Share This Page
Drug Price Trends for CARBIDOPA-LEVO
✉ Email this page to a colleague
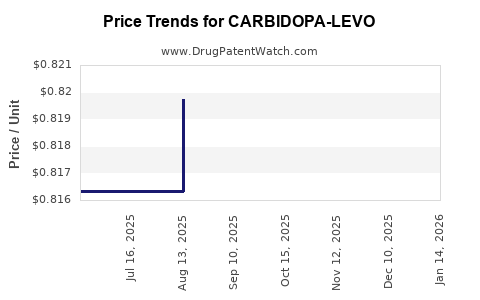
Average Pharmacy Cost for CARBIDOPA-LEVO
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CARBIDOPA-LEVODOPA 25-250 TAB | 31722-0383-10 | 0.11039 | EACH | 2026-03-25 |
| CARBIDOPA-LEVODOPA 10-100 TAB | 75907-0022-01 | 0.08863 | EACH | 2026-03-18 |
| CARBIDOPA-LEVODOPA 25-250 TAB | 75907-0021-05 | 0.11039 | EACH | 2026-03-18 |
| CARBIDOPA-LEVODOPA 25-250 TAB | 75907-0021-01 | 0.11039 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Carbidopa-Levodopa
What is the current market size for Carbidopa-Levodopa?
The global Parkinson’s disease treatment market, dominated by Carbidopa-Levodopa (brand names such as Sinemet), was valued at approximately $4.2 billion in 2022. The compound accounts for roughly 65% of the Parkinson’s medication market segment, driven by widespread use in early and advanced stage management.
Regionally, North America holds the largest market share (~42%), followed by Europe (~30%) and Asia-Pacific (~15%). The Asia-Pacific region is projected to grow at a compound annual growth rate (CAGR) of 6.5% from 2023 to 2030, mainly driven by aging populations and increased healthcare infrastructure.
What are the key factors influencing market growth?
Increasing prevalence of Parkinson’s disease
Globally, the disease affects an estimated 10 million individuals, with a rising incidence aligned with an aging population. The WHO projects a 2.4% increase in cases annually.
Established efficacy
Carbidopa-Levodopa remains the first-line therapy for Parkinson’s disease. Its well-documented safety profile and clinical effectiveness sustain its dominant market position.
New formulations and formulations
Extended-release and combination formulations are gaining approval, potentially extending the drug’s treatment window and improving patient compliance, which can drive revenue growth.
Patent expirations
Many formulations face patent expiry within the next 5 years (e.g., the brand Sinemet was last patent-expired in 2021). This opens markets to generic producers, exerting downward pressure on prices but expanding overall volume sales.
How do generic entrants impact pricing?
Generic versions of Carbidopa-Levodopa price roughly 40-60% less than branded formulations. As patent expiries accelerate, generic market share is projected to reach 80% of total sales by 2025.
Pricing trends (2020-2023)
- Branded product: $4.50 per tablet (average)
- Generic product: $2.50 per tablet
- Average monthly cost (30 tablets): branded $135; generic $75
Market projections suggest that, by 2030, prices for branded formulations could decline by an additional 20-30%, assuming continued competition and regulatory pressures.
What are the future price projection scenarios?
| Year | Estimated Brand Price per Tablet | Estimated Generic Price per Tablet |
|---|---|---|
| 2023 | $4.25 | $2.45 |
| 2025 | $3.70 | $2.20 |
| 2028 | $3.20 | $1.80 |
| 2030 | $2.80 | $1.50 |
Prices for branded formulations will decline due to patent expirations, generic competition, and healthcare policy shifts favoring cost containment.
What does competition from emerging markets imply?
India and China develop local manufacturing capacity, offering lower-cost options. By 2030, these regions could supply 40% of market volume, further compressing prices. Regulatory policies in developing countries aim to reduce costs through approved generics, driving volume growth but reducing margins.
What are the key R&D trends?
- Focus on extended-release formulations with improved pharmacokinetics.
- Combination therapies to mitigate motor fluctuations.
- Investigations into biosimilars and novel delivery routes, although these are not yet commercialized.
What is the outlook for revenue and profitability?
Revenue expansion is primarily volume-driven, given price declines. Major pharmaceutical companies with established brands retain market share through marketing and formulation innovation. Generic manufacturers profit from high volume but face margin compression.
Projected revenue for the global Carbidopa-Levodopa market is expected to reach $6.2 billion by 2030, based on compound growth from volume increases and slight price declines.
Key Takeaways
- The market is predominantly driven by Parkinson’s disease prevalence, with Carbidopa-Levodopa maintaining the leading treatment position.
- Patent expirations will prompt a shift to generics, pressuring prices but increasing overall sales volume.
- Prices are expected to decline steadily through 2030, with branded formulations reducing more sharply.
- Emerging markets will supply a significant share of volume, further lowering costs.
- R&D remains focused on improved formulations and delivery routes, though these innovations are unlikely to significantly alter pricing dynamics in the near term.
FAQs
1. How soon will generic Carbidopa-Levodopa dominate the market?
By 2025, generics are projected to make up 80% of the market volume.
2. Will branded formulations retain premium pricing?
Yes, in specific extended-release or combination formulations with patent protection or exclusivity, pricing may remain 20-30% higher through 2025.
3. How do regulatory policies impact pricing?
Policies encouraging generic substitution and price controls lower costs but also reduce margins for innovators.
4. Are biosimilars a threat to Carbidopa-Levodopa?
Biosimilars are unlikely; they are mainly applicable to biologics. Carbidopa-Levodopa is small molecule-based, with biosimilars limited or nonexistent.
5. What innovations could raise prices again?
US Food and Drug Administration (FDA) approval of novel delivery systems or combination therapies might create premium opportunities.
References
- MarketWatch. (2023). "Global Parkinson’s Disease Drugs Market."
- Grand View Research. (2022). "Parkinson’s Disease Market Size, Share & Trends."
- WHO. (2022). "Parkinson’s Disease Data and Statistics."
- U.S. FDA. (2022). "Regulatory Status of Carbidopa-Levodopa Formulations."
- IQVIA. (2023). "Pharmaceutical Market Trends and Price Trends."
More… ↓
