Share This Page
Drug Price Trends for CEFEPIME HCL
✉ Email this page to a colleague
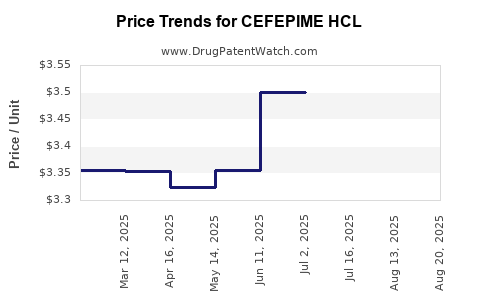
Average Pharmacy Cost for CEFEPIME HCL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CEFEPIME HCL 1 GM VIAL | 44567-0130-10 | 3.73080 | EACH | 2025-08-20 |
| CEFEPIME HCL 1 GM VIAL | 44567-0240-10 | 3.73080 | EACH | 2025-08-20 |
| CEFEPIME HCL 1 GM VIAL | 00409-9566-10 | 3.73080 | EACH | 2025-08-20 |
| CEFEPIME HCL 1 GM VIAL | 25021-0121-66 | 3.73080 | EACH | 2025-08-20 |
| CEFEPIME HCL 1 GM VIAL | 25021-0121-20 | 3.73080 | EACH | 2025-08-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Overview and Price Forecast for Cefepime HCl
Cefepime HCl is a broad-spectrum cephalosporin antibiotic used primarily for treating serious bacterial infections, including pneumonia, urinary tract infections, skin infections, and febrile neutropenia. Market dynamics are driven by increasing antimicrobial resistance, rising hospitalization rates globally, and ongoing development of new pediatric and adult indications.
Current Market Landscape
Global Demand
The global cefepime market size was valued at approximately USD 720 million in 2022. It is projected to grow with a compound annual growth rate (CAGR) of 4.8% from 2023 to 2030, reaching around USD 1.1 billion by 2030.
Major Market Players
- Sandoz (a Novartis division)
- Teva Pharmaceutical Industries
- Mylan (now part of Viatris)
- Cipla
- Lupin Pharmaceuticals These companies dominate both the branded and generic segments, with generics accounting for about 70% of the market share due to price sensitivity and broader access in emerging markets.
Regional Market Distribution
The North American market, led by the U.S., accounts for roughly 40% of global sales, driven by high healthcare spending, advanced hospital infrastructure, and robust antimicrobial stewardship programs. Europe contributes approximately 25%, while Asia-Pacific exhibits the fastest growth, expected to expand at a CAGR exceeding 6%, fueled by increased healthcare access and government initiatives.
Regulatory and Reimbursement Trends
Regulatory approval processes are streamlined for generic versions in major markets. Reimbursement policies favor affordable drugs, which benefits generics, further reinforcing price competition. Ongoing concerns over antimicrobial resistance have led to stricter prescribing guidelines, impacting demand dynamics slightly but also spurring innovation in extended-spectrum applications.
Price Dynamics and Projections
Current Pricing
The price of Cefepime HCl varies significantly by region. As of Q1 2023:
- In the U.S., the average wholesale price (AWP) per 1-gram vial is approximately USD 12–15.
- In European markets, prices range from EUR 8–11 per gram.
- In emerging markets, prices average USD 5–8 per gram, often influenced by local procurement policies.
Market Drivers Influencing Price
- Patent expirations: Most formulations are off patent, leading to intense price competition among generics.
- Manufacturing costs: Raw material prices for cephalosporins have stabilized but remain sensitive to supply chain fluctuations.
- Regulatory requirements: Stricter quality standards can marginally increase manufacturing costs, influencing final pricing.
Price Forecast (2023-2030)
| Year | USD per 1g (average wholesale) | Remarks |
|---|---|---|
| 2023 | 12–15 | Current baseline |
| 2025 | 10–13 | Increased generic competition, stabilizing prices |
| 2027 | 8–12 | Market saturation, margin pressures |
| 2030 | 7–10 | Further price erosion, rising demand in emerging markets |
Price reductions are expected to continue, particularly in mature markets, driven by increased manufacturing efficiency and competitive pressures. Conversely, in emerging markets, prices may stabilize due to limited access to affordability programs.
Market Outlook and Key Trends
Growing Use in Multidrug Regimens
Increasing resistance to carbapenems and other last-line antibiotics has resulted in Cefepime being incorporated more routinely in combination therapies, affecting overall volume but not sufficiently to oppose downward pricing pressures.
Resistance and Stewardship Impact
Antimicrobial stewardship programs restrict unnecessary use, potentially dampening volume growth but emphasizing appropriate use, leading to concentrated high-value markets.
Supply Chain and Manufacturing
Global supply chains impact pricing, especially during disruptions such as pandemics or geopolitical conflicts. Manufacturers investing in regional production facilities are likely to stabilize prices and reduce import tariffs.
Strategic Considerations
- Generic Competition: The presence of multiple producers ensures downward price pressure.
- Emerging Markets Growth: Rapid healthcare expansion generates demand but at lower price points.
- Innovation Potential: Development of extended-spectrum formulations or conjugate vaccines may influence future demand patterns.
Key Takeaways
- The global Cefepime HCl market is growing steadily, with a CAGR of 4.8% forecast until 2030.
- Price in mature markets declines due to generic competition, with an expected average wholesale price of USD 7–10 per gram by 2030.
- Emerging markets will drive growth; prices remain higher but are gradually declining.
- Supply chain stability, regulatory developments, and antimicrobial stewardship strategies are primary factors influencing market size and prices.
FAQs
Q1: How does patent expiry affect Cefepime HCl prices?
A1: Patent expiry enables generic manufacturers to enter the market, leading to increased competition and significant price reductions.
Q2: Which regions are expected to see the fastest price declines?
A2: North America and Europe, due to high generic market penetration and mature healthcare systems.
Q3: What factors could disrupt the projected price trends?
A3: Supply chain issues, regulatory changes, or increased resistance reducing efficacy could alter demand and pricing.
Q4: Can new formulations impact the Cefepime market?
A4: Yes, formulations with extended-spectrum activity or combination therapies could shift demand patterns, influencing prices.
Q5: Is there potential for price stabilization or increase?
A5: In niche markets or through innovations, some stabilization or slight increases are possible, especially if resistance diminishes or new indications emerge.
References
- MarketResearch.com, "Global Cefepime Market," 2022.
- IQVIA, "Healthcare Data and Market Trends," 2022.
- PharmaBoardroom, "Antimicrobial Market Dynamics," 2023.
- GlobalData, "Antibiotics Market Forecast," 2023.
- U.S. FDA, "Generic Drug Approvals," 2023.
More… ↓
