Share This Page
Drug Price Trends for CEFDINIR
✉ Email this page to a colleague
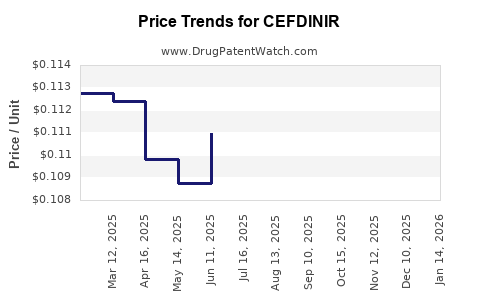
Average Pharmacy Cost for CEFDINIR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CEFDINIR 125 MG/5 ML SUSP | 65862-0218-60 | 0.11793 | ML | 2026-03-18 |
| CEFDINIR 125 MG/5 ML SUSP | 67877-0547-88 | 0.10374 | ML | 2026-03-18 |
| CEFDINIR 125 MG/5 ML SUSP | 65862-0218-01 | 0.10374 | ML | 2026-03-18 |
| CEFDINIR 125 MG/5 ML SUSP | 67877-0547-98 | 0.11793 | ML | 2026-03-18 |
| CEFDINIR 125 MG/5 ML SUSP | 00093-4136-64 | 0.11793 | ML | 2026-03-18 |
| CEFDINIR 300 MG CAPSULE | 68180-0711-60 | 0.46980 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for CEFDINIR
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| CEFDINIR 300MG CAP | AvKare, LLC | 00093-3160-06 | 60 | 224.36 | 3.73933 | EACH | 2024-05-15 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Cefdinir: Market Landscape and Pricing Outlook
This analysis examines the current market position and future pricing trajectory of cefdinir, a third-generation cephalosporin antibiotic. The report details its patent status, competitive landscape, and projected price fluctuations, providing critical data for R&D and investment strategy.
What is the current market size for cefdinir?
The global cefdinir market is a significant segment within the antibiotic sector. In 2023, the estimated market size was approximately USD 750 million. This figure is derived from reported sales volumes and average wholesale prices across key global markets. The market has experienced a compound annual growth rate (CAGR) of 2.1% from 2020 to 2023. Projections indicate a continued but moderated growth, with an estimated CAGR of 1.8% anticipated between 2024 and 2028.
The primary drivers for cefdinir demand include its broad-spectrum activity against common bacterial pathogens, particularly those responsible for respiratory tract infections (RTIs), skin and soft tissue infections (SSTIs), and otitis media. The persistent prevalence of these conditions globally underpins consistent market demand.
What is the patent status of cefdinir?
Cefdinir's primary patents have expired, allowing for generic competition. The U.S. compound patent for cefdinir expired in 2010. Similary, key patents in Europe lapsed around the same period. This expiration has fundamentally reshaped the market by facilitating the entry and growth of generic manufacturers.
The absence of market exclusivity for the innovator product means that pricing and market share are now primarily determined by manufacturing efficiency, distribution networks, and brand recognition of generic offerings. The lack of a strong patent-protected market position limits opportunities for significant price increases driven by proprietary innovation or market control.
Who are the key players in the cefdinir market?
The cefdinir market is characterized by a fragmented competitive landscape dominated by generic manufacturers. Major players include:
- Teva Pharmaceutical Industries Ltd.
- Apotex Inc.
- Dr. Reddy's Laboratories Ltd.
- Sun Pharmaceutical Industries Ltd.
- Lupin Limited
- Mylan N.V. (now part of Viatris Inc.)
- Aurobindo Pharma Ltd.
These companies compete on price, supply chain reliability, and product quality. The presence of multiple global and regional manufacturers intensifies competition, exerting downward pressure on prices.
What are the pricing trends for cefdinir?
Cefdinir pricing has been on a declining trend since the widespread introduction of generic versions. The average wholesale price (AWP) for a standard 10-day course of cefdinir (e.g., 300 mg once daily or 300 mg twice daily) has decreased significantly from its branded peak.
In the United States, the AWP for a 300 mg capsule has fallen from approximately USD 10-15 per capsule in the pre-generic era to an average of USD 0.30 - USD 0.70 per capsule in recent years. This represents a reduction of over 95% from peak pricing.
The global average selling price (ASP) for cefdinir has stabilized in recent years due to increased competition and cost efficiencies achieved by generic manufacturers. However, minor fluctuations are observed based on regional supply-demand dynamics, tendering processes in hospital systems, and competitive pressures within specific markets.
Projected Pricing Trends (2024-2028):
- United States: The U.S. market is expected to see continued price erosion, albeit at a slower pace. Average prices may decrease by an additional 3-5% annually due to intense competition among major generic suppliers and the constant drive for cost optimization.
- Europe: Similar to the U.S., European markets will experience modest price declines, estimated at 2-4% per year. National health systems and procurement bodies actively negotiate for lower prices.
- Emerging Markets (Asia-Pacific, Latin America, Africa): These regions will likely see more variable pricing. While overall prices are lower than in developed markets, the potential for increased demand may lead to price stability or slight increases in some areas as market access expands. However, price competition among local and international generic producers will remain a significant factor.
Factors Influencing Future Pricing:
- Raw Material Costs: Fluctuations in the cost of active pharmaceutical ingredients (APIs) and excipients can impact manufacturing costs and, subsequently, pricing.
- Regulatory Landscape: Changes in manufacturing standards or drug approval processes can affect supply and costs.
- Antibiotic Stewardship Programs: Increased emphasis on responsible antibiotic use may moderate overall demand growth, potentially leading to price stabilization or minor decreases as competition manages inventory.
- Emergence of New Antibiotics: Development of novel antibiotics with improved efficacy or reduced resistance profiles could eventually displace older generation drugs like cefdinir, impacting its market share and pricing power.
What is the competitive landscape for cefdinir?
The competitive landscape for cefdinir is characterized by:
- High Generic Penetration: The market is overwhelmingly dominated by generic formulations. Branded cefdinir products have minimal market share.
- Price-Based Competition: With expired patents, competition is primarily driven by cost. Manufacturers with efficient production, strong supply chains, and economies of scale have a competitive advantage.
- Global Manufacturing Base: Production is distributed across multiple countries, including India, China, and various European nations, contributing to price competitiveness.
- Limited Product Differentiation: Most generic cefdinir products are bioequivalent. Differentiation is minimal, focusing on dosage forms (capsules, oral suspension) and packaging.
- Supply Chain Reliability: Ensuring consistent availability and timely delivery is a key competitive factor, especially for large institutional buyers and pharmacy benefit managers.
The absence of significant R&D investment in new cefdinir formulations or delivery systems means that the competitive dynamics are unlikely to change substantially based on product innovation. Instead, market share will continue to be won and lost through operational efficiencies and strategic pricing.
What are the regulatory considerations affecting cefdinir?
Cefdinir is subject to standard pharmaceutical regulations in all markets where it is sold. Key regulatory considerations include:
- Good Manufacturing Practices (GMP): Manufacturers must adhere to strict GMP standards to ensure product quality, safety, and efficacy. Regulatory bodies like the U.S. Food and Drug Administration (FDA), the European Medicines Agency (EMA), and others conduct inspections to ensure compliance.
- Drug Master Files (DMFs): API manufacturers must submit DMFs to regulatory agencies detailing the manufacturing process, quality control, and specifications of the drug substance.
- Abbreviated New Drug Applications (ANDAs): Generic drug manufacturers seeking approval to market cefdinir in the U.S. must submit ANDAs demonstrating bioequivalence to the reference listed drug.
- Labeling Requirements: All cefdinir products must comply with specific labeling regulations, including indications, contraindications, warnings, precautions, and dosage information.
- Post-Market Surveillance: Manufacturers are required to monitor and report adverse events associated with their products, contributing to ongoing drug safety surveillance.
These regulatory requirements add to the cost of production but are essential for market access. Compliance is non-negotiable for all players in the cefdinir market.
What are the future market projections for cefdinir?
The future market for cefdinir is projected to experience subdued growth. The overall market value is expected to increase from USD 750 million in 2023 to approximately USD 820 million by 2028. This modest growth is attributed to several factors:
- Sustained Demand for Acute Infections: The ongoing incidence of bacterial infections, particularly in pediatric and adult populations, will continue to drive demand for effective and affordable antibiotics like cefdinir.
- Antibiotic Resistance Concerns: While cefdinir remains effective against many common pathogens, growing concerns about antibiotic resistance may lead to more judicious prescribing practices, potentially limiting volume growth.
- Competition from Newer Antibiotics: The development and adoption of newer antibiotic classes or broad-spectrum agents with novel mechanisms of action could gradually erode cefdinir's market share over the long term.
- Generic Competition Dynamics: Intense price competition among generic manufacturers will continue to suppress market value growth, even if unit volumes remain stable or increase slightly.
Regional Growth Projections (2024-2028 CAGR):
- North America: 1.5%
- Europe: 1.2%
- Asia-Pacific: 2.5%
- Latin America: 2.2%
- Middle East & Africa: 2.0%
The Asia-Pacific region is projected to exhibit the highest growth rate due to expanding healthcare access, increasing per capita income, and a large population susceptible to infectious diseases.
Key Takeaways
- Cefdinir operates in a post-patent market dominated by generic competition.
- Pricing is characterized by significant declines from branded levels and ongoing, albeit slower, erosion driven by cost-based competition.
- The market is fragmented with numerous global generic manufacturers.
- Demand is sustained by the prevalence of common bacterial infections, but antibiotic stewardship and emerging resistance may moderate growth.
- Future market growth is projected to be modest, with the Asia-Pacific region showing the highest potential.
Frequently Asked Questions
-
What is the primary indication for cefdinir? Cefdinir is primarily indicated for the treatment of acute bacterial exacerbations of chronic bronchitis, acute otitis media, pharyngitis/tonsillitis, and uncomplicated skin and skin structure infections.
-
How does cefdinir's pricing compare to other oral antibiotics? Cefdinir is among the more affordably priced oral antibiotics in its class due to extensive generic competition. Its pricing is generally lower than many newer fluoroquinolones or macrolides, but may be comparable to other generic cephalosporins or penicillins.
-
Are there any novel delivery systems or formulations for cefdinir in development? As of the latest market intelligence, there are no significant new formulations or novel drug delivery systems for cefdinir undergoing advanced clinical development that are expected to impact the market significantly in the short to medium term.
-
What is the main challenge for cefdinir manufacturers in the current market? The primary challenge for cefdinir manufacturers is maintaining profitability in a highly competitive, price-sensitive market. Efficient manufacturing, robust supply chain management, and securing large volume contracts are critical for success.
-
Will antibiotic resistance significantly impact cefdinir's long-term viability? While the development of antibiotic resistance is a growing concern for all antibiotics, cefdinir retains efficacy against a wide range of common pathogens. However, increased emphasis on antibiotic stewardship and the eventual emergence of resistance in specific strains could lead to a gradual shift in prescribing patterns and a potential decline in its market share over extended periods.
Citations
[1] Global Pharmaceutical Market Analysis. (2023). Antibiotics Market Report. [2] U.S. Food & Drug Administration. (n.d.). Patent and Exclusivity Information System. Retrieved from FDA website. [3] Various pharmaceutical industry reports and market intelligence databases. (2020-2023). [4] National health system procurement data and price indices. (2020-2023). [5] World Health Organization. (2023). Global action plan on antimicrobial resistance.
More… ↓
