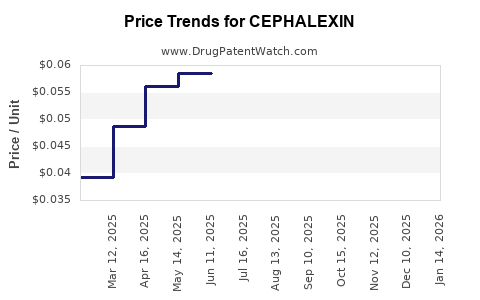
Last updated: February 20, 2026
Cephalexin, an oral antibiotic from the first-generation cephalosporin class, treats skin, bone, and respiratory infections. The drug is available as a generic and under brand names such as Keflex. The market’s dynamics, including manufacturing, competition, and regulatory factors, influence pricing and revenue projections.
Market Size and Growth
Global Demand:
As of 2022, the global cephalexin market was valued at approximately USD 1.2 billion. The compound annual growth rate (CAGR) from 2023 to 2030 is projected at 3.2%. Growth drivers include increasing bacterial infections, rising antibiotic prescriptions, and a steady approval rate for generic versions.
| Regional Breakdown: |
Region |
Market Share (2022) |
CAGR (2023–2030) |
Key Factors |
| North America |
40% |
3.0% |
High prescription rates; established healthcare system |
| Europe |
25% |
3.5% |
Aging population; antibiotic stewardship programs |
| Asia-Pacific |
20% |
4.0% |
Growing healthcare infrastructure; large population |
| Rest of World |
15% |
2.5% |
Variable access; emerging markets |
Market Drivers and Challenges
Drivers
- Rising bacterial infections: Urbanization and antibiotic use drive infections like skin, respiratory, and urinary tract infections.
- Generic availability: Multiple manufacturers produce cephalexin, lowering prices and increasing accessibility.
- Regulatory approvals: Growing approvals for formulations and indications expand market penetration.
Challenges
- Antimicrobial resistance (AMR): Increasing resistance reduces efficacy, prompting shifts to newer antibiotics.
- Regulatory scrutiny: Evolving policies on antibiotic usage and stewardship programs may limit prescriptions.
- Competition: Presence of other antibiotics like amoxicillin and doxycycline limits market growth.
Pricing Trends
Current Prices (2023):
- A typical 500 mg capsule costs USD 0.10–0.25 retail.
- Wholesale prices tend to be approximately 20–30% lower.
- Brand-name Keflex remains priced approximately 25–30% higher than generics.
Price Dynamics:
- Generic cephalexin prices declined by an average of 12% annually from 2015 to 2022.
- The increase in competition has stabilized prices despite inflation.
- Geographic differences influence price: North America and Europe maintain higher prices due to regulation and healthcare reimbursement models.
Price Projection (2023–2030)
| Year |
Estimated Price (per 500 mg capsule) |
Notes |
| 2023 |
USD 0.10 – 0.25 |
Current market range |
| 2025 |
USD 0.09 – 0.23 |
Slight decline due to increased generics penetration |
| 2030 |
USD 0.08 – 0.20 |
Continued price erosion, stabilization with new generics |
Projection assumptions include increased generic market share, stable regulatory landscape, and limited resistance issues in the short term.
Competitive Landscape
Major Manufacturers
- Lupin
- Sandoz
- Teva Pharmaceuticals
- Mylan
Patent Status
- No patents for cephalexin formulations exist since 2015.
- Market dominated by generics, which collectively account for ~90% of sales.
Therapeutic Alternatives
- Amoxicillin
- Doxycycline
- Cefadroxil
These alternatives may influence future demand and pricing stability.
Regulatory Environment
- Generic drug approvals increasingly emphasis on bioequivalence.
- In the U.S., the FDA has approved multiple generic cephalexin products since 2018.
- In Europe, EMA approvals follow similarly, supporting price competition.
Summary of Outlook
| Factor |
Impact on Market & Pricing |
| Increased generic competition |
Continues downward pressure on unit prices |
| Rising AMR concerns |
Potential for prescription restrictions, limiting sales |
| Regional growth in APAC |
Expanding markets with growing demand, potentially higher prices |
| Regulatory developments |
May increase costs or delay entry of new formulations |
Key Takeaways
- The global cephalexin market is expected to grow modestly, driven by increasing bacterial infections and generic sales.
- Prices are likely to decrease gradually, with a projection of USD 0.08–0.20 per 500 mg capsule by 2030.
- Competition, AMR, and regulatory factors will significantly influence revenue and market share.
- The majority of sales in mature markets are generics, contributing to stable and declining prices.
- Emerging markets, especially in Asia-Pacific, present growth opportunities with potentially higher pricing initially.
FAQs
1. What factors most influence cephalexin pricing?
Market competition, generic availability, regional healthcare regulations, and AMR trends.
2. How does resistance affect future demand?
Rising resistance could lower prescriptions, reducing demand and stabilizing or decreasing prices.
3. What is the main competitor for cephalexin?
Amoxicillin, doxycycline, and cefadroxil, which are often prescribed as alternatives.
4. Will new formulations affect the market?
New formulations could command higher prices but require regulatory approval, which may delay market entry.
5. What regions are most likely to see pricing stability or increases?
Emerging markets like Asia-Pacific, where demand is expanding, may see stable or higher initial prices due to less saturation.
References
[1] MarketWatch. (2023). Global cephalexin market size, share, growth, trends, and forecast.
[2] Grand View Research. (2023). Antibiotics Market Size, Share & Trends Analysis Report.
[3] U.S. Food and Drug Administration. (2022). Approved drug products with therapeutic equivalence evaluations.
[4] European Medicines Agency. (2022). Mandatory requirements for generic medicines.