Drug Price Trends for CHILD ACETAMINOPHEN
✉ Email this page to a colleague
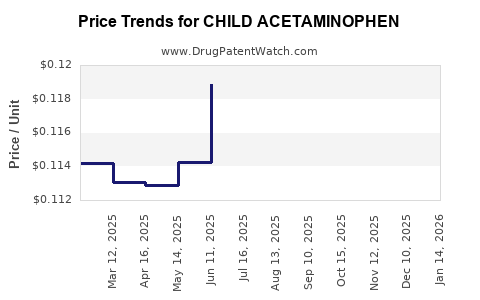
Average Pharmacy Cost for CHILD ACETAMINOPHEN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CHILD ACETAMINOPHEN 160 MG | 70000-0310-01 | 0.11990 | EACH | 2026-03-18 |
| CHILD ACETAMINOPHEN 160 MG | 70000-0309-01 | 0.11990 | EACH | 2026-03-18 |
| CHILD ACETAMINOPHEN 160 MG | 70000-0310-01 | 0.12056 | EACH | 2026-02-18 |
| CHILD ACETAMINOPHEN 160 MG | 70000-0309-01 | 0.12056 | EACH | 2026-02-18 |
| CHILD ACETAMINOPHEN 160 MG | 70000-0310-01 | 0.12007 | EACH | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Child Acetaminophen Market Analysis and Price Projections
Market Size and Growth Dynamics
Global pediatric acetaminophen (paracetamol) market is valued at approximately $2.8 billion in 2022. It is projected to reach $4.5 billion by 2030, expanding at a compound annual growth rate (CAGR) of 6.8% (2022-2030). Growth drivers include the increasing prevalence of fever and pain-related conditions among children, rising awareness of pediatric health, and expanding penetration in emerging markets.
Major geographic segments include North America, Europe, Asia-Pacific, and Latin America. North America accounts for around 45% of sales, driven by high healthcare expenditure and product availability. The Asia-Pacific region is expected to register the highest CAGR due to rising healthcare infrastructure and population growth.
Market Drivers and Challenges
Drivers:
- Rising pediatric population
- Increased use for fever relief and pain management
- Growing acceptance of OTC medications for children
- Regulatory approvals and formulations designed for children
Challenges:
- Safety concerns regarding overdose or misuse
- Regulatory scrutiny over formulations and labeling
- Competition from alternative over-the-counter and prescription medications
Market Segmentation
| Segment | Characteristics | Market Share (2022) | Growth Rate |
|---|---|---|---|
| Formulation | Liquid suspension, chewables, suppositories | Liquid suspension (60%) | CAGR 7.1% in liquids |
| Distribution Channel | Pharmacy, hospital, online retail | Pharmacy (70%) | Growth in online channels (~12%) |
| Age Group | Under 2 years, 2-5 years, 6-12 years | Under 2 years (25%) | Increasing use in infants |
Pricing Trends and Projections
Current Price Range (2023):
- Retail liquid suspension: $3.50 to $5.00 per 100 ml bottle
- Chewables: $4.00 to $6.50 per pack (30-50 tablets)
Price Factors:
- Production costs, including raw materials (acetaminophen active ingredient)
- Regulatory compliance expenditures
- Demand-supply dynamics; raw material prices have been stable but are sensitive to global supply chain fluctuations
- Competition levels, with generic brands pricing 10-20% lower than brand-name equivalents
Projection for 2025-2030:
- Prices for liquid formulations are expected to decrease by approximately 2% annually due to economies of scale and increased generic competition
- Premium pediatric formulations, such as organic or allergen-free variants, may command prices 15-20% higher
- Overall, retail prices are projected to decline to $3.00-$4.50 per 100 ml by 2030, assuming moderate inflation and steady demand
Competitive Landscape
Key Players:
- Johnson & Johnson (Children’s Tylenol)
- Pfizer (Infants’ Acetaminophen)
- Perrigo (Store Brands)
- Sanofi (Glenmark formulations)
Market Share (2022):
- Johnson & Johnson: 35%
- Pfizer: 20%
- Perrigo: 15%
- Others (including generics): 30%
The market is dominated by generics, which account for over 60% of sales, undercutting brand-name products on price.
Regulatory Environment and Future Trends
Regulatory authorities, including the US FDA and European Medicines Agency, tighten labeling accuracy, dosage standards, and safety warnings. New formulations are under review to improve tolerability and efficacy.
Increased adoption of digital health apps and telepharmacy enhances access. Development of precise dosing devices aims to reduce overdose risk and could influence product pricing structures.
Summary of Key Data
| Metric | 2022 Info | 2030 Projection |
|---|---|---|
| Global Pediatric Acetaminophen Market | $2.8 billion | $4.5 billion |
| CAGR | 6.8% | 6.8% |
| Retail Price (per 100 ml) | $3.50 - $5.00 | $3.00 - $4.50 |
| Market Share (Leading Brands) | Johnson & Johnson (35%) | N/A (market consolidation likely persists) |
Key Takeaways
- The pediatric acetaminophen market is expected to grow steadily, driven by increased demand in emerging markets and a stable regulatory environment.
- Pricing will trend downward marginally, with generic brands pressuring margins of branded products.
- Price variations will depend on formulation type, branding, and distribution channels.
- Regulatory focus on safety and precise dosing will shape product development and may affect pricing strategies.
- Market competition is intense, with leading brands holding significant shares but facing strong generic competition.
FAQs
1. What factors influence pediatric acetaminophen pricing?
Raw material costs, regulatory compliance expenses, manufacturing scale, competition, and patent status influence pricing.
2. How will emerging markets impact global prices?
Increasing demand and local manufacturing will exert downward pressure on prices, especially in countries with high birth rates.
3. What is the main driver of market growth?
The growing pediatric population and widespread use for fever and pain relief.
4. Are there safety concerns affecting market development?
Yes. Overdose potential and regulatory scrutiny lead to stricter dosing guidelines, influencing product formulations and labeling.
5. How is competition shaping pricing strategies?
Mainly through generic entrants offering lower prices, leading branded products to focus on formulation differentiation and safety features.
Citations
More… ↓
