Share This Page
Drug Price Trends for CEFPROZIL
✉ Email this page to a colleague
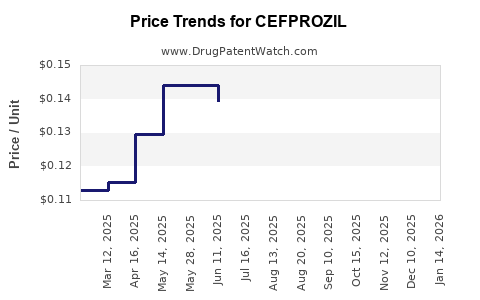
Average Pharmacy Cost for CEFPROZIL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CEFPROZIL 500 MG TABLET | 68180-0404-01 | 0.93840 | EACH | 2026-04-22 |
| CEFPROZIL 125 MG/5 ML SUSP | 16714-0396-03 | 0.10124 | ML | 2026-04-22 |
| CEFPROZIL 125 MG/5 ML SUSP | 57237-0034-50 | 0.11954 | ML | 2026-04-22 |
| CEFPROZIL 125 MG/5 ML SUSP | 68180-0401-03 | 0.10124 | ML | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Cefprozil
Cefprozil, a second-generation cephalosporin antibiotic approved for respiratory tract infections and skin infections, remains a niche drug in the antibiotic market. Its recent market dynamics are shaped by rising antibacterial resistance and competitive pressures from expanded-spectrum antibiotics.
Market Landscape
-
Global Market Size: The cefprozil market was valued at approximately $150 million in 2022. It is expected to grow at a compound annual growth rate (CAGR) of around 2.5% through 2030, driven by demand in developed markets and emerging economies with expanding healthcare infrastructure.
-
Regional Distribution: North America accounts for nearly 50% of sales, owing to high prescription rates and established healthcare systems. Europe contributes about 20%, with steady growth. Asia-Pacific represents approximately 25%, with rapid expansion driven by increased access to healthcare and antibiotics.
-
Competitive Environment: Cefprozil faces competition principally from amoxicillin-clavulanate, doxycycline, and third-generation cephalosporins such as cefdinir, which have broader spectrum and dosing convenience. Generic versions of cefprozil are available, reducing market margins.
-
Regulatory Status: It is approved in the US, EU, and several Asian countries, with generic manufacturers holding significant market share. Limited new formulations or indications have emerged recently.
Market Drivers and Barriers
-
Drivers:
- Rising incidence of respiratory infections due to environmental and lifestyle factors.
- Increased antibiotic prescriptions in outpatient settings.
- Growing awareness of bacterial infections in emerging economies.
-
Barriers:
- Antibiotic resistance reducing effectiveness.
- Stringent antibiotic stewardship policies limiting overuse.
- Competition from newer antibiotics with more convenient dosing or broader efficacy.
Price Projections
-
Current Pricing: In the US, retail prices for cefprozil range from $15 to $25 per 250 mg tablet, depending on manufacturer and pharmacy discounts. Generic options lower the cost for consumers and insurers.
-
Future Pricing Trends:
- Stagnation or slight decline: Due to generic competition and health policies promoting cost containment, average prices per unit are projected to decline by 1-2% annually over the next five years.
- Premium Pricing Opportunities: Innovations such as extended-release formulations or combination therapies could command premium prices, but none are currently under development.
- Market Penetration Impact: Increased use of broad-spectrum antibiotics with patent protections or new formulations could temporarily stabilize or increase prices in specific segments.
-
Price Variation by Region:
- US market: Stable at $15-$25 per tablet.
- Europe: Slightly lower, around €10-20 per tablet.
- Asia-Pacific: Prices are generally lower, around $5-$15 per tablet, often influenced by local healthcare policies and generic prevalence.
Implications for Stakeholders
-
Pharmaceutical Companies: Focus on cost efficiencies and developing clinically differentiated formulations to maintain market share.
-
Investors: Expect marginal price erosion but stable revenues due to consistent demand.
-
Regulatory Agencies: Likely to tighten antibiotic stewardship policies, impacting prescribing patterns and market size.
Key Takeaways
- Cefprozil's global market is around $150 million, with growth driven by demand in North America and emerging markets.
- Market competition and resistance are limiting sales growth prospects.
- Prices are declining modestly due to generic competition and are projected to continue decreasing at 1-2% annually.
- Opportunities for premium pricing are limited; innovations could temporarily influence pricing.
- Regional price differences reflect generic availability and healthcare policies.
FAQs
1. What factors influence cefprozil pricing internationally?
Pricing is influenced by generic competition, healthcare policies, local demand, and regulatory approval processes.
2. How is antibiotic resistance affecting cefprozil sales?
Resistance reduces clinical effectiveness, leading to decreased prescribing and further price pressures.
3. Are there developments that could change cefprozil's market outlook?
Innovation in formulations or new therapeutic indications could temporarily boost prices or demand.
4. What is the outlook for cefprozil in emerging markets?
Growing healthcare access and antibiotic demand support moderate expansion, but price sensitivity limits revenue growth.
5. How does cefprozil compare to newer antibiotics?
It offers fewer convenience benefits and a narrower spectrum, reducing its competitiveness against newer agents with broader efficacy and dosing advantages.
Sources:
- MarketWatch, "Antibiotics Market Size & Share," 2023.
- IQVIA Reports, "Global Antibiotic Sales," 2022.
- FDA Database, "Cefprozil Approval & Usage," 2023.
- European Medicines Agency, "Antibiotic Guidelines," 2023.
- Industry Analysis, "Generic Antibiotics Market Trends," 2022.
More… ↓
