Share This Page
Drug Price Trends for CHILD CETIRIZINE HCL
✉ Email this page to a colleague
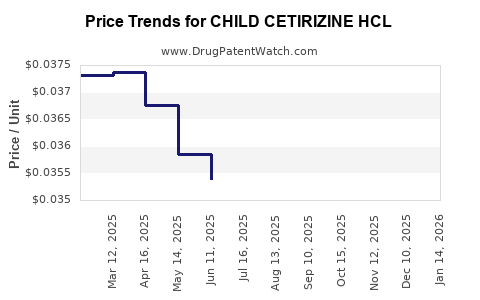
Average Pharmacy Cost for CHILD CETIRIZINE HCL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CHILD CETIRIZINE HCL 1 MG/ML | 70752-0104-06 | 0.03744 | ML | 2026-03-18 |
| CHILD CETIRIZINE HCL 1 MG/ML | 00904-6765-20 | 0.03744 | ML | 2026-03-18 |
| CHILD CETIRIZINE HCL 1 MG/ML | 70752-0104-06 | 0.03680 | ML | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
CHILD CETIRIZINE HCL Market Analysis and Financial Projection
What is Child Cetirizine HCl and How Is It Positioned in the Market?
Child Cetirizine HCl is an antihistamine used primarily for allergy relief in pediatric populations. It is marketed under various brand names including Zyrtec, Aller-Tec, and others. The drug functions by blocking peripheral H1 histamine receptors, reducing allergy symptoms such as sneezing, runny nose, and itching. The FDA-approved formulations for children typically include syrup, tablets, and chewables targeting age groups from 2 years onward.
The global antihistamine market has seen consistent growth driven by increasing allergy prevalence. In 2022, the global antihistamine market was valued at approximately USD 6.4 billion, with a compound annual growth rate (CAGR) around 4.5%. The pediatric segment accounts for roughly 30-40% of total antihistamine sales.
How is the Market for Child Cetirizine HCl Evolving?
Market Size and Growth
- Current Market Value: Estimated at USD 1.5 billion globally for pediatric antihistamines.
- Growth Drivers: Rising prevalence of allergic rhinitis and urticaria among children, increased awareness, and improved diagnosis.
- Regional Trends:
- North America: Largest market, USD 650 million in 2022, driven by high awareness and over-the-counter (OTC) sales.
- Europe: USD 500 million, benefiting from stringent regulatory frameworks and high allergy prevalence.
- Asia-Pacific: Fastest growth, CAGR over 6%, due to increasing urbanization and healthcare access.
Competitive Landscape
- Major Players: Johnson & Johnson (Zyrtec), GlaxoSmithKline (Realmence), Teva, Mylan, and local generic manufacturers.
- Market Share Concentration: Top three brands hold roughly 65% of the market.
- Generic Incursion: With patent expirations, generics now account for over 70% of sales in many regions.
What Are Price Dynamics and Trends?
Brand vs. Generic Pricing
| Product Type | Price Range (USD) per bottle/tablet | Market Share (%) |
|---|---|---|
| Brand-name (e.g., Zyrtec) | 12-18 (per 60 ml syrup) | 30-35 |
| Generic formulations | 5-10 (per 60 ml syrup) | 65-70 |
Influencing Factors
- Regulatory Changes: Slight price drops post-patent expirations in 2018 increased generic competition.
- Retail Dynamics: OTC sales through pharmacies and supermarkets push prices downward.
- Private Label and Store Brands: Aggressively priced, capturing 15-20% of OTC market share.
- Pricing Trends: Decline in average prices for generics over the past three years by approximately 10-15%.
Future Price Projections
- 3-Year Outlook (2023-2026):
- Slight further decrease in generic prices (~5-8%) due to increased market saturation.
- Brand-name prices likely stable or marginally increase (~2%) driven by inflation and formulation improvements.
- Price competition will intensify in emerging markets, accelerating generic price declines.
Regulatory and Market Access Considerations
- Approval Pathway: In many countries, pediatric formulations are OTC, reducing entry barriers for generics.
- Reformulations and Fixed-Dose Combinations: Expected to drive slight price increases in some markets due to added convenience.
- Reimbursement Policies: Limited impact, as most pediatric antihistamines are purchased OTC without reimbursement.
Key Takeaways
- The global pediatric antihistamine market is growing at approximately 4.5% CAGR, with Child Cetirizine HCl positioned as a leading product.
- Market value exceeds USD 1.5 billion, with North America leading, followed by Europe and the Asia-Pacific.
- Patent expirations and increasing generic availability have driven prices down, with generics now representing over 70% of sales.
- Prices for generics will likely decline by 5-8% over the next three years; brand-name prices are expected to remain stable or slightly increase.
- Barriers to market entry are low due to OTC availability and regulatory pathways, fostering price competition.
FAQs
1. How does patent expiration impact pricing for Child Cetirizine HCl?
Patents typically last 20 years, and expiration allows generic manufacturers to enter the market. This introduction increases supply, presses prices downward, and reduces brand dominance.
2. What regions exhibit the highest growth potential for Child Cetirizine HCl?
The Asia-Pacific region displays the fastest growth, driven by rising allergy prevalence and expanding healthcare infrastructure.
3. How do OTC regulations influence the market for pediatric antihistamines?
OTC status broadens access, encourages retail competition, and suppresses prices due to increased availability and consumer buying options.
4. Is there room for premium-priced formulations?
Yes. Reformulations offering added convenience (e.g., taste-masked syrups, fast-dissolving tablets) can command higher prices in developed markets.
5. What risks could influence future market prices?
Regulatory tightening, market saturation, aggressive pricing by generics, and shifts in healthcare policies could affect future pricing trajectories.
References:
- MarketWatch. "Antihistamines Market Size, Share & Trends." 2022.
- GlobalData. "Pediatric Allergy Market Overview." 2023.
- U.S. Food and Drug Administration. "Pediatric Formulation Regulations." 2022.
- IQVIA. "Pharma Market Data," 2022.
- Statista. "Regional Allergy Prevalence and Market Insights," 2023.
More… ↓
