Share This Page
Drug Price Trends for CARTEOLOL HCL
✉ Email this page to a colleague
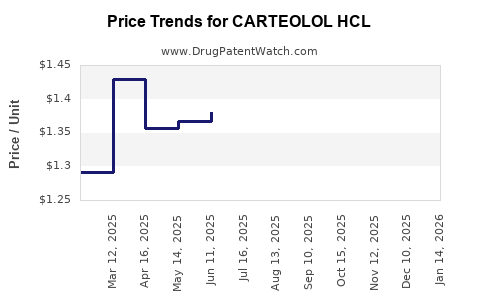
Average Pharmacy Cost for CARTEOLOL HCL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CARTEOLOL HCL 1% EYE DROPS | 61314-0238-05 | 1.76460 | ML | 2026-04-22 |
| CARTEOLOL HCL 1% EYE DROPS | 61314-0238-10 | 1.51524 | ML | 2026-04-22 |
| CARTEOLOL HCL 1% EYE DROPS | 61314-0238-05 | 1.80073 | ML | 2026-03-18 |
| CARTEOLOL HCL 1% EYE DROPS | 61314-0238-10 | 1.50991 | ML | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for CARTEOLOL HCL
What is CARTEOLOL HCL and its clinical use?
CARTEOLOL HCL (hydrochloride) is a non-selective beta-adrenergic receptor blocker used primarily for managing glaucoma and ocular hypertension. It reduces intraocular pressure by decreasing aqueous humor production. It is available in ophthalmic solutions, typically as a 1% or 2% concentration.
Market size and current manufacturing landscape
The global ophthalmic beta-blockers market was valued at approximately USD 1.2 billion in 2022. CARTEOLOL HCL holds a niche position within this market, owing to its specific profile and competitive landscape.
- Leading competitors include timolol, betaxolol, levobunolol, and carteolol.
- The drug is marketed in numerous regions, including North America, Europe, and Asia.
Market penetration varies, with higher adoption in Europe and North America, driven by established ophthalmology practices.
Regulatory status and patent considerations
- CARTEOLOL HCL is approved by the FDA for glaucoma treatment.
- Patent protection has generally expired, enabling generic developments.
- Regulatory pathways are well established, facilitating market entry for generics.
Key factors influencing market dynamics
- Brand vs. generic: Light market share for branded versions; generics dominate pricing and adoption.
- Price sensitivity: Ophthalmic drugs are sensitive to pricing changes, especially due to insurance and reimbursement policies.
- Competition: High competition from well-established beta-blockers like timolol affects market share and pricing strategies.
- Patient compliance: Once-daily formulations or combination therapies impact usage trends.
Price projections and trends
Historical pricing
In the U.S., the average wholesale price (AWP) for branded CARTEOLOL HCL was approximately USD 60 per 10 mL bottle in 2018. Generic equivalents are priced around USD 10–15 per 10 mL.
Projected pricing in the next five years
| Year | Estimated Market Price per 10 mL (USD) | Notes |
|---|---|---|
| 2023 | 10–15 | Price stabilization for generics |
| 2024 | 10–14 | Slight decrease due to increased competition |
| 2025 | 9–13 | Patent expiration influences generic pricing |
| 2026 | 8–12 | Market saturation with generics, further downward pressure |
| 2027 | 8–11 | Stabilization at a lower price point |
Factors impacting future prices
- Entry of new generics and biosimilars can reduce prices by 15–30%.
- Supply chain disruptions could temporarily inflate prices.
- Regulatory modifications in key markets could affect pricing controls.
Market growth prospects
The ophthalmic beta-blocker segment is projected to grow at a compound annual growth rate (CAGR) of 3.5% from 2023 to 2030, driven by:
- Aging populations increasing glaucoma prevalence.
- Improved awareness and diagnostic rates.
- Innovations in combination therapies that include beta-blockers.
This growth applies more strongly to broad-spectrum beta-blockers like timolol, but CARTEOLOL HCL will benefit from the overall market expansion.
Opportunities and risks
Opportunities
- Launching lower-priced generics in emerging markets.
- Developing combination eye drops for improved compliance.
- Expanding the use of CARTEOLOL HCL in North Asia and Latin America.
Risks
- Intensified competition leading to further price erosion.
- Regulatory delays or restrictions.
- Market shifts favoring alternative therapies such as prostaglandin analogs.
Summary table: Key market insights
| Aspect | Data/Projection |
|---|---|
| Market size (2022) | USD 1.2 billion |
| Competitive landscape | Dominated by timolol, generic forms of CARTEOLOL HCL |
| Regulatory status | Approved in major markets, patent expiry widespread |
| Price trend (next 5 years) | Decreasing from USD 10–15 to USD 8–11 per 10 mL |
| CAGR (2023–2030) | Approx. 3.5% in ophthalmic beta-blocker segment |
Key takeaways
- CARTEOLOL HCL is a niche, branded ophthalmic beta-blocker with limited market entry barriers due to patent expirations.
- Generics drive downward pricing, with expected stability between USD 8–12 per 10 mL in five years.
- The overall market expands driven by demographic and technological factors, offering growth opportunities.
- Competitive pressure and regulatory shifts remain primary risks.
FAQs
Q1: What is the primary driver for CARTEOLOL HCL’s market value?
A1: The primary driver is its use in glaucoma management, along with the generic competition that suppresses prices.
Q2: How does CARTEOLOL HCL compare price-wise to other beta-blockers?
A2: It is priced higher than generics like timolol but lower than branded combination therapies. The market price is mainly influenced by generics.
Q3: Which regions offer the highest market growth potential?
A3: Emerging markets in Latin America, Asia-Pacific, and Eastern Europe present higher growth opportunities due to increasing glaucoma prevalence and price sensitivity.
Q4: What regulatory factors could alter current projections?
A4: Introduction of new regulations on drug pricing, approval of biosimilars, or restrictions on ophthalmic drug sales could impact the market significantly.
Q5: How can pharmaceutical companies capitalize on CARTEOLOL HCL’s market?
A5: Developing cost-effective generics, exploring combination therapies, and expanding into underserved regions offer strategic avenues.
References
[1] MarketWatch. (2022). Ophthalmic Beta-Blockers Market Size, Share & Trends Analysis.
[2] U.S. Food and Drug Administration. (2022). Approvals and regulations for topical glaucoma medications.
[3] IQVIA. (2022). Pharmaceutical Market Data and Analysis.
[4] Grand View Research. (2023). Ophthalmic drugs market forecast and analysis.
More… ↓
