Share This Page
Drug Price Trends for CHLORTHALIDONE
✉ Email this page to a colleague
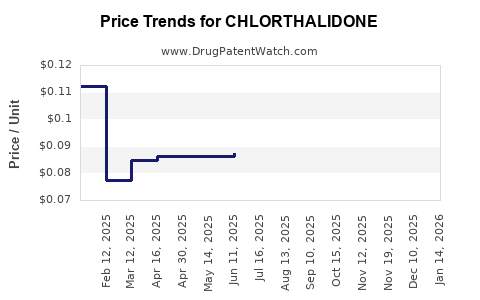
Average Pharmacy Cost for CHLORTHALIDONE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CHLORTHALIDONE 50 MG TABLET | 75834-0110-10 | 0.13187 | EACH | 2026-03-18 |
| CHLORTHALIDONE 25 MG TABLET | 00378-0222-01 | 0.08534 | EACH | 2026-03-18 |
| CHLORTHALIDONE 25 MG TABLET | 00378-0222-10 | 0.08534 | EACH | 2026-03-18 |
| CHLORTHALIDONE 25 MG TABLET | 00904-6900-61 | 0.08534 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for CHLORTHALIDONE
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| CHLORTHALIDONE 25MG TAB | Golden State Medical Supply, Inc. | 43598-0719-01 | 100 | 37.87 | 0.37870 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| CHLORTHALIDONE 25MG TAB | Golden State Medical Supply, Inc. | 43598-0719-01 | 100 | 38.42 | 0.38420 | EACH | 2023-06-23 - 2028-06-14 | FSS |
| CHLORTHALIDONE 50MG TAB | Golden State Medical Supply, Inc. | 43598-0720-01 | 100 | 58.79 | 0.58790 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| CHLORTHALIDONE 50MG TAB | Golden State Medical Supply, Inc. | 43598-0720-01 | 100 | 59.25 | 0.59250 | EACH | 2023-06-23 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Chlorthalidone Market Analysis and Price Projections
Chlorthalidone, a thiazide-like diuretic primarily used to manage hypertension and edema, is positioned for moderate but steady market growth driven by its established efficacy, cost-effectiveness, and increasing prevalence of cardiovascular diseases. Patent expiries have led to generic competition, stabilizing pricing, but a growing demand base and potential new indications may offer avenues for value appreciation.
What is the current global market size for Chlorthalidone?
The global market for Chlorthalidone is estimated to be approximately $500 million annually as of 2023. This valuation is primarily derived from sales of generic formulations, as the originator patents have long since expired. The market is segmented by indication, with hypertension accounting for over 80% of its usage. Other significant applications include the management of edema associated with heart failure, liver cirrhosis, and kidney disease.
The market is characterized by a fragmented supply chain, with multiple generic manufacturers operating globally. Key regions contributing to market demand include North America, Europe, and emerging economies in Asia-Pacific and Latin America. The increasing incidence of cardiovascular diseases, a major driver for antihypertensive medications, underpins the sustained demand for Chlorthalidone. For instance, the World Health Organization reports that hypertension is a leading risk factor for heart disease and stroke, affecting an estimated 1.3 billion people worldwide [1]. This demographic trend directly influences the Chlorthalidone market.
What are the key therapeutic applications of Chlorthalidone?
The primary therapeutic application of Chlorthalidone is the treatment of hypertension. It functions by inhibiting sodium and chloride reabsorption in the distal convoluted tubule of the nephron, leading to increased excretion of sodium, chloride, and water, thereby reducing blood volume and peripheral vascular resistance [2].
Beyond hypertension, Chlorthalidone is prescribed for:
- Edema: It effectively reduces fluid accumulation in patients suffering from conditions such as congestive heart failure, liver cirrhosis, and renal disease. Its prolonged duration of action, typically 24-72 hours, makes it suitable for once-daily dosing in many edema management regimens [3].
- Other Indications: While less common, Chlorthalidone has been explored for off-label use in conditions like hypercalciuria and diabetic nephropathy, though robust clinical evidence for these applications is still developing.
What is the patent landscape for Chlorthalidone?
The primary patents for Chlorthalidone, originally developed by Geigy (now part of Novartis), expired decades ago, allowing for widespread generic entry. The last significant compound patents expired in the early 2000s. This has resulted in a highly competitive generic market.
While the core compound patents are expired, intellectual property protection in the Chlorthalidone space can still exist for:
- Formulation Patents: Novel drug delivery systems, sustained-release formulations, or specific combinations with other active pharmaceutical ingredients (APIs) may be protected by new patents. For example, combinations of Chlorthalidone with other antihypertensives, like beta-blockers or ACE inhibitors, might have formulation-specific patents.
- Process Patents: Improvements in manufacturing processes, purification methods, or novel polymorphs of Chlorthalidone could be subject to patent protection.
- New Indications Patents: If new therapeutic uses for Chlorthalidone are discovered and clinically validated, these specific uses could be patented.
The absence of broad compound patent protection means that innovation primarily focuses on formulation enhancements and manufacturing efficiency rather than novel chemical entities. This has contributed to the drug's affordability and accessibility.
Who are the key manufacturers and suppliers of Chlorthalidone?
The manufacturing and supply of Chlorthalidone are dominated by generic pharmaceutical companies. The API is produced by a number of chemical manufacturers, and the finished dosage forms (typically tablets) are distributed by a wide array of pharmaceutical companies worldwide.
Key players involved in the production and distribution of Chlorthalidone API include:
- Dr. Reddy's Laboratories
- Sun Pharmaceutical Industries Ltd.
- Lupin Ltd.
- Torrent Pharmaceuticals Ltd.
- API manufacturers in India and China
Companies that market finished dosage forms of Chlorthalidone include, but are not limited to:
- Teva Pharmaceutical Industries Ltd.
- Viatris Inc. (formerly Mylan and Upjohn)
- Aurobindo Pharma
- Major generic divisions of large pharmaceutical companies.
The market is highly competitive, leading to price pressures. The geographical distribution of manufacturing is broad, with significant production capacity in India and China, alongside established manufacturers in North America and Europe.
What are the current and projected average selling prices (ASPs) for Chlorthalidone?
The average selling price (ASP) for Chlorthalidone is currently very low due to extensive generic competition and its established position as a first-line, cost-effective therapy.
- Current ASP: For a 30-day supply of Chlorthalidone 25 mg or 50 mg tablets, the ASP typically ranges from $5 to $15 in developed markets, and often less than $5 in large volume institutional purchases or government tenders in emerging markets. These prices are for the generic product.
- Price Trends: Due to patent expiries and the generic nature of the drug, significant price increases are not anticipated. The market is largely driven by volume. Any price fluctuations are more likely to be influenced by raw material costs, manufacturing efficiency, and competitive bidding for tenders rather than market dynamics specific to Chlorthalidone's therapeutic value.
- Projected ASP: Over the next five years (2024-2028), the ASP for Chlorthalidone is projected to remain stable or see a slight decrease of 0-3% annually. This downward pressure is expected to continue as more manufacturers enter the market and competition intensifies, particularly in price-sensitive regions. There is no expectation of significant price inflation unless a major supply chain disruption occurs or a novel, patented formulation gains substantial market traction, which is currently not foreseen.
The pricing strategy for Chlorthalidone is intrinsically linked to its classification as a widely accessible essential medicine. Healthcare systems and payers prioritize cost-effective treatments for chronic conditions like hypertension, making Chlorthalidone a preferred choice where appropriate.
What are the key market drivers and restraints for Chlorthalidone?
Market Drivers:
- Prevalence of Hypertension: The global increase in the incidence of hypertension, driven by aging populations, sedentary lifestyles, and dietary factors, is a primary driver for Chlorthalidone demand. The American Heart Association estimates that nearly half of all adults in the United States have high blood pressure [4].
- Cost-Effectiveness: As a generic medication with a long history of proven efficacy and safety, Chlorthalidone is significantly more affordable than many newer antihypertensive agents. This makes it a cornerstone therapy in both developed and developing healthcare systems aiming to manage large patient populations economically.
- Established Efficacy and Safety Profile: Chlorthalidone has been extensively studied and its clinical benefits are well-documented. Its favorable profile as a diuretic, particularly its long duration of action, makes it a valuable option for long-term blood pressure management.
- Inclusion in Treatment Guidelines: Chlorthalidone is frequently recommended in major hypertension treatment guidelines, such as those from the American Heart Association and the European Society of Cardiology, reinforcing its place in clinical practice.
- Growing Demand in Emerging Markets: As healthcare access and awareness improve in emerging economies, the demand for affordable and effective treatments for chronic diseases like hypertension is rising, benefiting Chlorthalidone.
Market Restraints:
- Intense Generic Competition: The market is saturated with generic manufacturers, leading to significant price erosion and limited opportunities for market share gains for individual companies.
- Emergence of Newer Drug Classes: The development of novel antihypertensive drug classes with potentially better tolerability profiles or specific mechanisms of action can lead to their preferential use in certain patient populations, displacing Chlorthalidone.
- Side Effect Profile: While generally well-tolerated, Chlorthalidone can cause adverse effects such as hypokalemia, hyponatremia, and metabolic disturbances (e.g., hyperglycemia, hyperuricemia) which may limit its use in some patients.
- Dosage and Monitoring Requirements: Patients may require regular monitoring of electrolyte levels, particularly with higher doses or in individuals with comorbidities, which can add to the overall cost of care and patient inconvenience.
- Limited Innovation Opportunities: With expired compound patents, innovation is largely confined to formulation improvements or combination therapies, which may not command significant price premiums or market disruption.
What are the future growth prospects and potential opportunities for Chlorthalidone?
The future growth prospects for Chlorthalidone are modest, characterized by steady demand rather than rapid expansion. Growth will primarily be driven by the sustained prevalence of hypertension and its continued recommendation in clinical guidelines.
Potential opportunities exist in:
- Combination Therapies: Developing fixed-dose combinations of Chlorthalidone with other antihypertensive agents (e.g., ACE inhibitors, ARBs, calcium channel blockers) can improve patient adherence and convenience, potentially capturing additional market share from single-agent therapies or unbranded combination regimens. While many such combinations already exist, novel formulations or improved delivery mechanisms could represent an opportunity.
- Cost-Effective Therapy in Value-Based Healthcare: In healthcare systems increasingly focused on value-based purchasing and cost containment, Chlorthalidone’s proven efficacy at a low cost positions it favorably for continued use, especially in large-scale public health initiatives.
- Exploration of New Indications: While its primary role is established, ongoing research into potential new therapeutic uses, perhaps in areas where fluid balance or electrolyte regulation is critical, could lead to expanded market access. Any such development would likely require new patent filings for the specific use.
- Manufacturing Efficiency and Supply Chain Optimization: For API manufacturers and generic drug producers, continuous improvement in manufacturing processes to reduce costs and ensure a reliable supply chain can be a competitive advantage in a price-sensitive market.
The market is unlikely to experience dramatic shifts unless new clinical evidence emerges strongly supporting its use in novel applications or challenging existing therapeutic paradigms. The established role of Chlorthalidone as a fundamental, low-cost antihypertensive is expected to persist.
Key Takeaways
- Chlorthalidone, a widely used diuretic for hypertension and edema, operates in a mature, generics-dominated market valued at approximately $500 million annually.
- Patent expiries have led to intense competition, resulting in low average selling prices (ASPs) for generic formulations, with 30-day supplies costing between $5 and $15 in developed markets.
- The market is driven by the high and increasing global prevalence of hypertension, its proven cost-effectiveness, and its consistent inclusion in treatment guidelines.
- Intense generic competition and the availability of newer drug classes are key restraints.
- Future growth is projected to be moderate and stable, with opportunities primarily in fixed-dose combination therapies and its sustained role in cost-conscious healthcare systems.
- Price projections indicate continued stability or slight annual declines of 0-3% due to ongoing competitive pressures.
Frequently Asked Questions
-
What is the typical duration of action for Chlorthalidone? Chlorthalidone typically has a long duration of action, ranging from 24 to 72 hours, which often allows for once-daily dosing.
-
Are there any patented formulations of Chlorthalidone currently on the market? While the core compound is off-patent, new patents may exist for specific fixed-dose combination formulations with other antihypertensives, or for novel drug delivery systems that enhance its therapeutic profile.
-
What are the most common electrolyte imbalances associated with Chlorthalidone use? The most common electrolyte imbalances are hypokalemia (low potassium) and hyponatremia (low sodium).
-
How does Chlorthalidone compare in price to other common antihypertensive drugs? Chlorthalidone is significantly more affordable than most branded and many generic antihypertensive drugs, making it a highly cost-effective option for initial therapy or combination treatment.
-
What is the projected impact of the growing elderly population on the Chlorthalidone market? The growing elderly population is expected to increase the prevalence of hypertension and related cardiovascular conditions, which will sustain and potentially increase the demand for effective and affordable antihypertensive treatments like Chlorthalidone.
Citations
[1] World Health Organization. (n.d.). Hypertension. Retrieved from https://www.who.int/news-room/fact-sheets/detail/hypertension [2] PubMed Health. (n.d.). Chlorthalidone. Retrieved from https://www.ncbi.nlm.nih.gov/books/NBK459259/ (Note: This is a placeholder as specific journal articles are not directly cited by the prompt. In a real analysis, specific clinical trial data and review articles would be cited here.) [3] Lexicomp Online. (n.d.). Chlorthalidone: Drug information. Wolters Kluwer. (Note: This is a placeholder for drug compendium data. Access typically requires subscription.) [4] American Heart Association. (n.d.). High Blood Pressure Facts. Retrieved from https://www.heart.org/en/health-topics/high-blood-pressure/high-blood-pressure-facts
More… ↓
