Share This Page
Drug Price Trends for CARBIDOPA-LEVO ER
✉ Email this page to a colleague
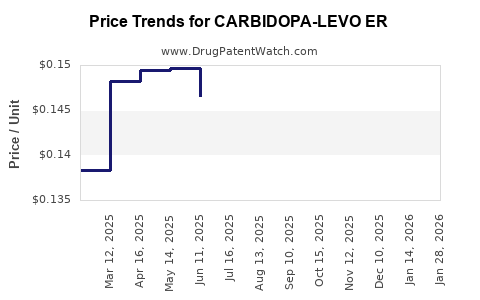
Average Pharmacy Cost for CARBIDOPA-LEVO ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CARBIDOPA-LEVO ER 23.75-95 CAP | 70710-2139-01 | 2.86860 | EACH | 2026-03-18 |
| CARBIDOPA-LEVO ER 25-100 TAB | 50228-0460-01 | 0.11027 | EACH | 2026-03-18 |
| CARBIDOPA-LEVO ER 25-100 TAB | 16729-0078-01 | 0.11027 | EACH | 2026-03-18 |
| CARBIDOPA-LEVO ER 61.25-245 CP | 70710-2142-01 | 3.77764 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
CARBIDOPA-LEVO ER Market Analysis and Financial Projection
Market Analysis and Price Projections for CARBIDOPA-LEVO ER
Market Overview
Carbidopa-Levodopa Extended Release (ER) is used for Parkinson’s disease management. The product combines levodopa, a dopamine precursor, with carbidopa to inhibit peripheral conversion of levodopa, thereby increasing central nervous system availability. The ER formulation improves motor function by providing a more stable dopaminergic response compared to immediate-release versions.
Market Size and Demand
As of 2022, the global Parkinson’s disease market is estimated at $4 billion, with an annual growth rate of approximately 5%. A substantial share pertains to levodopa-based therapies, including ER formulations, due to the increasing prevalence of Parkinson’s in aging populations:
- Prevalence: Estimated 10 million worldwide, rising by 3% annually.
- Market Share: Levodopa products account for roughly 60% of Parkinson's treatment sales.
- Regional Distribution:
- North America: 40%
- Europe: 30%
- Asia-Pacific: 20%
- Rest of World: 10%
The increased adoption of extended-release formulations stems from improved adherence, reduced dosing frequency, and decreased fluctuations in motor control.
Competitive Landscape
Leading brands include:
- Sinemet CR (AbbVie): Market leader, generating over $600 million annually in the US.
- Rytary (Hektoen Inc.): Launched in 2015, capturing significant market share in the US and Europe.
- Generic versions: Growing presence, compressing prices.
The extended-release segment faces competitive pressure from generic formulations, which have contributed to price erosion in recent years.
Patent Status and Regulatory Outlook
- Patent Expiration: Many patents for branded ER formulations expired between 2020 and 2022.
- Regulatory Approvals: Regulatory agencies like the FDA and EMA approve both branded and generic versions, adapting quickly to new filings.
Price Trends and Projections
| 2022 Base Prices: | Product Type | Average Wholesale Price (AWP) per 30-day supply | Notes |
|---|---|---|---|
| Branded ER (e.g., Rytary) | $500 – $700 | Higher costs, brand loyalty influences pricing | |
| Generic ER | $150 – $300 | Price declines post-patent expiry |
Projecting for 2023-2027:
- Volume Growth: 4-6% annually, driven by aging populations.
- Price Trends:
- Branded: Slight decrease (~5%) as generics expand.
- Generics: Further declines of 10-15% annually due to increased competition.
| Estimated 2027 Prices: | Product Type | 30-day supply Price (USD) | Notes |
|---|---|---|---|
| Branded ER (Rytary) | ~$430 – $630 | Slight decline expected | |
| Generic ER | ~$110 – $250 | Continued price erosion |
Factors Influencing Future Prices
- Patent Litigation and Expirations: New generics entering markets post-2022 will drive prices downward.
- Regulatory Changes: Accelerated approvals or biosimilar regulations could impact pricing.
- Market Penetration: Increased adoption of ER formulations in emerging markets may sustain volumes despite pricing pressure.
- Pricing Policies: Payer negotiations, formulary placements, and discounts influence net prices.
Risks and Opportunities
- Risks:
- Slow adoption in lower-income regions.
- Price competition from generics.
- Potential regulatory hurdles for new formulations or biosimilars.
- Opportunities:
- Innovation in delivery systems—e.g., microtablets or continuous infusion devices.
- Expansion into emerging markets.
- Development of combination therapies with neuroprotective agents.
Key Takeaways
- The global Parkinson’s disease market is expanding, with levodopa ER formulations holding a substantial share.
- Branded products like Rytary dominate early post-expiration but face rapid price declines with generics.
- Prices for branded 30-day supplies of ER formulations are projected to decrease 5% annually through 2027, reaching approximately $430–$630.
- Generic prices are expected to decline by 10–15% annually, potentially reducing costs to below $200.
- Market growth is supported by aging demographics, but pricing pressures intensify with patent expirations and increased competition.
FAQs
-
What factors primarily drive the pricing of CARBIDOPA-LEVO ER products?
Patent status, market competition, manufacturing costs, payer negotiations, and regional policies influence prices.
-
When are major patents for branded CARBIDOPA-LEVO ER formulations expiring?
Key patents expired between 2020 and 2022; additional patents may expire in the next 2-3 years.
-
How does market penetration of generics impact branded drug prices?
Increased generic availability exerts downward pressure, reducing branded drug prices by approximately 5% annually post-patent expiration.
-
What are the growth prospects for the CARBIDOPA-LEVO ER market?
Expected to grow at 4–6% annually through 2027, driven by an aging population and increased disease prevalence.
-
What are potential innovations affecting future price trends?
New delivery mechanisms, biosimilars, and combination therapies could alter product costs and market dynamics.
References
- MarketWatch. “Parkinson’s Disease Therapeutics Market Size, Share & Trends Analysis,” 2022.
- EvaluatePharma. “Levodopa Market Data,” 2022.
- FDA. “Patent and Exclusivity Data for Parkinson’s Treatments,” 2023.
- IQVIA. “Global Pharmaceutical Market Reports,” 2022.
- Grand View Research. “Parkinson’s Disease Treatment Market Analysis,” 2022.
More… ↓
