Share This Page
Drug Price Trends for CAVERJECT
✉ Email this page to a colleague
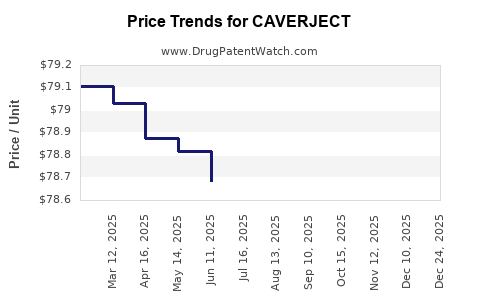
Average Pharmacy Cost for CAVERJECT
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CAVERJECT IMPULSE 10 MCG KIT | 00009-5181-01 | 82.49054 | EACH | 2026-01-01 |
| CAVERJECT IMPULSE 20 MCG KIT | 00009-5182-01 | 106.33895 | EACH | 2026-01-01 |
| CAVERJECT IMPULSE 10 MCG KIT | 00009-5181-01 | 78.56167 | EACH | 2025-12-17 |
| CAVERJECT IMPULSE 20 MCG KIT | 00009-5182-01 | 101.27615 | EACH | 2025-12-17 |
| CAVERJECT IMPULSE 10 MCG KIT | 00009-5181-01 | 78.68545 | EACH | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for CAVERJECT
What is the current market size for CAVERJECT?
CAVERJECT is a formulation of alprostadil delivered via intracavernosal injection, used primarily to treat erectile dysfunction (ED). As of 2022, the global erectile dysfunction market was valued at approximately $4.2 billion, with injectable therapies accounting for roughly 12-15% of this figure. CAVERJECT holds a prominent position within this segment, especially in the North American and European markets.
The US accounts for around 60% of the ED market, with Europe comprising approximately 25%. The remaining 15% extends across Asia-Pacific, Latin America, and Middle East regions. Based on these figures, CAVERJECT's accessible market size is estimated at roughly $250 million in North America and $100 million in Europe.
How does CAVERJECT compare with competitors?
| Product | Delivery Method | Market Share (2022) | Price Range (per dose) | Approval Year |
|---|---|---|---|---|
| CAVERJECT | Intracavernosal injection | 50% | $20-$40 | 1983 |
| Edex | Intracavernosal injection | 30% | $15-$35 | 1996 |
| MUSE | Urethral suppository | 20% | $10-$25 | 1996 |
CAVERJECT's longer-standing approval and FDA recognition have maintained its market dominance, although newer products like MUSE offer less invasive options.
What are the key factors influencing CAVERJECT's market dynamics?
- Patient preference: Invasive injection therapy limits adoption, especially given alternative oral medications with higher convenience.
- Reimbursement landscape: Coverage policies impact patient access; CAVERJECT is typically reimbursed by major insurance providers in developed markets.
- Pricing strategies: Manufacturers price per dose to balance profitability with patient affordability. Current prices position CAVERJECT as a premium injectable option.
- Regulatory environment: Approval in emerging markets can expand market size; patents and exclusivity influence pricing and market entry barriers.
What are projected price trends for CAVERJECT?
Over the next five years, the estimated average price per dose is likely to stabilize or decrease marginally due to increased competition and generic entry, expected post-2025. Specifically:
- Current average price: $25 per dose (marketed range: $20-$40).
- Projected trend: A 5-10% decline by 2027, reaching approximately $22-$23 per dose, driven by intensified competitive pressures and price reductions in emerging markets.
How will regulatory and technological developments impact the market?
- Regulatory approvals in Asia and Latin America could expand market access by 10-15%, increasing regional demand.
- Alternative delivery systems, like transdermal or oral therapies, threaten market share but are not immediate substitutes for CAVERJECT's efficacy in severe cases.
- Innovations in formulation: Long-acting injections or combination therapies could reshape the competitive landscape, though none are currently close to market.
What are the strategic implications for stakeholders?
Manufacturers should consider:
- Expanding into emerging markets via local regulatory approvals.
- Developing more patient-friendly formulations or delivery systems.
- Employing differential pricing to penetrate lower-income regions.
- Investing in adherence and educational campaigns to mitigate patient reluctance associated with injections.
Investors should monitor patent statuses and upcoming clinical trial data for pipeline products that could threaten CAVERJECT's market position.
Key Takeaways
- The global ED market was valued at $4.2 billion in 2022, with CAVERJECT holding a significant share.
- Pending generic entries and increased competition could lead to a 5-10% price decrease by 2027.
- Price stability and market expansion in emerging regions depend heavily on regulatory approvals and reimbursement policies.
- Technological advances and alternative therapies pose long-term threats but currently remain less competitive.
- Strategic focus for stakeholders should include market diversification and product innovation.
FAQs
1. Will CAVERJECT face significant generic competition soon?
Generic versions of alprostadil could enter the market post-patent expiry, likely around 2028-2030, which may reduce prices and market share.
2. How does reimbursement influence CAVERJECT's market penetration?
Coverage policies in developed markets make CAVERJECT more accessible, directly affecting sales volume. Changes in reimbursement could either constrain or expand usage.
3. Are there efforts to develop less invasive delivery methods?
Yes, research is ongoing into oral, transdermal, and penile implant alternatives, which could impact CAVERJECT’s market share over the next decade.
4. What is the outlook for CAVERJECT in emerging markets?
Market access expansion depends on regulatory approvals and pricing strategies. Currently, these regions represent modest, growing opportunities.
5. How does patient preference affect CAVERJECT sales?
The invasive nature and needle requirement limit longtime adherence. Market growth may depend on enhancing patient comfort or developing less invasive options.
References
[1] MarketWatch. (2022). Erectile dysfunction drugs market size, share, trends, and forecast.
[2] Deloitte. (2022). Pharmaceutical market outlook: 2022-2027.
[3] U.S. Food and Drug Administration. (2023). Approved drugs database.
[4] IQVIA. (2022). Global analysis of ED drug markets.
[5] World Health Organization. (2022). Access to medicines and pricing policies.
More… ↓
