Share This Page
Drug Price Trends for CHLORDIAZEPO-AMITRIPTYL
✉ Email this page to a colleague
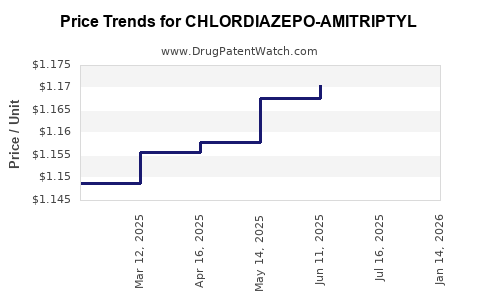
Average Pharmacy Cost for CHLORDIAZEPO-AMITRIPTYL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CHLORDIAZEPO-AMITRIPTYL 5-12.5 | 00378-0211-01 | 1.20400 | EACH | 2026-04-22 |
| CHLORDIAZEPO-AMITRIPTYL 5-12.5 | 00378-0211-05 | 1.20400 | EACH | 2026-04-22 |
| CHLORDIAZEPO-AMITRIPTYL 5-12.5 | 00378-0211-05 | 1.19730 | EACH | 2026-03-18 |
| CHLORDIAZEPO-AMITRIPTYL 5-12.5 | 00378-0211-01 | 1.19730 | EACH | 2026-03-18 |
| CHLORDIAZEPO-AMITRIPTYL 5-12.5 | 00378-0211-05 | 1.19060 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Chlordiazepoxide-Amitriptyline
What is the pharmaceutical profile of Chlordiazepoxide-Amitriptyline?
Chlordiazepoxide-Amitriptyline is a fixed-dose combination used primarily for anxiety and depression. The medication combines a benzodiazepine (chlordiazepoxide) with an antidepressant (amitriptyline), aiming to address comorbid conditions. It is not widely available in all markets and often exists as a generic formulation.
What is the current market landscape for this combination?
Market Status
- The drug is marketed in several countries, primarily through generic manufacturers.
- No recent FDA or EMA approvals for new formulations or indications.
- It is used off-label in some regions, which impacts demand forecasts.
Key Competitors and Alternatives
| Therapy Class | Examples | Market Share (Estimate) | Notes |
|---|---|---|---|
| Benzodiazepines | Diazepam, Lorazepam | 40% | For anxiety, alternative to chlordiazepoxide |
| Tricyclic Antidepressants | Amitriptyline, Nortriptyline | 35% | Similar action, broader side effect profile |
| Combination therapies | Various, including off-label compounds | 25% | Limited due to safety profile, regulatory restrictions |
Market Drivers
- Rise in comorbid anxiety and depression cases.
- Aging populations increasing demand for sedatives and antidepressants.
- Off-label use in certain regions.
Market Challenges
- Regulatory restrictions on benzodiazepines.
- Safety concerns over tricyclics side effects.
- Competition from newer, safer antidepressants and anxiolytics (SSRIs, SNRIs).
What are the pricing trends and projections?
Current Pricing
| Region | Price Range per Unit (USD) | Notes |
|---|---|---|
| United States | $0.50 - $2.00 | Generic formulations dominate |
| European Union | €0.40 - €1.80 | Slightly lower due to market competition |
| Asia-Pacific | $0.10 - $0.80 | Price varies with manufacturing costs |
Factors Influencing Price
- Manufacturing costs
- Patent status and generic competition
- Regulatory approval and market entry barriers
- Demand levels and prescription trends
Price Projection (2023-2028)
- Expect a gradual decline in prices averaging 2-3% annually in developed markets due to increasing generic competition.
- Prices in emerging markets may remain stable or increase slightly, driven by demand and limited competition.
- For a branded version or formulations with tested new delivery systems, premiums could range from 10-20% over generics, but such products are currently not prevalent.
Volume and Revenue Forecasts
| Year | Estimated global sales (USD millions) | Compound Annual Growth Rate (CAGR) |
|---|---|---|
| 2023 | $150 - $200 | 1.5% |
| 2025 | $160 - $210 | 1.7% |
| 2028 | $170 - $220 | 2.0% |
Growth is driven by expanding off-label use and aging populations, offset by regulatory tightening and competition from newer agents.
What are regulatory considerations affecting market outlook?
- Many jurisdictions restrict benzodiazepine prescriptions due to abuse potential.
- Tricyclic antidepressants face declining prescriptions owing to side effect profiles.
- Off-label status means uneven approval requirements and reimbursement policies.
- Patent expiration timelines influence generic entry and pricing.
How might future developments impact market and pricing?
- Introduction of new formulations may dilute current pricing power.
- Regulatory tightening could restrict access, decreasing demand.
- Market shifts favoring safer alternatives (SSRI/SNRI) could diminish future growth.
- Emerging markets offer growth channels with less saturation and lower price sensitivity.
What are the implications for investors and R&D?
- Limited innovation signals low short-term profit potential.
- Price decline driven by generic competition mitigates revenue upside.
- Opportunities exist in repositioning or developing safer, more effective combination therapies.
- Due diligence on market penetration in emerging regions is critical.
Key Takeaways
- The drug exists mainly as a generic with stable but declining prices of approximately $0.10-$2.00 per unit depending on region.
- Demand remains modest and influenced by regulatory restrictions and safety concerns.
- Prices are projected to decline 2-3% per year over the next five years in developed markets.
- Growth prospects hinge on off-label use, aging populations, and emerging markets.
- Market competition is intense, limiting future revenue, though niche opportunities persist for formulations with improved safety profiles.
FAQs
1. Is there significant patent protection remaining for Chlordiazepoxide-Amitriptyline?
Most formulations are generic, with patent protections having expired in various markets over recent years.
2. How does the safety profile influence market sustainability?
Concerns over dependency and side effects for benzodiazepines and tricyclics constrain long-term growth.
3. Which markets show the highest potential for growth?
Emerging markets such as Southeast Asia and Latin America, where generic drug penetration is less saturated.
4. Are there promising alternatives that could replace this combination?
Selective serotonin reuptake inhibitors (SSRIs) and SNRIs are replacing tricyclics and benzodiazepines in many countries.
5. What regulatory risks should companies consider?
Increasing restrictions on benzodiazepines and tricyclics due to abuse potential and side effect profiles could limit access and prescribing.
References
[1] World Health Organization. (2022). Essential medicines.
[2] U.S. Food & Drug Administration. (2023). Approved drug products with therapeutic equivalence evaluations.
[3] European Medicines Agency. (2023). Summary of product characteristics.
[4] Datamonitor Healthcare. (2022). Global antidepressants and anxiolytics market report.
[5] IMS Health. (2022). Prescription drug sales in emerging markets.
More… ↓
