Last updated: February 12, 2026
What Is the Current Market Size for Carbamazepine?
Carbamazepine is a longstanding anticonvulsant and mood-stabilizing medication, primarily indicated for epilepsy, trigeminal neuralgia, and bipolar disorder. The drug has been on the market for over 50 years. The global market revenue for carbamazepine was approximately $500 million in 2022, with anticipated compound annual growth rate (CAGR) of 3.2% through 2030. The United States accounts for roughly 40% of the market share, followed by Europe and Asia-Pacific regions.
The drug's popularity stems from its low-cost manufacturing and widespread acceptance as a first- or second-line therapy for focal seizures. Patent protections expired in the early 2000s, resulting in an increase in generic versions that dominate sales.
How Is the Patent and Regulatory Landscape Shaping the Market?
Carbamazepine is off-patent globally. No new patent protections exist for the original molecule, which constrains the potential for brand-name pricing above generics. The Food and Drug Administration (FDA) approved formulations primarily include immediate-release and controlled-release versions. Recent regulatory strategies are focused on optimizing bioavailability and reducing side effects, such as Stevens-Johnson syndrome, especially through the development of formulations with improved safety profiles.
Generic manufacturers, including Teva, Mylan, Sun Pharma, and others, account for over 90% of global sales. Patent challenges are minimal, but some regions have regulatory hurdles or market access barriers, affecting regional price dynamics.
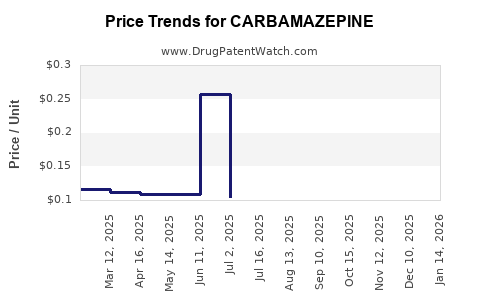
What Are the Main Price Trends for Carbamazepine?
Generic carbamazepine tablets are sold at an average wholesale price (AWP) of around $0.10 per 200 mg tablet. Brand-name formulations, such as Tegretol, are priced approximately 4-5 times higher, with AWPs around $0.50 per 200 mg tablet.
Price erosion has persisted over the last decade due to increased competition. In North America, generic prices fell by approximately 30% between 2015 and 2020, stabilizing thereafter. The price difference between generic and brand-name versions can reach 80% in regions where generics dominate.
What Factors Influence Price Projections?
Several factors influence future pricing and market volume:
-
Patent Status: As patents have expired worldwide, new patent protections are unlikely. This limits real price increases for existing formulations.
-
Generic Competition: The presence of multiple generic manufacturers maintains low prices. Any reduction in competition, such as market exits, could lead to price increases.
-
Formulation Innovations: Development of extended-release or combination products may command premium prices, potentially creating select niche markets.
-
Regulatory Changes: Adoption of policies promoting generic substitution or price controls in major markets could suppress prices further.
-
Pricing Policies: National health systems and insurance reimbursement practices significantly influence actual transaction prices.
What Are the Future Price Projections?
Based on current trends, no significant price increases are foreseen for standard formulations of carbamazepine. Prices are expected to remain stable or decline slightly over the next five years, primarily driven by increased generic competition and market saturation.
Premium formulations, such as extended-release versions, may see modest price increases, potentially reaching up to $0.20 per 200 mg tablet, reflecting improved safety or convenience attributes. However, these niche products are unlikely to significantly impact the global market value, which predominantly relies on low-cost generics.
How Will Market Dynamics Evolve?
Market demand will likely grow at a CAGR of around 3% through 2030, driven by unmet needs in developing regions and increased off-label use in bipolar disorder. Opportunities for growth pertain mainly to formulations with improved tolerability and compliance, as well as combination therapies.
Market consolidation and manufacturing efficiencies will further pressure prices downward. Regulatory emphasis on biosimilar and generic drug proliferation will keep costs low but may restrict profit margins for manufacturers seeking higher prices.
Key Takeaways
- Market size for carbamazepine stood at $500 million in 2022, with steady growth projected through 2030.
- Off-patent status and widespread generic competition keep prices stable or declining.
- Average generic tablet prices hover around $0.10 per 200 mg, with brand-name versions up to five times as expensive.
- Innovations in formulations may command premium pricing but are unlikely to alter the overall market trend.
- Price stability is expected absent regulatory or market disruptions, with minimal upward movement anticipated.
FAQs
1. Are there any new patented formulations of carbamazepine in development?
No. Current research focuses on improved formulations rather than new patents. Most development aims to enhance safety, tolerability, or compliance.
2. How does regional pricing vary?
Prices are lowest in regions with high generic penetration, such as India and Latin America, and higher in markets with regulations limiting generic substitution, like certain European countries and the US.
3. Will biosimilars or novel formulations impact market prices?
Given carbamazepine is a small molecule, biosimilars are not applicable. Novel formulations might lead to slight price increases in certain segments but will not disrupt overall pricing trends.
4. What are the main drivers for market growth?
Increasing epilepsy prevalence, especially in developing countries, and off-label use for bipolar disorder are primary growth factors.
5. Are supply chain disruptions likely to affect prices?
Potential supply-side issues—like shortages or manufacturing delays—could temporarily impact prices, but sustained effects are unlikely given the generic manufacturing landscape.
References
- Grand View Research, “Carbamazepine Market Size, Share & Trends,” 2022.
- IQVIA, “Global Pharmaceutical Market Data,” 2022.
- FDA Drug Database, “Approved formulations of Carbamazepine,” 2023.
- IMS Health, “Generic Drug Pricing Trends,” 2021.
- European Medicines Agency, “Regulatory overview of Carbamazepine,” 2023.