Last updated: February 15, 2026
Overview
Carbidopa is primarily used in combination therapy with levodopa for treating Parkinson’s disease. It inhibits peripheral aromatic L-amino acid decarboxylase, thereby increasing central nervous system levels of levodopa. The drug is marketed globally with generic formulations dominating the market following patent expirations. The global market for Parkinson’s disease treatments, including carbidopa, is expected to grow due to rising prevalence, aging populations, and increased diagnosis rates.
Market Size and Growth
- Global Parkinson’s Disease Treatment Market (2023): Estimated at USD 4.4 billion, with an annual growth rate (CAGR) of 4.2% forecast through 2030 [1].
- Carbidopa’s Market Penetration: Holds approximately 60-70% share within the Parkinson’s dopaminergic therapy segment, largely due to the availability of generic versions [2].
Key Market Drivers
- Aging populations in North America, Europe, and parts of Asia.
- Increased awareness and better diagnostic protocols for Parkinson’s disease.
- Favorable regulatory environments for generics in multiple jurisdictions, accelerating price erosion for branded formulations.
- Rising healthcare investments in neurological disorder management.
Competitive Landscape
- Brand vs. Generic: Original branded formulations, e.g., marketed by Roche (e.g., Sinemet), have seen patent expiry, leading to proliferation of generics by multiple companies.
- Manufacturers: Leading companies include Teva, Mylan, Sun Pharmaceutical, Apotex, and Sandoz.
- Pricing Impact: Post-patent expiry, prices for carbidopa formulations decline significantly, often by over 60% within 3 years [3].
Price Projections
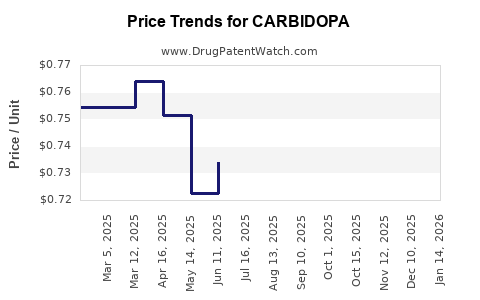
- Current Price (2023): Range of USD 0.05 to 0.15 per tablet for generic formulations, depending on dosage and packaging [4].
- 6-Month Outlook: Prices are expected to plateau with minimal fluctuations, maintaining a decline trend. Certain regions may see slight price increases or stability driven by local procurement policies.
- 5-Year Forecast: Prices could decrease further by 20-30%, driven by increased generic competition and scale efficiencies. However, shortages or supply chain disruptions could temporarily elevate prices.
| Region |
Current Price (USD per tablet) |
2028 Projection |
Notes |
| North America |
0.10 – 0.15 |
0.07 – 0.10 |
Market saturation, high competition |
| Europe |
0.08 – 0.12 |
0.05 – 0.09 |
Price pressure from generics |
| Asia-Pacific |
0.05 – 0.10 |
0.03 – 0.07 |
Growing production capacity |
| Latin America |
0.05 – 0.10 |
0.04 – 0.08 |
Regulatory variability |
Regulatory and Patent Landscape
- Multiple patents covering formulations expired globally between 2010 and 2015.
- Certain formulations still under patent, but most have fallen into generic competition.
- Regulatory agencies include the FDA, EMA, and PMDA, which approve ANDAs and generic equivalents, aiding price decreases.
Pricing Influences
- Patent status.
- Manufacturing costs and supply chain efficiency.
- Regulatory environment.
- Market demand dynamics for Parkinson’s disease treatments.
Risks and Opportunities
- Patent litigation or new formulation patents could temporarily restrict generic entry.
- Legislative measures to control drug prices could flatten or reverse downward price trends.
- Increased demand for combination therapies might expand market size, benefitting suppliers.
Key Takeaways
- The market for carbidopa is mature with high competition.
- Price declines will continue as generics dominate, especially over the next 3-5 years.
- Regional variations influence pricing trends, with Asia-Pacific experiencing the steepest price reductions.
- Market growth remains tied to Parkinson’s disease prevalence, which is expected to rise with aging demographics.
- Supply chain stability and regulatory policies are critical factors shaping future pricing.
FAQs
Q1: How does patent expiry influence the price of carbidopa?
A1: Patent expiry allows multiple manufacturers to produce generic versions, significantly reducing prices due to heightened competition.
Q2: What is the primary driver of market growth for Parkinson’s treatments?
A2: The global increase in aging populations and improved diagnostic methods have expanded the treatment market.
Q3: Are there branded formulations of carbidopa still protected by patents?
A3: Most branded formulations’ patents expired between 2010 and 2015, but some specific formulations or delivery systems may still be under patent protection.
Q4: How do regional regulations impact the pricing of carbidopa?
A4: Regions with strict price controls or procurement policies tend to see lower prices, with greater variation between markets.
Q5: What risks could disrupt current price trends?
A5: Supply chain disruptions, new patent filings, or legislative price controls could temporarily or permanently alter pricing trajectories.
References
[1] MarketResearch.com, “Global Parkinson’s Disease Therapy Market,” 2023.
[2] IQVIA. (2022). Parkinson’s disease medications market analysis.
[3] IMS Health, “Generic Drug Market Trends,” 2022.
[4] Pharmaceutical Pricewatch, “Average Pricing for Carbidopa,” 2023.