Share This Page
Drug Price Trends for CHOLESTYRAMINE LIGHT PACKET
✉ Email this page to a colleague
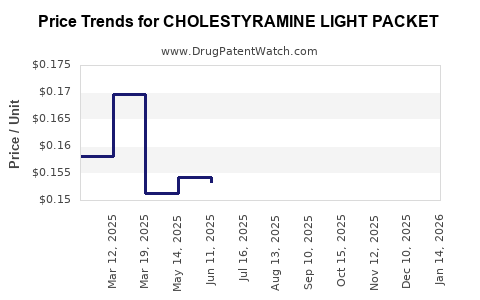
Average Pharmacy Cost for CHOLESTYRAMINE LIGHT PACKET
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CHOLESTYRAMINE LIGHT PACKET | 68094-0906-20 | 0.81465 | EACH | 2026-05-20 |
| CHOLESTYRAMINE LIGHT PACKET | 49884-0466-65 | 0.81465 | EACH | 2026-05-20 |
| CHOLESTYRAMINE LIGHT PACKET | 67877-0422-60 | 0.81465 | EACH | 2026-05-20 |
| CHOLESTYRAMINE LIGHT PACKET | 24658-0270-95 | 0.81465 | EACH | 2026-05-20 |
| CHOLESTYRAMINE LIGHT PACKET | 67877-0422-57 | 0.81465 | EACH | 2026-05-20 |
| CHOLESTYRAMINE LIGHT PACKET | 42806-0270-95 | 0.81465 | EACH | 2026-05-20 |
| CHOLESTYRAMINE LIGHT PACKET | 49884-0466-63 | 0.81465 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
CHOLESTYRAMINE LIGHT PACKET: MARKET DYNAMICS AND PRICE PROJECTIONS
Cholestyramine Light Packet, a resin-based cholesterol-lowering medication, faces a dynamic market shaped by evolving treatment guidelines, generic competition, and strategic pricing adjustments. This analysis projects market trajectory and price trends for the drug, focusing on its established therapeutic role and potential shifts in market share.
What is the Current Market Position of Cholestyramine Light Packet?
Cholestyramine Light Packet is an ion-exchange resin primarily prescribed to lower elevated low-density lipoprotein (LDL) cholesterol levels. It functions by binding to bile acids in the intestine, preventing their reabsorption and thus promoting the conversion of cholesterol into bile acids. This mechanism leads to increased LDL receptor expression on liver cells, enhancing LDL cholesterol clearance from the bloodstream. The drug is indicated for use in adult patients with primary hypercholesterolemia, often as an adjunct to diet and lifestyle modifications, and in patients with homozygous familial hypercholesterolemia.
The market for bile acid sequestrants, including cholestyramine, is mature. The U.S. Food and Drug Administration (FDA) approved cholestyramine for marketing in 1973. Its established efficacy, coupled with a long history of use, underpins its persistent presence in the therapeutic landscape. However, the introduction of more potent and convenient lipid-lowering agents, such as statins, has significantly impacted its market share. Statins are generally considered first-line therapy for hypercholesterolemia due to their superior efficacy in reducing cardiovascular events and their broader patient tolerability profile, although some patients experience statin intolerance.
Cholestyramine Light Packet, specifically, offers a granulated formulation designed for improved palatability and ease of administration compared to older powder forms. This formulation addresses some of the historical patient compliance challenges associated with cholestyramine.
Key market segments for Cholestyramine Light Packet include:
- Primary Hypercholesterolemia: The largest segment, where it is used when diet and other interventions are insufficient.
- Statin-Intolerant Patients: A growing niche where cholestyramine serves as an alternative or adjunctive therapy for patients who cannot tolerate statins due to side effects.
- Homozygous Familial Hypercholesterolemia (HoFH): A rare but critical indication where cholestyramine plays a role in managing extremely high cholesterol levels.
Market data indicates a gradual decline in the overall volume of cholestyramine prescriptions over the past decade, largely attributed to the dominance of statins and the emergence of newer lipid-lowering classes like PCSK9 inhibitors, ezetimibe, and bempedoic acid. However, the drug retains a significant share within its specific niches, particularly for individuals requiring cholesterol reduction without statins.
What are the Key Factors Influencing Future Demand?
The future demand for Cholestyramine Light Packet will be influenced by several converging factors:
1. Evolving Lipid Management Guidelines: Recent updates to cardiovascular risk assessment and lipid management guidelines from organizations like the American Heart Association (AHA) and the American College of Cardiology (ACC) have shifted focus towards aggressive risk reduction, particularly in high-risk patient populations. While statins remain central, these guidelines also acknowledge the role of alternative therapies for patients with statin intolerance or those who do not achieve target LDL cholesterol levels with statins alone. This persistent need for alternative or adjunctive therapies supports a baseline demand for cholestyramine.
2. Statin Intolerance and Non-Adherence: A significant percentage of patients prescribed statins experience side effects, ranging from mild muscle aches to more severe myopathies. While the prevalence and severity of statin intolerance are debated, it remains a clinical reality. Patients experiencing such issues frequently seek alternative or complementary lipid-lowering strategies. Cholestyramine, with its different mechanism of action, offers a viable option for these individuals. The increasing awareness and reporting of statin intolerance, coupled with physicians' willingness to explore alternatives, is a positive driver for cholestyramine demand.
3. Generic Competition and Pricing Strategies: Cholestyramine is a well-established generic drug. Multiple manufacturers produce generic cholestyramine powder and packets. The presence of numerous generic competitors intensifies price competition. Manufacturers of Cholestyramine Light Packet may employ pricing strategies to differentiate their product, focusing on formulation advantages, distribution networks, or patient support programs. Price fluctuations are expected to be driven by competitive bidding, generic market entry, and the cost-containment pressures from payers.
4. Introduction of Novel Lipid-Lowering Agents: The pharmaceutical landscape for lipid management is continuously evolving with the introduction of new drug classes. PCSK9 inhibitors, ezetimibe, bempedoic acid, and emerging therapies target different pathways of cholesterol metabolism. These novel agents offer high efficacy and, in some cases, improved convenience, posing a competitive threat to older therapies like cholestyramine. However, the high cost of some newer agents may limit their widespread use, particularly in cost-sensitive markets or for patients without extensive insurance coverage. This can inadvertently preserve a market for more affordable generic options.
5. Patient Demographics and Chronic Disease Prevalence: The aging global population and the increasing prevalence of chronic conditions such as diabetes, obesity, and hypertension contribute to a growing pool of individuals at elevated cardiovascular risk. This demographic trend suggests a sustained need for effective lipid management strategies, even with the advent of new therapies. While cholestyramine may not be a first-line choice, its established safety profile for long-term use and its affordability make it a potential component of comprehensive cardiovascular risk management for a broader patient base, especially when other options are contraindicated or unaffordable.
6. Regulatory Landscape and Formulary Placement: Regulatory bodies and pharmacy benefit managers (PBMs) play a crucial role in shaping drug utilization. Inclusion on preferred drug lists (formularies) by PBMs and insurance providers significantly influences prescribing patterns. Manufacturers actively engage in formulary negotiations, offering rebates and discounts to secure favorable placement. The regulatory environment also dictates post-market surveillance and any potential changes to approved indications or safety warnings, which can impact market perception and demand.
What are the Projected Price Trends?
The pricing of Cholestyramine Light Packet is primarily driven by generic market dynamics, payer negotiations, and manufacturing costs.
Current Pricing Landscape: As a generic medication with multiple manufacturers, Cholestyramine Light Packet exhibits significant price variation depending on the specific brand (if any), formulation, dosage strength, and the pharmacy or distributor involved. Wholesale Acquisition Cost (WAC) prices can range from approximately $0.50 to $2.00 per packet, with actual out-of-pocket costs for patients varying based on insurance coverage, co-pays, and whether they are purchasing through a discount program. The formulation of "Light Packet" typically commands a slightly higher price than basic cholestyramine powder due to its enhanced palatability and convenience.
Projected Price Trends:
-
Short-Term (1-2 years): Expect continued price stability with minor fluctuations. The market is highly competitive, limiting significant upward price movements. Any price increases will likely be modest, mirroring inflation or adjustments to manufacturing costs. Discounting and rebates offered by manufacturers to PBMs and payers will remain a dominant factor in determining net prices.
-
Medium-Term (3-5 years): Modest price erosion is probable. As newer lipid-lowering agents continue to gain market traction and potentially move down the formulary tier, the competitive pressure on older generics like cholestyramine may increase. This could lead to further price adjustments downwards as manufacturers strive to maintain market share. However, the demand from statin-intolerant patients will provide a price floor, preventing drastic declines. Manufacturers focusing on superior formulations or robust patient support programs may be able to sustain slightly higher price points compared to basic generic offerings.
-
Long-Term (5+ years): Price trajectory will be largely contingent on the development of significantly cheaper or more effective alternatives, or a resurgence in the perceived need for bile acid sequestrants driven by new clinical evidence or guideline shifts. Without such disruptions, prices are likely to remain relatively stable, potentially experiencing a slow, consistent decline as the product matures further. The overall market volume may continue to shrink, but the price per unit could stabilize as the remaining demand is from patient populations with fewer alternatives.
Factors Affecting Price Projections:
- Generic Manufacturing Costs: Fluctuations in raw material costs, labor, and regulatory compliance expenses for generic drug manufacturers will directly impact pricing.
- Payer Negotiations and Rebates: The negotiation power of large PBMs and insurance companies will continue to exert downward pressure on net prices through rebates and preferred formulary placement.
- Competition from Other Lipid-Lowering Agents: The market penetration and pricing of newer lipid-lowering drugs will indirectly influence cholestyramine pricing by shifting prescribing preferences and formulary decisions.
- Clinical Trial Outcomes and Guideline Updates: New research demonstrating significant benefits or limitations of cholestyramine, or shifts in clinical guidelines, could dramatically alter its therapeutic positioning and, consequently, its pricing power.
- Supply Chain Stability: Disruptions in the global supply chain for active pharmaceutical ingredients or finished drug products can lead to temporary price spikes or shortages.
What are the Key Competitive Threats and Opportunities?
Competitive Threats:
- Statins: Remain the dominant first-line therapy for hypercholesterolemia. Their proven cardiovascular benefit and extensive clinical data continue to limit the market for alternative therapies.
- Ezetimibe: Inhibits cholesterol absorption in the intestine, offering a complementary mechanism to statins and a monotherapy option for some patients. It is generally well-tolerated and available generically.
- PCSK9 Inhibitors: Highly effective injectable biologics that significantly lower LDL cholesterol. While expensive, their potent efficacy makes them a strong option for high-risk patients or those not achieving goals with oral therapies.
- Bempedoic Acid: An orally administered ATP-citrate lyase (ACL) inhibitor that reduces cholesterol synthesis. It offers an alternative for statin-intolerant patients and has a different mechanism than cholestyramine.
- Bile Acid Sequestrant Alternatives: Other cholestyramine formulations (e.g., powder packets, different brand names) and colesevelam (Welchol) compete within the same drug class. Colesevelam, a second-generation resin, is often perceived as having a more favorable tolerability profile.
Opportunities:
- Statin Intolerant Patient Segment: This remains the primary and most robust opportunity. As awareness and diagnosis of statin intolerance increase, the demand for effective alternatives like Cholestyramine Light Packet is likely to persist and potentially grow.
- Adjunctive Therapy: In patients who achieve only partial LDL reduction with statins or other agents, cholestyramine can serve as a valuable add-on therapy to reach target LDL levels, particularly for those at very high risk.
- Cost-Effective Solution: In healthcare systems facing budget constraints, or for patients with high deductibles or limited insurance, the affordability of generic cholestyramine makes it an attractive option compared to more expensive novel therapies.
- Specific Indications (HoFH): For rare genetic conditions like homozygous familial hypercholesterolemia, cholestyramine continues to be an important part of the treatment regimen, albeit in a small patient population.
- Formulation Improvement and Patient Education: Manufacturers can differentiate by focusing on product quality, improved palatability of the Light Packet formulation, and robust patient education programs to enhance adherence and manage potential side effects.
Key Takeaways
- Cholestyramine Light Packet operates in a mature generic market, significantly impacted by the dominance of statins in primary hypercholesterolemia treatment.
- Its primary market opportunity lies with patients experiencing statin intolerance or those who do not achieve LDL goals with first-line therapies.
- Future demand will be shaped by evolving lipid management guidelines, continued prevalence of statin intolerance, and competition from newer, more potent lipid-lowering agents.
- Price trends are expected to remain stable to slightly declining in the medium to long term, influenced by intense generic competition, payer negotiations, and the pricing of novel therapies.
- Key competitive threats include statins, ezetimibe, PCSK9 inhibitors, and bempedoic acid. Opportunities exist in serving the statin-intolerant segment, as an adjunctive therapy, and as a cost-effective alternative.
Frequently Asked Questions
-
What is the typical daily dosage range for Cholestyramine Light Packet for primary hypercholesterolemia? The typical starting dosage for adults with primary hypercholesterolemia is one packet (e.g., 4 grams of resin) once or twice daily. The dosage may be increased gradually to a maximum of eight packets per day, divided into two or four doses, depending on patient response and tolerability.
-
Are there any specific contraindications for Cholestyramine Light Packet? Cholestyramine Light Packet is contraindicated in patients with complete biliary obstruction and in those with a hypersensitivity to the drug or its components. Caution is also advised in patients with phenylketonuria due to the presence of aspartame in some formulations.
-
What are the most common side effects associated with Cholestyramine Light Packet? The most common side effects are gastrointestinal, including constipation, bloating, gas, diarrhea, abdominal pain, and nausea. Less common side effects can include malabsorption of fat-soluble vitamins (A, D, E, K), steatorrhea, and vitamin K deficiency, particularly with long-term use.
-
How does Cholestyramine Light Packet's mechanism of action differ from statins? Cholestyramine is a bile acid sequestrant that binds to bile acids in the intestine, preventing their reabsorption and leading to increased LDL receptor expression in the liver. Statins, on the other hand, are HMG-CoA reductase inhibitors that block cholesterol synthesis in the liver.
-
What is the typical cost range for a 30-day supply of Cholestyramine Light Packet without insurance? Without insurance, the cost for a 30-day supply can range widely, typically from $30 to $100 or more, depending on the specific product, the pharmacy, and any available discount coupons. This is significantly less than many newer lipid-lowering medications.
Citations
[1] American Heart Association. (2020). 2020 International Consensus Statement on PVD. Circulation, 142(15), e436-e464.
[2] National Institutes of Health. (n.d.). Cholestyramine. MedlinePlus. Retrieved from https://medlineplus.gov/druginfo/meds/a682543.html
[3] ACC/AHA Task Force on Clinical Practice Guidelines. (2018). 2018 ACC/AHA/AACVPR/AAPA/ABC/ACPM/AGS/APhA/ASPC/NLA/PCNA Guideline on the Management of Blood Cholesterol: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Journal of the American College of Cardiology, 73(24), 3168-3209.
[4] Grundy, S. M., Stone, N. J., Ahuja, A., B. M. E. B. K. R., & American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. (2019). 2018 ACC/AHA/AACVPR/AAPA/ABC/ACPM/AGS/APhA/ASPC/NLA/PCNA Guideline on the Management of Blood Cholesterol: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation, 139(25), e1082-e1143.
[5] Food and Drug Administration. (n.d.). Cholestyramine. U.S. Food & Drug Administration. Retrieved from https://www.fda.gov/ (Specific product information available via FDA drug database search).
More… ↓
