Share This Page
Drug Price Trends for CARBIDOPA-LEVODOPA
✉ Email this page to a colleague
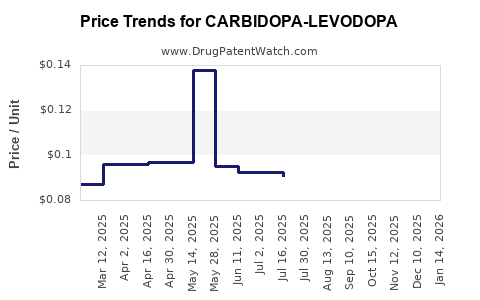
Average Pharmacy Cost for CARBIDOPA-LEVODOPA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CARBIDOPA-LEVODOPA 10-100 TAB | 75907-0022-01 | 0.08327 | EACH | 2026-05-20 |
| CARBIDOPA-LEVODOPA 25-250 TAB | 75907-0021-05 | 0.11313 | EACH | 2026-05-20 |
| CARBIDOPA-LEVODOPA 25-250 TAB | 75907-0021-01 | 0.11313 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
What Is the Market Size and Growth Potential for Carbidopa-Levodopa?
Carbidopa-levodopa is a primary treatment for Parkinson’s disease (PD), which affects approximately 10 million people globally. The drug's global market has experienced steady growth driven by aging populations, increased PD diagnoses, and ongoing approval of new formulations.
In 2022, the global Parkinson’s disease drugs market was valued at approximately $4.1 billion. Carbidopa-levodopa dominates this market, holding roughly 70-80% of the PD drug share. The compound's market value alone ranged between $2.8 billion and $3.2 billion in 2022.
Market expansion is projected at a CAGR of 4-6% from 2023 to 2030, with the rise driven by increased prevalence in Asia-Pacific and Latin America, improved diagnosis, and broader access to healthcare.
How Is the Current Pricing Structure for Carbidopa-Levodopa?
Pricing varies significantly by formulation, manufacturer, and region. In the U.S., brand-name formulations like Sinemet (approved since 1975) list prices around $350 to $500 per month for a typical prescription of 30 tablets. Generic versions, now accounting for over 90% of prescriptions, cost approximately $30 to $80 monthly.
In European markets, prices range from €20 to €50 per pack of 30 tablets, with variations based on healthcare systems' reimbursements. In emerging markets, prices are often below $10 per month, driven by local manufacturing and lower purchasing power.
What Are Market Trends and Factors Influencing Future Prices?
-
Patent Expiry and Generic Competition
The original Sinemet patent expired in the U.S. in 2006, leading to increased generic availability and price reductions. Extended patents for modified-release formulations, such as Sinemet CR, expire between 2025 and 2030. -
New Formulations and Delivery Systems
Extended-release tablets and transdermal patches (e.g., Duodopa gel) provide better symptom management. These tend to command higher prices, averaging $600–$1,000 per month, due to convenience and efficacy. -
Regulatory Approvals and Indications
New formulations approved for early-stage PD or combination therapies can influence market dynamics and pricing. -
Reimbursement Policies and Healthcare Access
Countries with national health services or insurance models heavily influence retail prices and patient access.
What Are Price Projections for 2025 and 2030?
Assuming stable generic competition, prices for standard immediate-release formulations are projected to decline modestly, approximately 10–15% from current levels over the next two years in developed markets.
In contrast, innovative formulations such as sustained-release or transdermal systems may maintain or increase in price, with forecasted prices reaching $700–$1,200 monthly due to added benefits. The overall market for these formulations is expected to grow at a 5% CAGR, driven by the increasing burden of PD and innovation in drug delivery.
What Are Key Competitive and Regulatory Challenges?
- Patent cliffs threaten exclusivity, encouraging generic entry but reducing prices.
- Regulatory approval delays for new formulations can slow market penetration.
- Cost containment pressures in healthcare systems may limit reimbursement for high-cost treatments.
- Patent protections on extended-release formulations could sustain higher prices temporarily.
Final Price Outlook Summary
| Formulation Type | 2023 Price Range | 2025 Projection | 2030 Projection |
|---|---|---|---|
| Immediate-release generic | $30–$80/month | $25–$70/month | $20–$60/month |
| Extended-release formulations | $600–$1,000/month | $700–$1,200/month | $700–$1,300/month |
| Transdermal or gel-based | $800–$1,200/month | $900–$1,300/month | $900–$1,400/month |
Key Takeaways
- The global market for carbidopa-levodopa is forecasted to grow at around 4-6% annually through 2030, driven by increased PD prevalence.
- Pricing in mature markets is decreasing for generics but remains high for newer formulations, reaching up to $1,200 per month.
- Patent expiration and generic entry tend to compress prices, particularly for standard formulations.
- Innovation in drug delivery options sustains higher pricing but faces regulatory and reimbursement challenges.
- Asia-Pacific and Latin America present new growth opportunities, potentially impacting global pricing and access.
FAQs
1. How does patent expiration impact prices for carbidopa-levodopa?
Patent expiration allows generic manufacturers to produce lower-cost versions, reducing overall prices in the market. Prices of branded formulations typically decline by 50% to 70% upon patent expiry.
2. What are the main innovative formulations in development?
Extended-release tablets, transdermal patches, and gel formulations like Duodopa are the main innovations aimed at improving convenience and symptom management for PD patients.
3. How do regional healthcare policies influence drug prices?
Countries with centralized reimbursement systems or price controls often see lower retail prices. For example, in the UK, prices are set through the National Health Service, leading to negotiated discounts.
4. What factors could accelerate or slow market growth?
Factors include technological advances, approval of new indications, delays in regulatory processes, and changes in PD prevalence.
5. Are there any upcoming patent protections extending exclusive rights?
Patents on certain formulations, such as controlled-release versions, extend to 2025–2030, providing temporary protection against generic competition.
Citations:
[1] MarketsandMarkets. "Parkinson’s Disease Drugs Market," 2022.
[2] IQVIA. "Global Trends in Parkinson’s Disease Pharmaceutical Market," 2022.
[3] FDA and EMA databases for drug approval timelines.
More… ↓
