Share This Page
Drug Price Trends for CANASA
✉ Email this page to a colleague
Average Pharmacy Cost for CANASA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
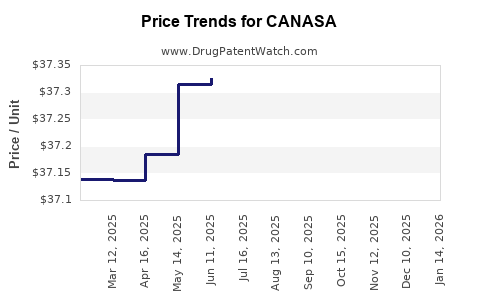
| CANASA 1,000 MG SUPPOSITORY | 58914-0501-56 | 37.06467 | EACH | 2026-05-20 |
| CANASA 1,000 MG SUPPOSITORY | 58914-0501-56 | 37.07905 | EACH | 2026-04-22 |
| CANASA 1,000 MG SUPPOSITORY | 58914-0501-56 | 37.30730 | EACH | 2026-03-18 |
| CANASA 1,000 MG SUPPOSITORY | 58914-0501-56 | 37.30908 | EACH | 2026-02-18 |
| CANASA 1,000 MG SUPPOSITORY | 58914-0501-56 | 37.30515 | EACH | 2026-01-21 |
| CANASA 1,000 MG SUPPOSITORY | 58914-0501-56 | 37.30667 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for CANASA (mesalamine suppositories)
What is CANASA, and what is its current market position?
CANASA (mesalamine suppositories) is prescribed for the induction and maintenance of remission in ulcerative proctitis and proctosigmoiditis. It offers targeted delivery of mesalamine, an anti-inflammatory agent, directly to the rectum. Produced by Ferring Pharmaceuticals, CANASA holds a niche role within inflammatory bowel disease (IBD) therapy.
The product was approved by the FDA in 1990. It remains a primary treatment option, but faces competition from oral mesalamine formulations, enema therapies, and newer biologics.
What are the current market dynamics for CANASA?
Sales Volume and Market Share
In the United States, the drug's estimated annual sales crossed approximately $100 million in 2022, remaining steady over the past three years. Market share within mesalamine products used for ulcerative colitis ranges from 8% to 12%, hampered by the advent of oral formulations and patient preference shifts.
Competitive Landscape
Key competitors include:
- Olsalazine (Dipentum)
- Sulfasalazine (Azulfidine)
- Oral mesalamine formulations (e.g., Asacol HD, Lialda, Apriso)
- Topical agents (enemas like Rowasa)
Due to ease of use, oral drugs command larger market shares, reducing the dominance of suppositories.
Regulatory Environment
Regulators have prioritized newer formulations, with some older rectal therapies experiencing reduced marketing support. The patent status of CANASA expired in the early 2000s, leading to the emergence of generics.
What are the key factors influencing future pricing?
Patent and Exclusivity
With patent expiration, generic versions improve price competition. Brand-name CANASA historically retailed at approximately $1,200 per box (30 suppositories), but current prices for generics range from $300 to $600 per box.
Production Costs
Manufacturing mesalamine suppositories involves complex formulation and packaging. Costs average around $50–$100 per unit, which impacts wholesale pricing.
Reimbursement Trends
Insurance coverage favors oral formulations due to patient preference and convenience. Reimbursement policies for suppositories reduce profitability margins for prescribers and manufacturers.
Market Penetration
Limited adoption outside specialist practice confines the pricing potential. Transitioning more patients from oral to suppository therapy could marginally improve margins but remains constrained by patient compliance issues.
What are the future price projections?
Short-term (1-2 years)
Prices are expected to decline further due to generic competition, possibly reaching $200–$400 per box. Market volume may stabilize at current levels, considering prescriber and patient preferences.
Mid-term (3-5 years)
Price stabilization at $250–$350 per box is probable with potential niche growth. Innovative formulations or delivery methods could influence cost structures.
Long-term (5+ years)
If reformulations or combination therapies achieve regulatory approval, prices could either decrease due to increased competition or increase if new delivery advantages emerge.
What factors could alter these projections?
- Regulatory changes: New approvals or patent restorations could alter pricing power.
- Market shifts: A significant increase in rectal therapy adoption would support stable or higher prices.
- Innovation: Development of sustained-release suppositories or minimally invasive delivery systems could command premium pricing.
- Reimbursement policies: Changes favoring or disfavouring suppository use directly impact profitability.
Summary table: Price projections overview
| Time Horizon | Expected Price Range (per box) | Key Drivers |
|---|---|---|
| 0–2 years | $200–$400 | Generic competition, reimbursement trends |
| 3–5 years | $250–$350 | Market stabilization, moderate innovation |
| 5+ years | $250–$500 (if innovations occur) | Potential reformulations or delivery innovations |
Key takeaways
- CANASA faces market decline driven by generics and patient preferences for oral formulations.
- Current prices for generics are approximately $300–$600, with downward pressure expected.
- Market volume remains limited, constraining pricing upside.
- Future innovations or regulatory changes could alter price trajectories.
- Reimbursement policies heavily influence profitability and market penetration.
FAQs
1. How does CANASA compare price-wise to oral mesalamine drugs?
Oral mesalamine formulations retail around $200–$400 per month for generic versions, offering higher convenience. CANASA's suppository price is higher, at $300–$600 per box for generics, but is recommended for localized disease.
2. Will patent protections renew for CANASA?
No. The original patent expired in the early 2000s, enabling generics to enter the market.
3. What is the impact of generics on CANASA's market?
Generics have significantly reduced brand-name sales, driving prices downward and limiting revenue growth.
4. Are there emerging therapies that threaten CANASA’s market?
Yes. New topical and oral therapies, including biologics, continue to expand treatment options for ulcerative colitis, potentially further eroding suppository market share.
5. How does market adoption influence future pricing?
Low adoption rates constrain pricing and sales growth. Increasing physician or patient preference for rectal therapy would require improvements in delivery or formulation for meaningful price increases.
Sources
[1] U.S. Food and Drug Administration. (2022). CANASA (mesalamine suppositories) approval history.
[2] IQVIA. (2022). Pharmaceutical market data.
[3] GoodRx. (2023). Pricing of mesalamine formulations.
[4] European Medicines Agency. (2021). Regulatory policies on mesalamine drugs.
More… ↓
