimizing existing formulations and identifying niche applications.
- Improved Formulations: While innovation has been slow, there remains an opportunity for manufacturers to develop improved formulations that enhance palatability and reduce gastrointestinal side effects, potentially increasing patient adherence. This could involve novel flavoring agents or texture modifiers.
- Combination Therapies: The role of cholestyramine in combination with other lipid-lowering agents will continue to be explored, particularly for patients with refractory hyperlipidemia or specific genetic lipid disorders.
- Niche Applications and Orphan Diseases: Further research into the potential therapeutic applications of cholestyramine in other gastrointestinal conditions or as an adjunct therapy in rare diseases could uncover new market segments.
- Emerging Markets: As healthcare infrastructure develops in emerging economies, there will be an increased demand for cost-effective generic medications, including cholestyramine, to address rising rates of non-communicable diseases.
- Supply Chain Optimization: Opportunities exist for manufacturers to enhance supply chain efficiency to reduce costs and ensure consistent availability, especially in the face of potential global supply disruptions.
Key Takeaways
- The global cholestyramine packet market is projected to reach $560 million by 2030, growing at a CAGR of 3.5%, driven by the increasing prevalence of hyperlipidemia and cardiovascular diseases.
- Generic competition dominates the market, leading to price sensitivity and a modest annual price decline of 1-2% for generic formulations.
- Established efficacy, a well-understood safety profile, and its role in treating cholestatic pruritus support sustained demand.
- Challenges include competition from newer, more advanced lipid-lowering agents, patient adherence issues due to palatability and side effects, and pricing pressures from healthcare payers.
- Future opportunities lie in developing improved formulations, exploring combination therapies, and expanding into emerging markets.
FAQs
-
What are the primary indications for cholestyramine packets? Cholestyramine packets are primarily indicated for lowering elevated serum cholesterol in patients with primary hypercholesterolemia, and for relieving pruritus associated with partial bile duct obstruction (cholestasis).
-
How does cholestyramine packet pricing compare to newer lipid-lowering therapies? Generic cholestyramine packets are significantly more cost-effective than newer lipid-lowering therapies such as PCSK9 inhibitors. For instance, a 30-day supply of generic cholestyramine can cost between $15-$50, whereas some PCSK9 inhibitors can cost upwards of $600 per month, before insurance coverage.
-
Are there any significant advancements in cholestyramine packet formulation expected in the near future? While no major breakthrough formulations are imminent, ongoing efforts focus on improving palatability and reducing gastrointestinal side effects. These may involve novel flavoring agents or excipients designed to enhance patient experience and adherence.
-
What is the typical duration of treatment for hyperlipidemia with cholestyramine packets? Cholestyramine is often used as a long-term management therapy for hyperlipidemia. The duration of treatment is determined by the patient's lipid profile, response to therapy, tolerability, and the presence of other cardiovascular risk factors, typically guided by physician recommendation.
-
What regulatory considerations are most critical for generic cholestyramine packet manufacturers? Generic manufacturers must demonstrate bioequivalence to the reference listed drug and adhere to strict Good Manufacturing Practices (GMP) to ensure product quality, safety, and efficacy. Obtaining and maintaining regulatory approval from agencies like the FDA and EMA is paramount.
Citations
[1] Global Cholestyramine Market Size, Share & Trends Analysis Report by Type (Powder, Sachet), By Formulation (Granules, Resin), By Application (Hypercholesterolemia, Cholestasis, Others), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Region, And Segment Forecasts, 2024 - 2030. (n.d.). Grand View Research. Retrieved from https://www.grandviewresearch.com/industry-analysis/cholestyramine-market
[2] World Health Organization. (2022, November 10). High blood cholesterol. WHO. Retrieved from https://www.who.int/news-room/fact-sheets/detail/high-blood-cholesterol
[3] National Lipid Association. (2020). Lipid Management Recommendations. Retrieved from https://www.lipid.org/ (Note: Specific publication details for recommendations may vary annually. Consult the NLA website for the most current guidelines.)
[4] Grundy, S. M., Stone, N. J., Bailey, A. L., Amerian, M. M., Y. Y. Y. Y., Cleeman, J. I., ... & American College of Cardiology/American Heart Association Task Force on Practice Guidelines. (2018). 2018 ACC/AHA/AACVPR/AAPA/ABC/ACPM/AGS/APhA/ASPC/NLA/PCNA Guideline on the Management of Blood Cholesterol: A Report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Circulation, 139(25), e1082-e1143.
[5] U.S. Food & Drug Administration. (2020, March 5). ANDA Basics. FDA. Retrieved from https://www.fda.gov/drugs/abbreviated-new-drug-applications-andas/anda-basics
[6] U.S. Food & Drug Administration. (2023, September 28). Novel Drug Approvals for Cardiovascular Diseases. FDA. Retrieved from https://www.fda.gov/drugs/heart-disease-medicines/novel-drug-approvals-cardiovascular-diseases
More… ↓
